MOFA+ analysis - WM, edgeR library sizes
Will Macnair
Neurogenomics, Neuroscience and Rare Diseases, RocheMarch 24, 2022
Last updated: 2022-03-24
Checks: 4 3
Knit directory: MS_lesions/
This reproducible R Markdown analysis was created with workflowr (version 1.7.0). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
The R Markdown file has unstaged changes. To know which version of the R Markdown file created these results, you’ll want to first commit it to the Git repo. If you’re still working on the analysis, you can ignore this warning. When you’re finished, you can run wflow_publish to commit the R Markdown file and build the HTML.
The global environment had objects present when the code in the R Markdown file was run. These objects can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment. Use wflow_publish or wflow_build to ensure that the code is always run in an empty environment.
The following objects were defined in the global environment when these results were created:
| Name | Class | Size |
|---|---|---|
| q | function | 1008 bytes |
The command set.seed(20210118) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
- setup_input
- setup_outputs
To ensure reproducibility of the results, delete the cache directory ms15_mofa_wm_edger_libs_cache and re-run the analysis. To have workflowr automatically delete the cache directory prior to building the file, set delete_cache = TRUE when running wflow_build() or wflow_publish().
Great job! Using relative paths to the files within your workflowr project makes it easier to run your code on other machines.
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version 9175fdc. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .DS_Store
Ignored: .RData
Ignored: .Rhistory
Ignored: .Rprofile
Ignored: .Rproj.user/
Ignored: ._F12_wm_chk.png
Ignored: ._MS_lesions.sublime-project
Ignored: ._fxs_genes.png
Ignored: .log/
Ignored: MS_lesions.sublime-project
Ignored: MS_lesions.sublime-workspace
Ignored: analysis/.__site.yml
Ignored: analysis/fig_muscat_cache/
Ignored: analysis/ms00_manuscript_figures_cache/
Ignored: analysis/ms02_doublet_id_cache/
Ignored: analysis/ms03_SampleQC_cache/
Ignored: analysis/ms03_SampleQC_summary_cache/
Ignored: analysis/ms04_conos_cache/
Ignored: analysis/ms05_splitting_cache/
Ignored: analysis/ms06_sccaf_cache/
Ignored: analysis/ms07_soup_cache/
Ignored: analysis/ms08_modules_cache/
Ignored: analysis/ms08_modules_pseudobulk_cache/
Ignored: analysis/ms09_ancombc_cache/
Ignored: analysis/ms09_ancombc_clean_1e3_cache/
Ignored: analysis/ms09_ancombc_clean_2e3_cache/
Ignored: analysis/ms09_ancombc_mixed_cache/
Ignored: analysis/ms10_muscat_run01_cache/
Ignored: analysis/ms10_muscat_run02_cache/
Ignored: analysis/ms10_muscat_template_broad_slim_cache/
Ignored: analysis/ms10_muscat_template_fine_slim_cache/
Ignored: analysis/ms11_paga_cache/
Ignored: analysis/ms11_paga_recalc_cache/
Ignored: analysis/ms11_paga_superclean_cache/
Ignored: analysis/ms12_markers_cache/
Ignored: analysis/ms13_labelling_cache/
Ignored: analysis/ms14_lesions_cache/
Ignored: analysis/ms15_mofa_gm_cache/
Ignored: analysis/ms15_mofa_gm_edger_libs_cache/
Ignored: analysis/ms15_mofa_heatmaps_cache/
Ignored: analysis/ms15_mofa_sample_gm_cache/
Ignored: analysis/ms15_mofa_sample_gm_final_meta_cache/
Ignored: analysis/ms15_mofa_sample_gm_superclean_cache/
Ignored: analysis/ms15_mofa_sample_gm_w_layers_final_meta_cache/
Ignored: analysis/ms15_mofa_sample_wm_cache/
Ignored: analysis/ms15_mofa_sample_wm_final_meta_bigger_cache/
Ignored: analysis/ms15_mofa_sample_wm_final_meta_bigger_edger_libsizes_cache/
Ignored: analysis/ms15_mofa_sample_wm_final_meta_cache/
Ignored: analysis/ms15_mofa_sample_wm_final_meta_edger_libsizes_cache/
Ignored: analysis/ms15_mofa_sample_wm_final_meta_neuro_ok_cache/
Ignored: analysis/ms15_mofa_sample_wm_new_meta_cache/
Ignored: analysis/ms15_mofa_sample_wm_superclean_cache/
Ignored: analysis/ms15_mofa_wm_bigger_cache/
Ignored: analysis/ms15_mofa_wm_cache/
Ignored: analysis/ms15_mofa_wm_coding_only_bigger_cache/
Ignored: analysis/ms15_mofa_wm_coding_only_cache/
Ignored: analysis/ms15_mofa_wm_edger_libs_cache/
Ignored: analysis/ms15_mofa_wm_neuro_ok_cache/
Ignored: analysis/ms15_mofa_wm_pearson_cache/
Ignored: analysis/ms15_patients_cache/
Ignored: analysis/ms15_patients_gm_cache/
Ignored: analysis/ms15_patients_sample_level_cache/
Ignored: analysis/ms15_patients_w_ms_cache/
Ignored: analysis/ms99_deg_figures_gm_cache/
Ignored: analysis/ms99_deg_figures_wm_cache/
Ignored: analysis/ms99_manuscript_figures_cache/
Ignored: analysis/ms99_test_cache/
Ignored: analysis/public/
Ignored: analysis/supp06_sccaf_cache/
Ignored: analysis/supp07_superclean_check_cache/
Ignored: analysis/supp09_ancombc_cache/
Ignored: analysis/supp09_ancombc_mixed_cache/
Ignored: analysis/supp09_ancombc_rowitch_cache/
Ignored: analysis/supp09_ancombc_superclean_cache/
Ignored: analysis/supp10_muscat_cache/
Ignored: analysis/supp10_muscat_ctrl_gm_vs_wm_cache/
Ignored: analysis/supp10_muscat_gm_layers_effects_cache/
Ignored: analysis/supp10_muscat_gsea_cache/
Ignored: analysis/supp10_muscat_heatmaps_cache/
Ignored: analysis/supp10_muscat_olg_pc1_cache/
Ignored: analysis/supp10_muscat_olg_pc2_cache/
Ignored: analysis/supp10_muscat_olg_pc_cache/
Ignored: analysis/supp10_muscat_regression_cache/
Ignored: analysis/supp10_muscat_soup_cache/
Ignored: analysis/supp10_muscat_soup_mito_cache/
Ignored: code/._ms10_muscat_fns_recover.R
Ignored: code/.recovery/
Ignored: code/adhoc_code/
Ignored: code/jobs/._muscat_run09_2021-10-11.slurm
Ignored: data/
Ignored: figures/
Ignored: output/
Ignored: tmp/
Untracked files:
Untracked: %
Untracked: check_absolutes.png
Untracked: wm_vs_gm.png
Unstaged changes:
Modified: analysis/ms09_ancombc_mixed.Rmd
Modified: analysis/ms15_mofa_gm_edger_libs.Rmd
Modified: analysis/ms15_mofa_wm_edger_libs.Rmd
Modified: code/ms00_utils.R
Modified: code/ms09_ancombc_mixed.R
Modified: code/ms15_mofa.R
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
These are the previous versions of the repository in which changes were made to the R Markdown (analysis/ms15_mofa_wm_edger_libs.Rmd) and HTML (public/ms15_mofa_wm_edger_libs.html) files. If you’ve configured a remote Git repository (see ?wflow_git_remote), click on the hyperlinks in the table below to view the files as they were in that past version.
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | b677f1a | Macnair | 2022-03-23 | Add “already done?” check to MOFA rmd |
| Rmd | 91ab359 | Will Macnair | 2022-03-23 | Tweak MOFA analysis |
| html | 91ab359 | Will Macnair | 2022-03-23 | Tweak MOFA analysis |
| Rmd | 7c17d96 | Macnair | 2022-03-18 | Add various MOFA run outputs |
| html | 7c17d96 | Macnair | 2022-03-18 | Add various MOFA run outputs |
| Rmd | 74935aa | wmacnair | 2022-03-06 | Fix MOFA WM, update some plots |
| html | 74935aa | wmacnair | 2022-03-06 | Fix MOFA WM, update some plots |
Setup / definitions
Libraries
Helper functions
source('code/ms00_utils.R')
source('code/ms08_modules.R')
source('code/ms09_ancombc.R')
source('code/ms10_muscat_runs.R')
source('code/ms15_mofa.R')
n_cores = 8
setDTthreads(n_cores)Inputs
# specify what goes into muscat run
meta_f = "data/metadata/metadata_checked_assumptions_2022-03-22.xlsx"
olg_grps_f = 'data/metadata/oligo_groupings.txt'
labels_f = 'data/byhand_markers/validation_markers_2021-05-31.csv'
labelled_f = 'output/ms13_labelling/conos_labelled_2021-05-31.txt.gz'
pb_f = file.path(soup_dir, 'pb_sum_broad_2021-10-11.rds')
pb_fine_f = file.path(soup_dir, 'pb_sum_fine_2021-10-11.rds')
soup_f = 'data/ambient/ambient.100UMI.txt'
# file with gene biotypes
gtf_f = 'data/gtf/Homo_sapiens.GRCh38.96.filtered.preMRNA.gtf'
# get summary QC metrics for each sample
qc_dir = "output/ms03_SampleQC"
qc_f = file.path(qc_dir, "ms_qc_dt.txt")
# define run to load
run_tag = 'run09'
time_stamp = '2021-10-13'
# define files
model_dir = file.path('output/ms10_muscat', run_tag)
muscat_f = '%s/muscat_res_dt_%s_%s.txt.gz' %>%
sprintf(model_dir, run_tag, time_stamp)
anova_f = '%s/muscat_goodness_dt_%s_%s.txt.gz' %>%
sprintf(model_dir, run_tag, time_stamp)
params_f = '%s/muscat_params_%s_%s.rds' %>%
sprintf(model_dir, run_tag, time_stamp)
ranef_dt_f = sprintf('%s/muscat_ranef_dt_%s_%s.txt.gz',
model_dir, run_tag, time_stamp)
mds_sep_f = sprintf('%s/mds_sep_dt_%s_%s.txt.gz',
model_dir, run_tag, time_stamp)Outputs
# where to save
save_dir = 'output/ms15_mofa'
date_tag = '2022-03-03_edger'
if (!dir.exists(save_dir))
dir.create(save_dir)
# file for summary of QC metrics
qc_stats_f = sprintf('%s/qc_stats_by_sample_%s.txt', save_dir, date_tag)
# parameters for gene selection
min_sd = log(2)
min_fc = log(2)
max_p = 0.01
n_factors = 5
sel_cl = c("OPCs / COPs", "Oligodendrocytes", "Astrocytes", "Microglia",
"Endothelial cells", "Pericytes", "Immune")
fgsea_cut = 0.1
sel_ps = c('go_bp', 'go_cc', 'go_mf', 'hallmark', 'kegg')
log_p_mad = 2
n_paths = 50
# parameters for plotting
min_var = 5
w_cut = 0.2
# checking if metadata can explain factors
formula_str = '~ lesion_type + sex + age_scale + pmi_cat'
random_var = 'subject_orig'
# restriction to protein-coding genes only for expression heatmaps
ok_types = c('protein_coding', 'IG_V_gene', 'IG_C_gene', 'IG_J_gene',
'TR_C_gene', 'TR_J_gene', 'TR_V_gene', 'TR_D_gene')
# output files
mofa_f = sprintf('%s/mofa_%s_%s.hdf5', save_dir, run_tag, date_tag)
fgsea_pat = sprintf('%s/mofa_fgsea_%s_%s_%s.txt',
save_dir, run_tag, '%s', date_tag)
interesting_f = sprintf('%s/mofa_interesting_genes_%s_%s.xlsx',
save_dir, run_tag, date_tag)
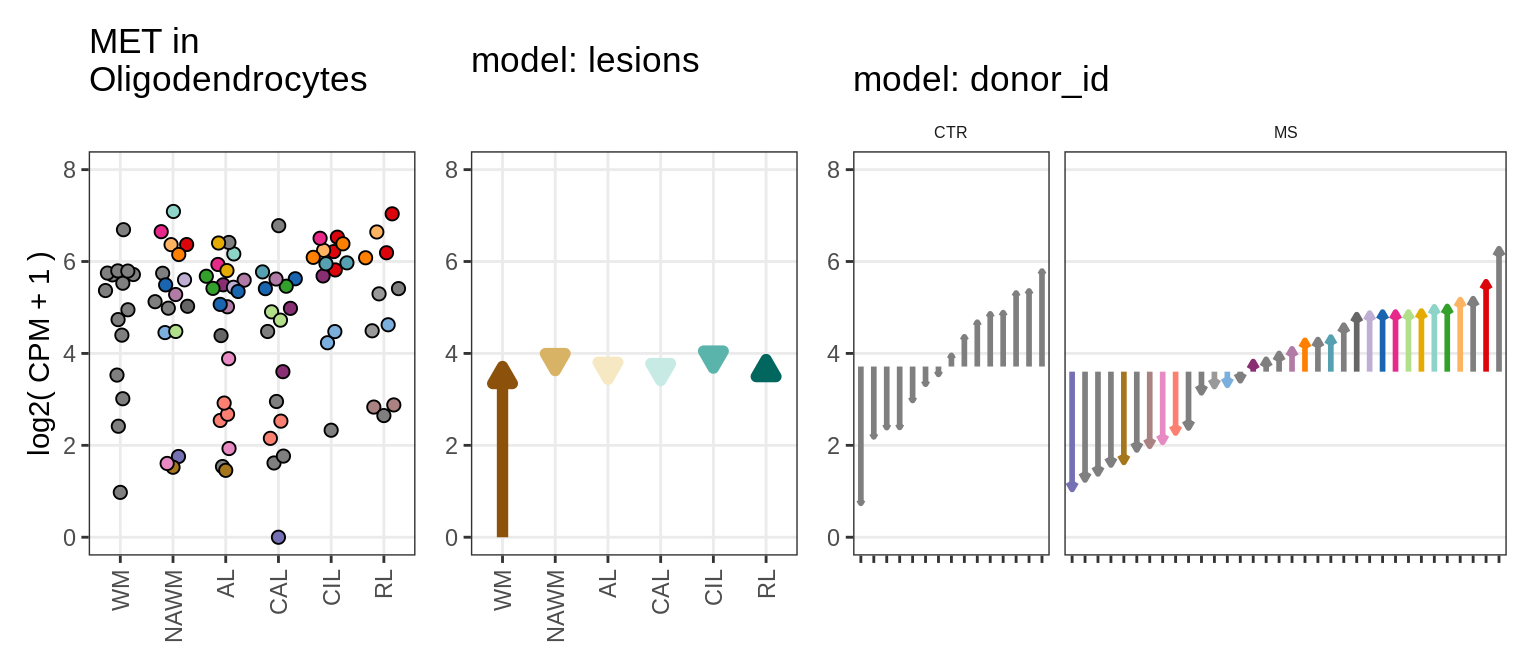
# what to use to illustrate random effects concept?
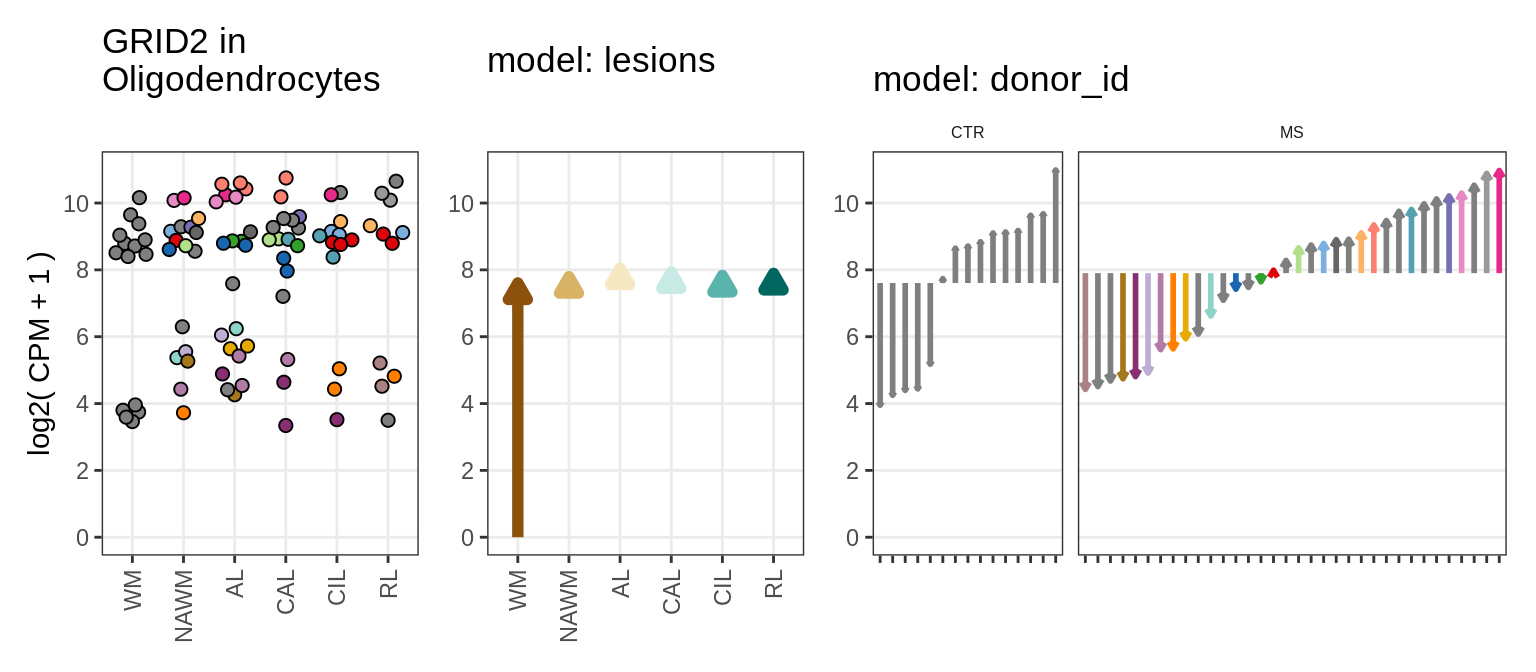
example_cl = 'Oligodendrocytes'
example_gs = c("NHLH1_ENSG00000171786", "CASP7_ENSG00000165806",
"RELN_ENSG00000189056", "KLB_ENSG00000134962", "NRTN_ENSG00000171119",
"EVI5L_ENSG00000142459", "PWP2_ENSG00000241945", "GRID2_ENSG00000152208",
"MET_ENSG00000105976")Load inputs
# load parameters
params = params_f %>% readRDS
# load pseudobulk object
pb = readRDS(params$pb_f) %>% .subset_pb(params$subset_spec) %>%
subset_pb_celltypes(sel_cl) subsetting pb object restricting to samples that meet subset criteria updating factors to remove levels no longer observed# check for any massive outliers
outliers_dt = calc_log_prop_outliers(pb, mad_cut = log_p_mad)no samples have half or more of celltypes with very extreme (2 > MADs)
log proportionsok_samples = outliers_dt[ props_ok == TRUE ]$sample_id
pb = pb[ , ok_samples ]
# load other useful things
labels_dt = .load_labels_dt(labels_f, params$cluster_var)Warning in FUN(X[[i]], ...): unable to translate '<U+00C4>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00D6>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00DC>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00E4>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00F6>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00FC>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00DF>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00C6>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00E6>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00D8>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00F8>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00C5>' to native encodingWarning in FUN(X[[i]], ...): unable to translate '<U+00E5>' to native encodingmagma_dt = .load_magma_dt(magma_f, pb)
tfs_dt = .load_tfs_dt(tfs_f, pb)
lof_dt = .load_lof_dt(lof_f, pb)
ok_genes = get_biotypes_dt(gtf_f) %>%
.[ gene_biotype %in% ok_types ] %>%
use_series('gene_id') %>% str_extract('^[^_]+')
assert_that( all(ok_genes %in% rowData(pb)$symbol) )[1] TRUE# load annotations
annots_dt = .get_cols_dt(pb) %>%
.[, sample := sample_id ] %>% .[, group := 'single_group'] %>%
.[, .(sample, sample_id, diagnosis, lesion_type, subject_id, subject_orig,
sample_source, batch = seq_pool, age = age_at_death, age_at_death, age_scale,
years_w_ms, sex, pmi_cat, pmi_cat2, smoker )] %>%
add_oligo_groups(olg_grps_f)
# get random effects
ranef_dt = .load_ranef_dt(ranef_dt_f, labels_dt, pb)
# get results
res_dt = muscat_f %>% fread %>%
.load_muscat_results(labels_dt, params) %>%
.[, .(cluster_id, gene_id, symbol, var_type, coef, test_var,
logCPM, mean_soup, padj = p_adj.soup, logFC)] %>%
.[ !is.na(padj) ]
# get anova results
anova_dt = .load_anova_dt(anova_f, res_dt) %>%
.[ is.na(full), full := 1 ]
# get MDS outputs
mds_sep_dt = mds_sep_f %>% fread
if (params$cluster_var == 'type_broad')
mds_sep_dt[, cluster_id := factor(cluster_id, levels = broad_ord)]# get random effects
sd_dt = ranef_dt %>% calc_ranef_melt %>% calc_sd_dt
filter_dt = calc_filter_dt(res_dt, sd_dt, pb, anova_dt,
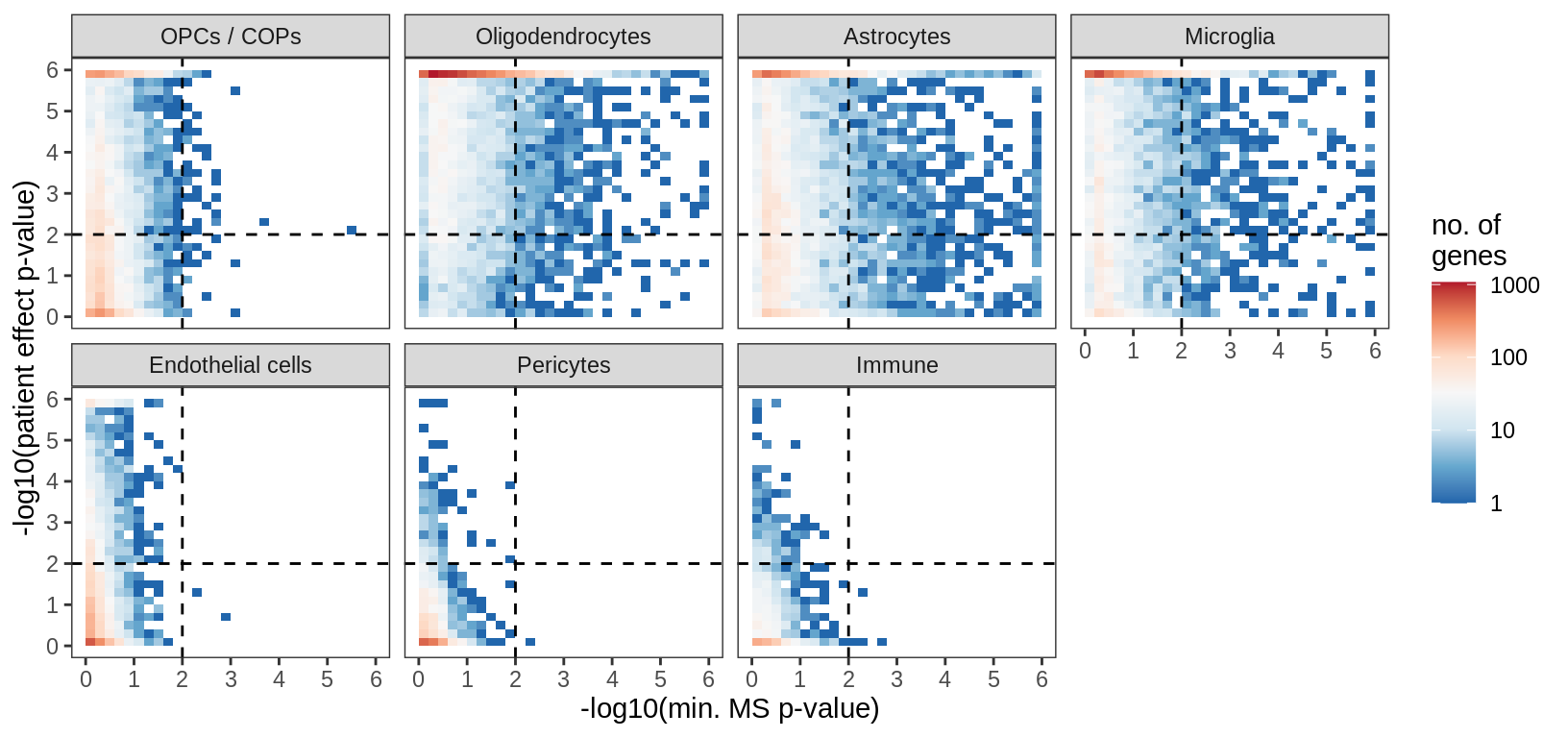
max_p = max_p, min_sd = min_sd, min_fc = min_fc)
filtered_dt = filter_dt[ ( (ms_signif == 'signif') & (ms_effect == 'big') ) |
( (pt_signif == 'signif') & (pt_variab == 'variable')) ] %>%
.[ cluster_id %in% sel_cl ] %>%
.[, is_ms := ifelse(ms_effect == "big" & ms_signif == "signif", "ms", "not") ] %>%
.[, is_pt := ifelse(pt_signif == "signif" & pt_variab == "variable", "pt", "not") ]
# check what we've got
filtered_dt[, .N, by = .(cluster_id, is_ms, is_pt)] %>%
.[, total := sum( N ), by = cluster_id ] %>%
dcast.data.table(cluster_id + total ~ is_ms + is_pt, fill = 0, value.var = "N") cluster_id total ms_not ms_pt not_pt
1: OPCs / COPs 97 20 2 75
2: Oligodendrocytes 507 259 16 232
3: Astrocytes 794 559 29 206
4: Microglia 667 255 48 364
5: Endothelial cells 86 2 0 84
6: Pericytes 27 1 0 26
7: Immune 27 4 0 23n_cells_dt = calc_n_cells_dt(pb_fine_f, annots_dt, sel_cl)soup_dt = get_soup_logcpms(soup_f, pb)qc_stats = calc_qc_stats_by_sample(qc_stats_f, qc_dir, qc_f,
meta_f, labels_f, labelled_f)Processing / calculations
mofa_obj = make_mofa_obj_samples(pb, filtered_dt, sel_cl,
lib_size_method = 'edger')Removing 5430 rows with all zero countsRemoving 3463 rows with all zero countsRemoving 3766 rows with all zero countsRemoving 4578 rows with all zero countsRemoving 6797 rows with all zero countsRemoving 8883 rows with all zero countsRemoving 8781 rows with all zero countsCreating MOFA object from a data.frame...if (file.exists(mofa_f)) {
model = load_model(mofa_f)
} else {
# set up
data_opts = get_default_data_options(mofa_obj)
model_opts = get_default_model_options(mofa_obj)
train_opts = get_default_training_options(mofa_obj)
# specify how many factors
model_opts$num_factors = n_factors
# train mofa
mofa_obj = prepare_mofa(
object = mofa_obj,
data_options = data_opts,
model_options = model_opts,
training_options = train_opts
)
model = run_mofa(mofa_obj, mofa_f)
}Warning in .quality_control(object, verbose = verbose): Factor(s) 1 are strongly correlated with the total number of expressed features for at least one of your omics. Such factors appear when there are differences in the total 'levels' between your samples, *sometimes* because of poor normalisation in the preprocessing steps.# add metadata
model = add_metadata(model, annots_dt)
# put weights and scores in MS order
model = put_model_in_ms_order(model)var_exp_dt = get_variance_explained(model, as.data.frame = TRUE) %>%
as.data.table %>%
.[, .(
view = r2_per_factor.view %>% factor(levels = broad_short),
factor = r2_per_factor.factor,
var_exp = r2_per_factor.value
)]
to_plot_dt = var_exp_dt[ var_exp > min_var ] %>% .[order(factor, -var_exp)]
var_exp_no_endoperi = var_exp_dt[ !(view %in% c('endo', 'peri')) ]# get weights, define expected files
ws_dt = extract_weights(model, sd_dt)
fgsea_fs = sapply(sel_ps, function(p) sprintf(fgsea_pat, p))
# if necessary, run FGSEA
if (all(file.exists(fgsea_fs))) {
gsea_list = lapply(fgsea_fs, fread)
} else {
# do fgsea for these
bpparam = MulticoreParam(workers = n_cores,
progressbar = TRUE, tasks = 50)
bpstart()
gsea_list = calc_mofa_fgsea(paths_list[ sel_ps ], ws_dt, fgsea_pat, fgsea_cut, bpparam)
bpstop()
}
# restrict to interesting ones
gsea_main = gsea_list %>% map( ~.x[ main_path == TRUE ]) %>% rbindlistr2_dt = calc_r2_for_factors(model, annots_dt, formula_str, random_var)
anova_dt = calc_lrts(model, annots_dt, formula_str, random_var)Analysis
muscat results vs SD
for (what in c('log10_padj', 'log2FC')) {
cat('### ', what, '\n', sep = '')
print(plot_muscat_vs_sd(res_dt, sd_dt, NULL, what = what))
cat('\n\n')
}log10_padj

Cytokine effects
cyto_gs = unique(res_dt$gene_id) %>% str_subset('(^IL[0-9]+|^CCL|^CXCL|^IFN|^TGF|^TNF|^CSF)')
(plot_muscat_vs_sd_min(res_dt[ gene_id %in% cyto_gs ], sd_dt[ gene_id %in% cyto_gs ],
sel_cl, min_sd, max_p, do_labels = TRUE))Ages vs duration of MS
(plot_age_duration(annots_dt))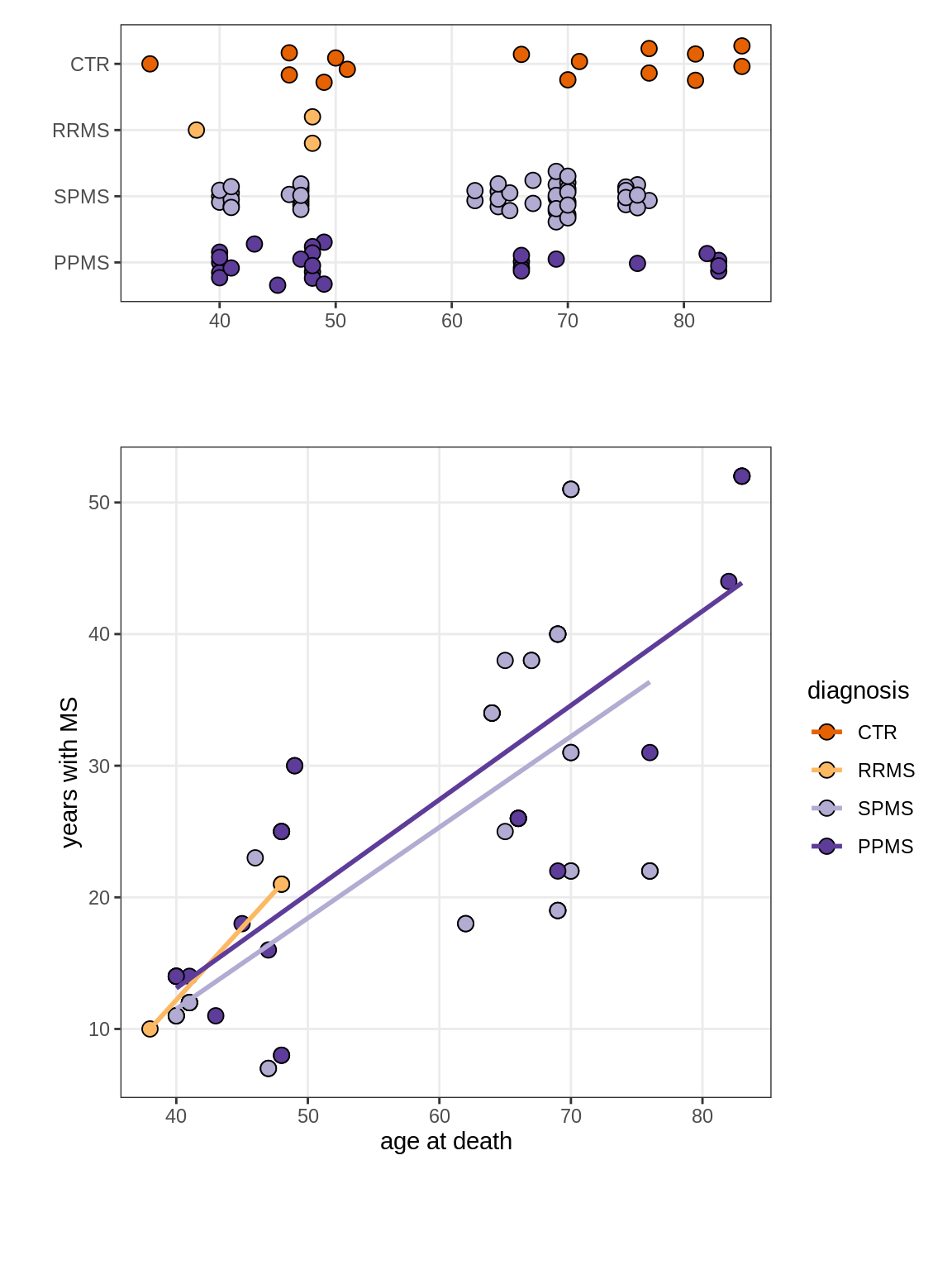
Data overview
(plot_data_overview(mofa_obj))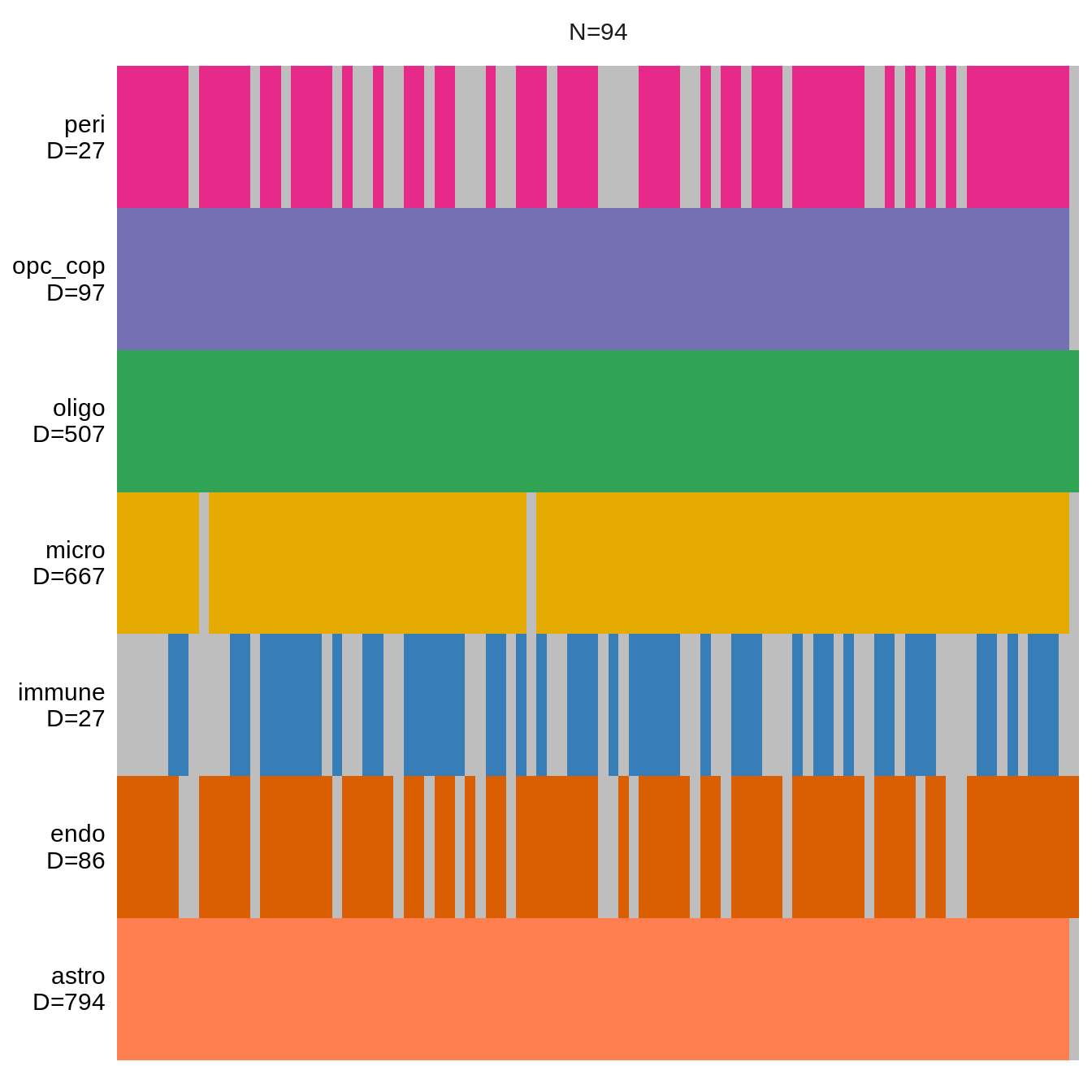
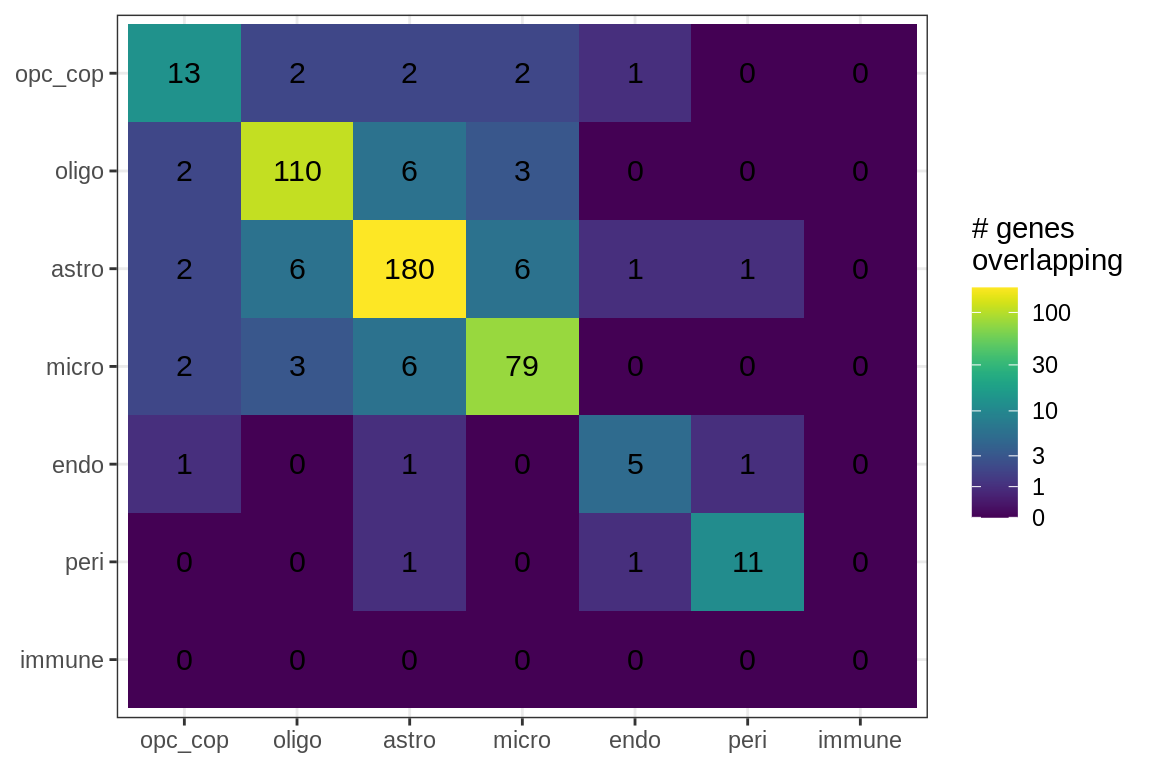
Overlapping genes
cat('### All genes\n')All genes
suppressWarnings(print(plot_gene_overlap(model)))
cat('\n\n')for (sel_f in factors_names(model)) {
cat('### Genes in ', sel_f, '\n', sep = '')
suppressWarnings(print(plot_gene_overlap(model, sel_f = sel_f, w_cut = w_cut)))
cat('\n\n')
}Genes in Factor1
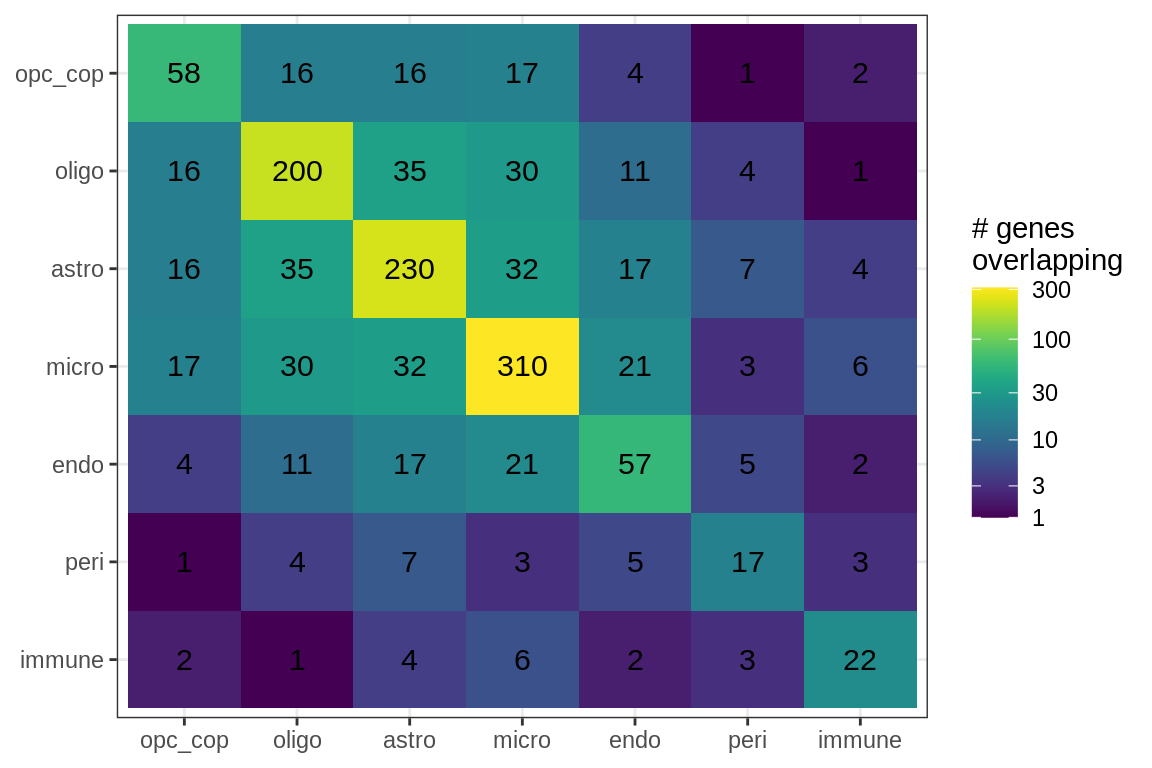
Genes in Factor2

Genes in Factor3
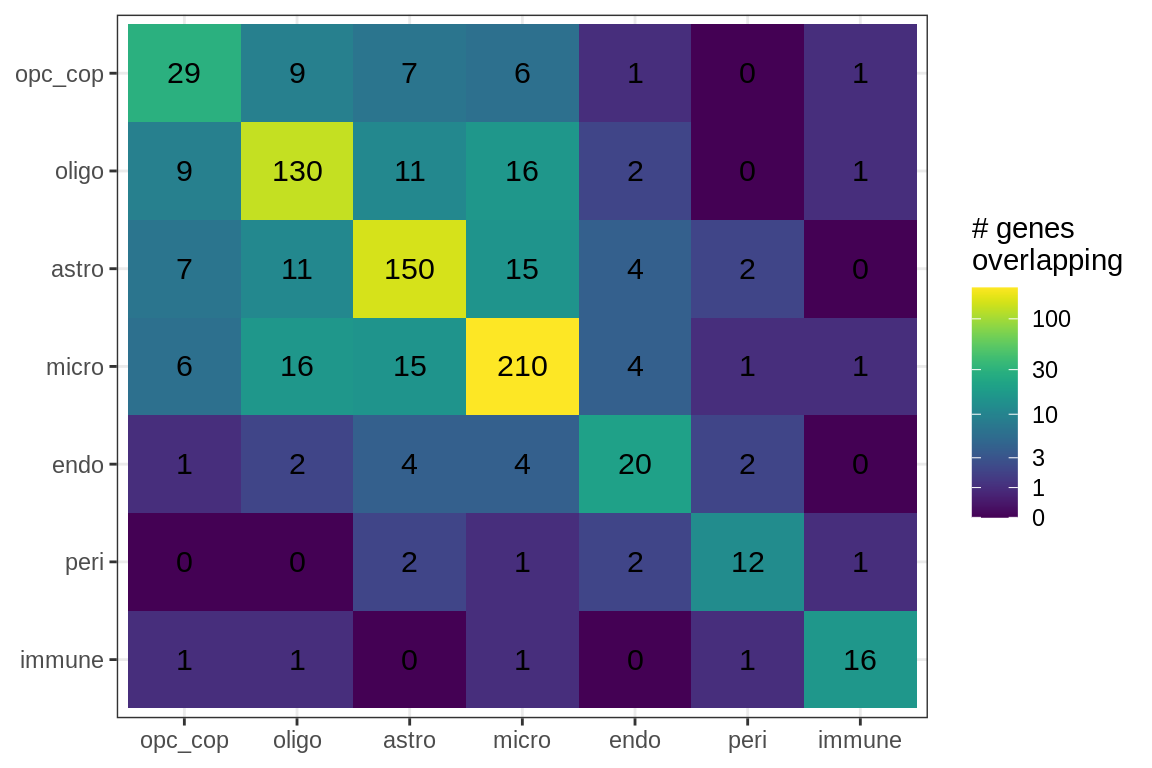
Genes in Factor4
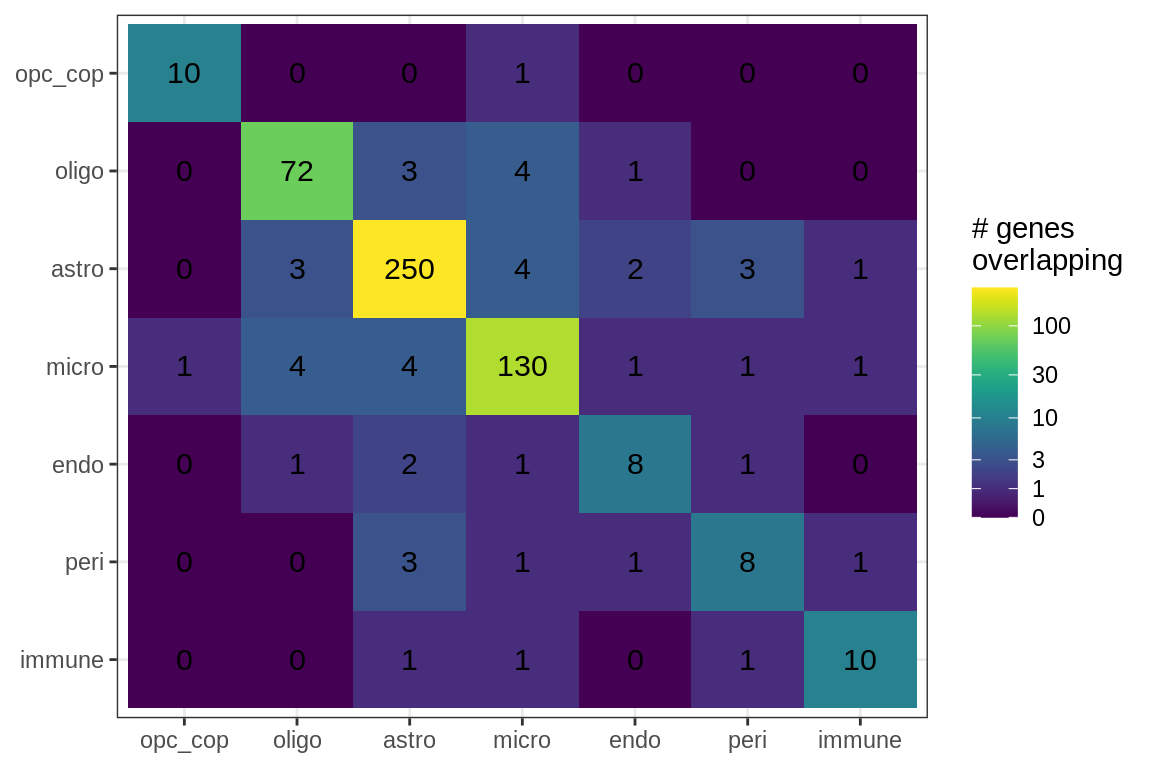
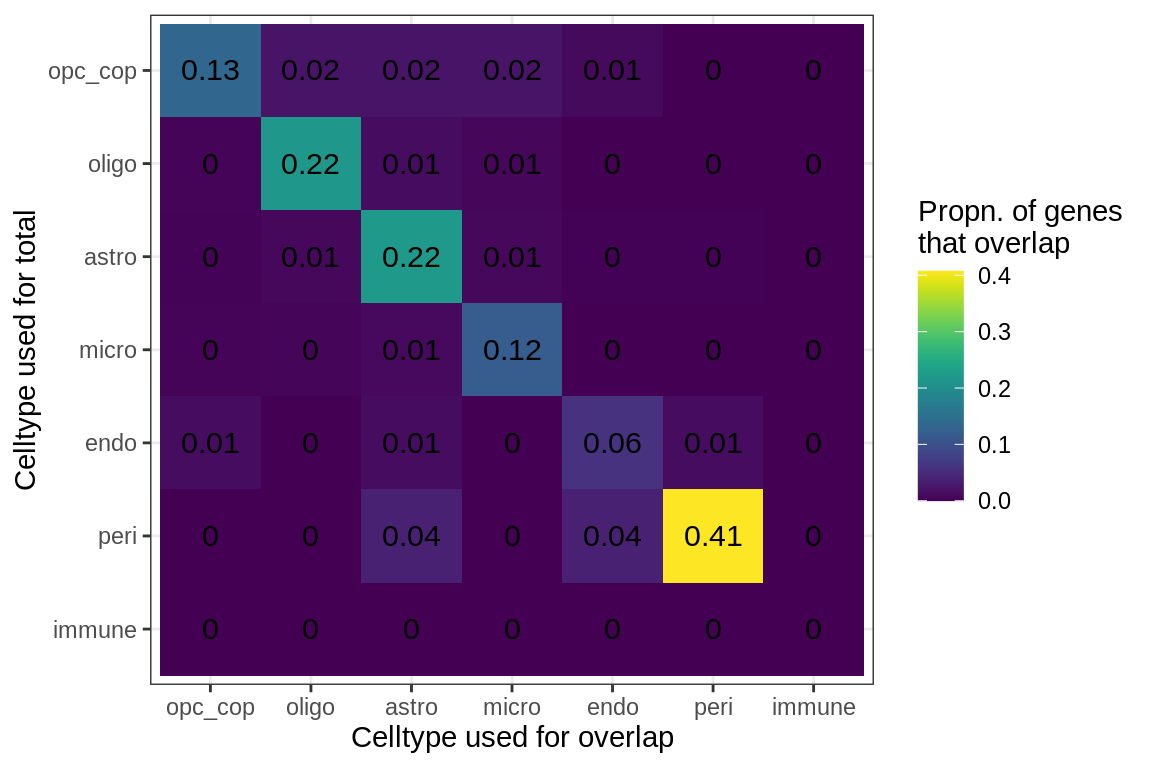
Overlapping genes (proportions)
cat('### All genes\n')All genes
suppressWarnings(print(plot_gene_overlap(model, what = 'prop')))
cat('\n\n')for (sel_f in factors_names(model)) {
cat('### Genes in ', sel_f, '\n', sep = '')
suppressWarnings(print(plot_gene_overlap(model, what = 'prop',
sel_f = sel_f, w_cut = w_cut)))
cat('\n\n')
}Genes in Factor1

Genes in Factor2

Genes in Factor3
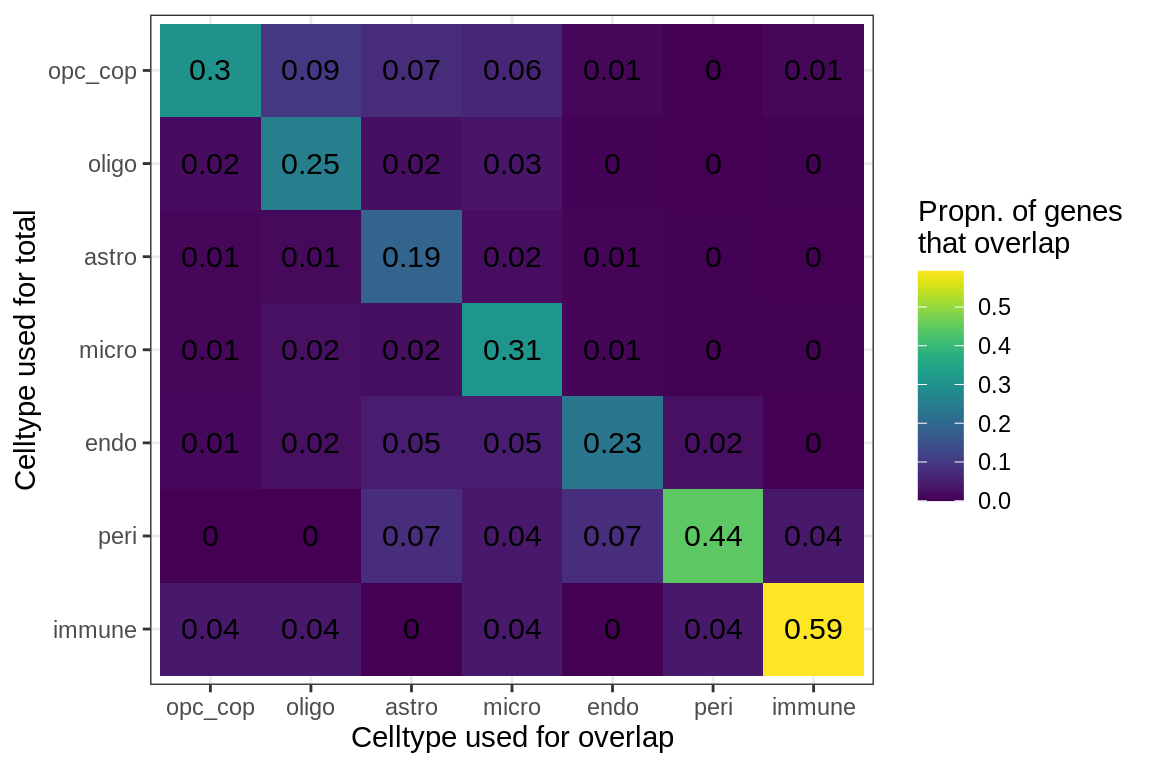
Genes in Factor4

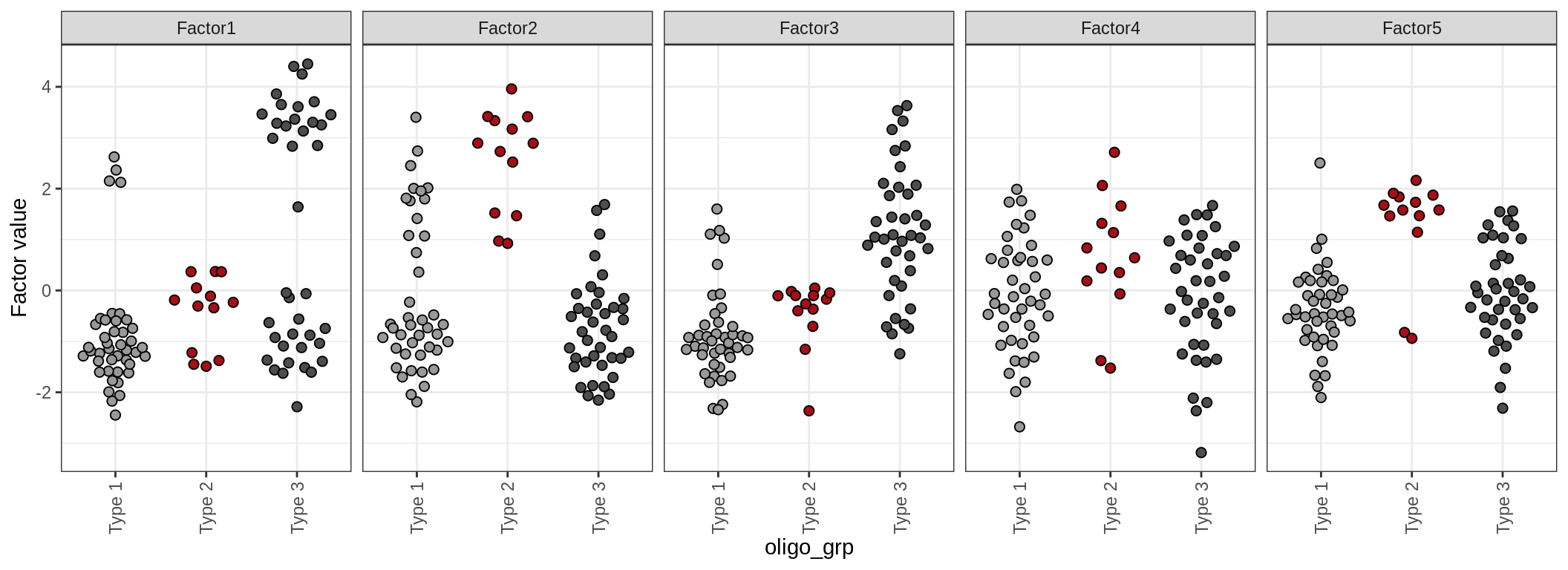
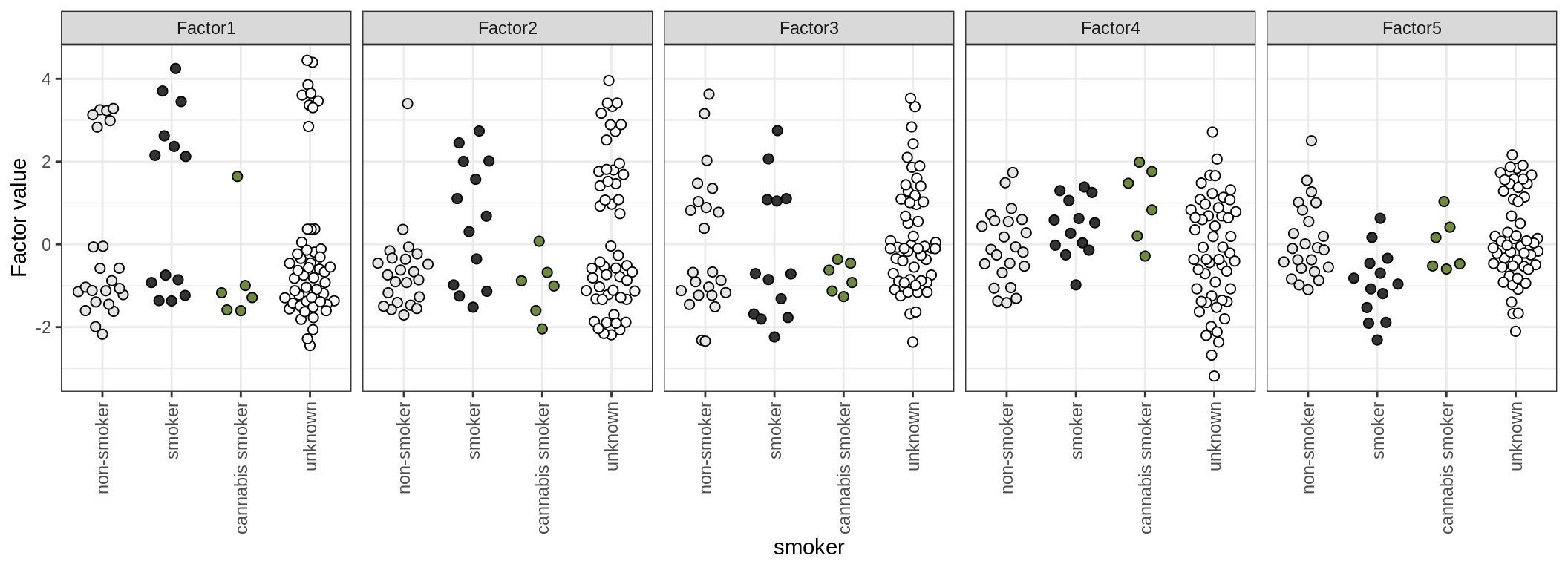
Factor distributions
for (annot in c('lesion_type', 'diagnosis', 'sex', 'sample_source', 'smoker', 'oligo_grp')) {
cat('### by ', annot, '\n', sep = '')
print(plot_factors_univariate(model, annots_dt, pb, by = annot))
cat('\n\n')
}by lesion_type

by diagnosis

by sex

by sample_source
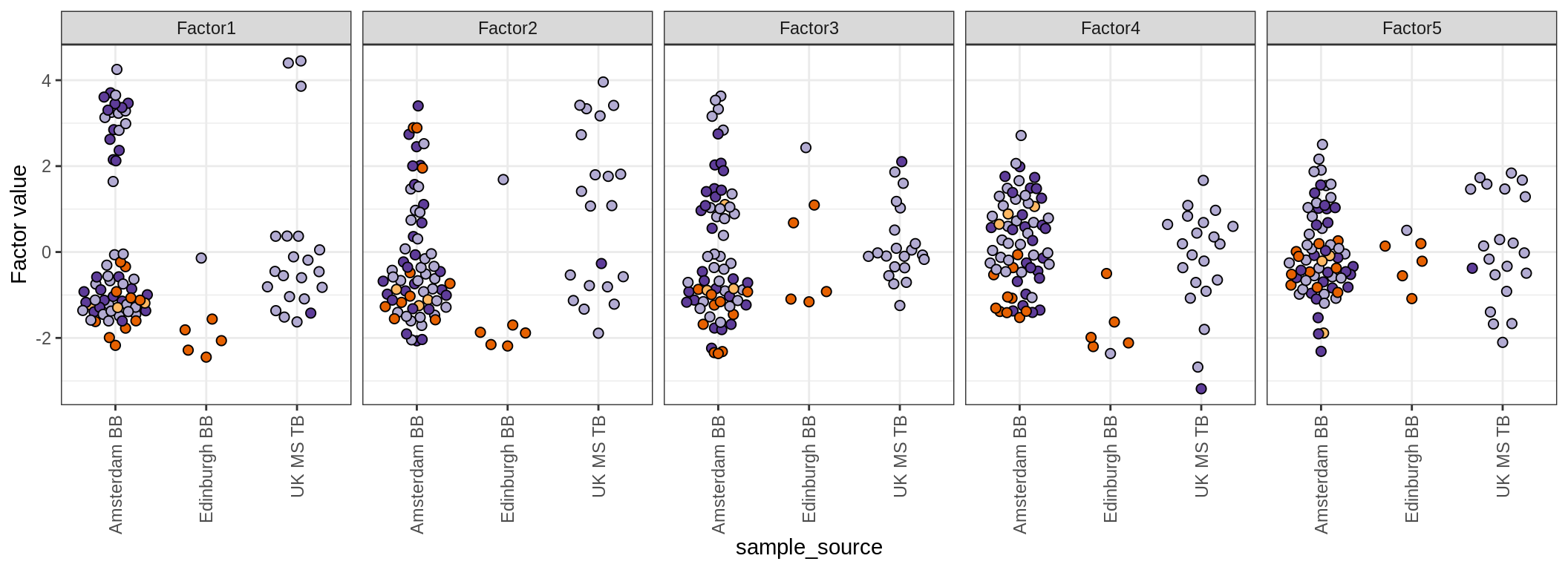
by smoker
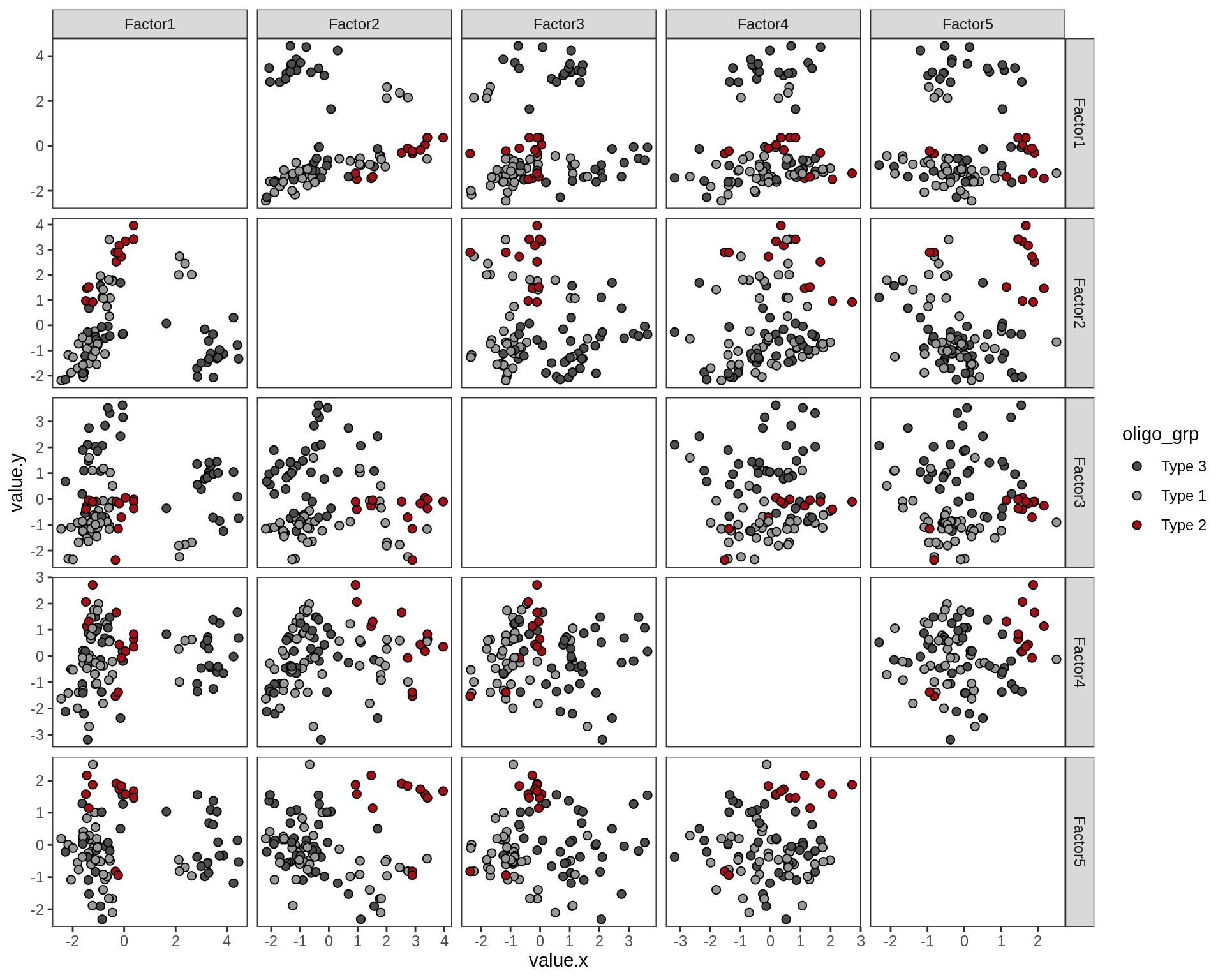
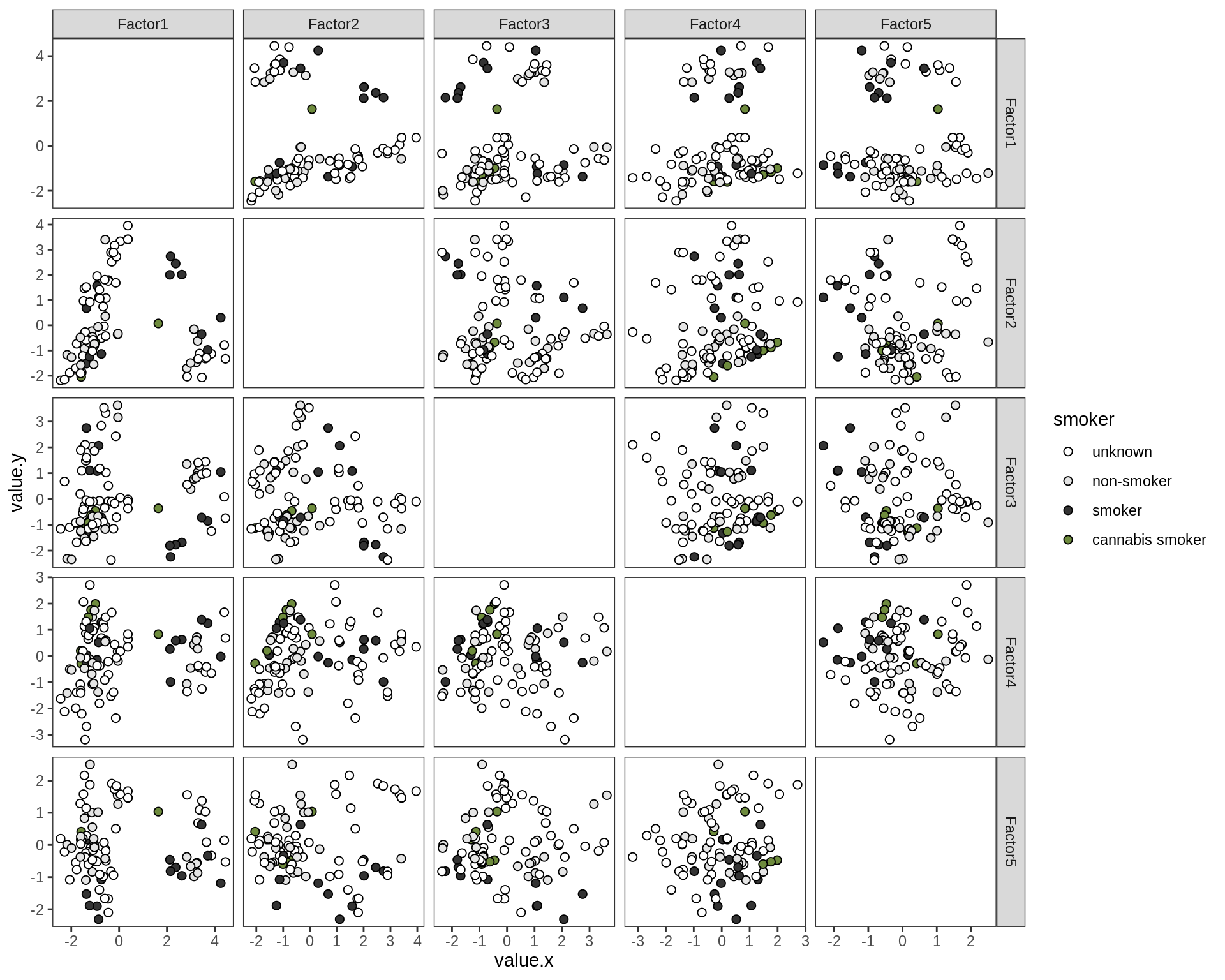
Factor distributions - pairwise
for (annot in c('subject_id', 'lesion_type', 'diagnosis', 'sex', 'sample_source', 'smoker', 'oligo_grp')) {
cat('### by ', annot, '\n', sep = '')
print(plot_factors_pairwise(model, annots_dt, pb, by = annot))
cat('\n\n')
}by subject_id
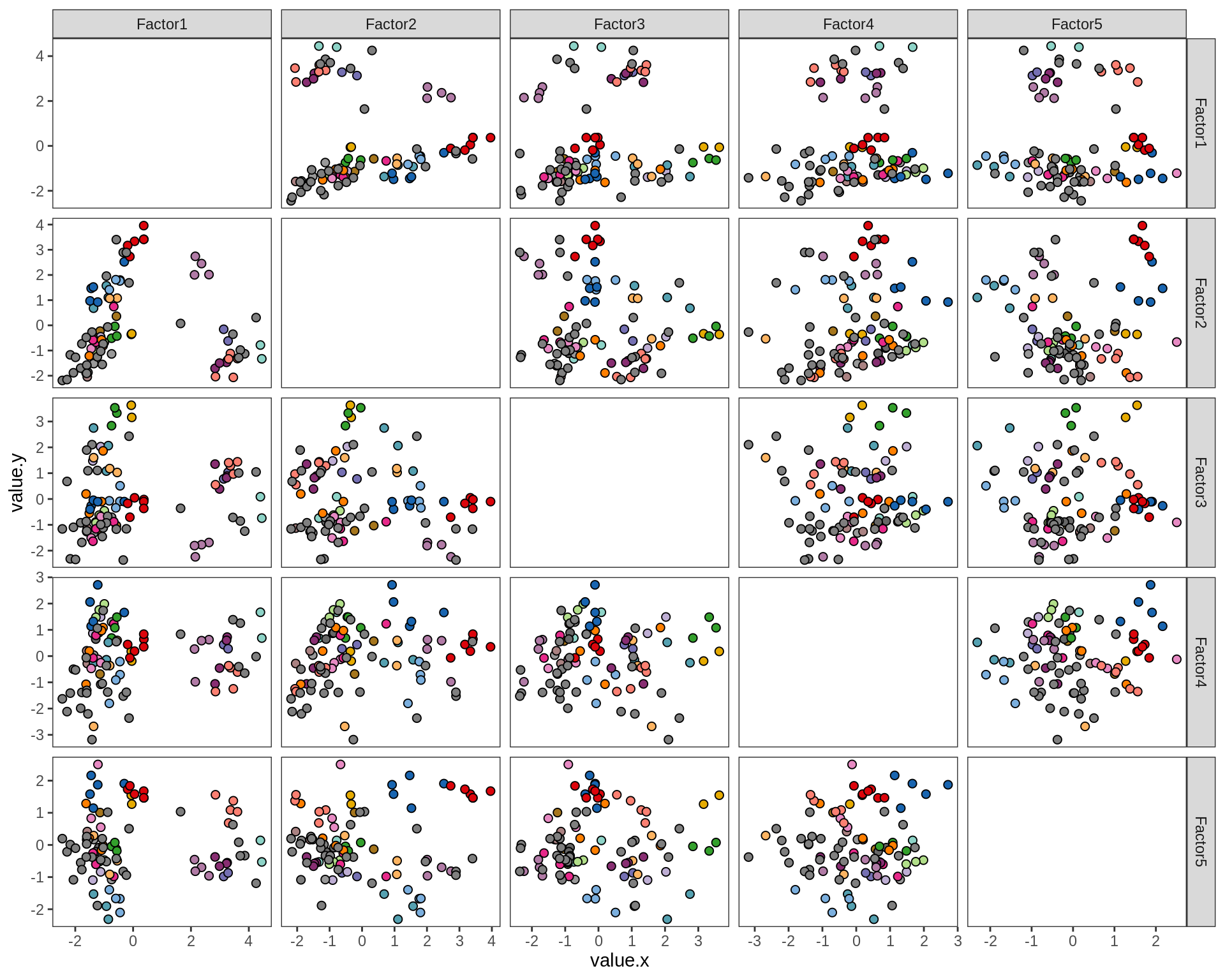
by lesion_type
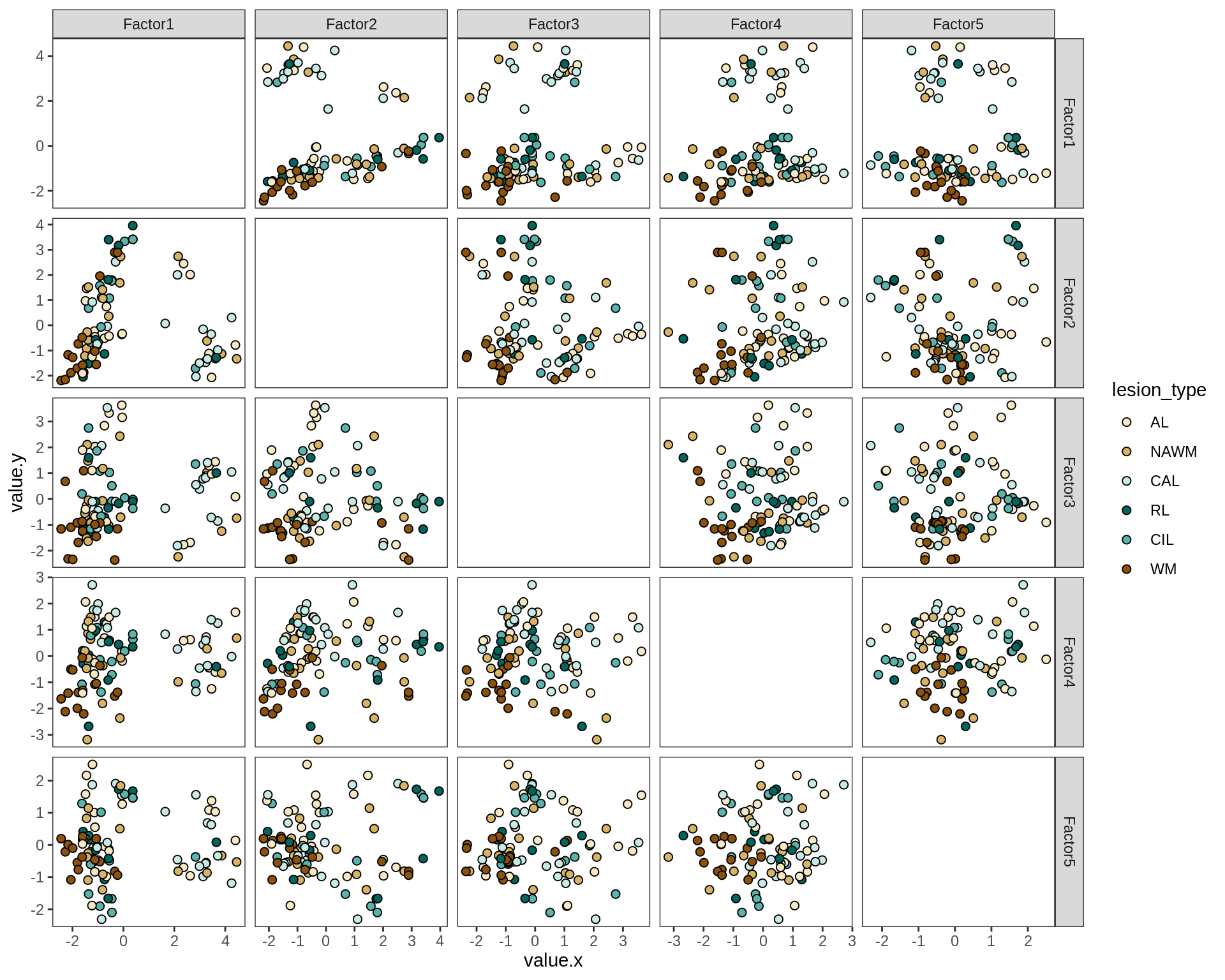
by diagnosis
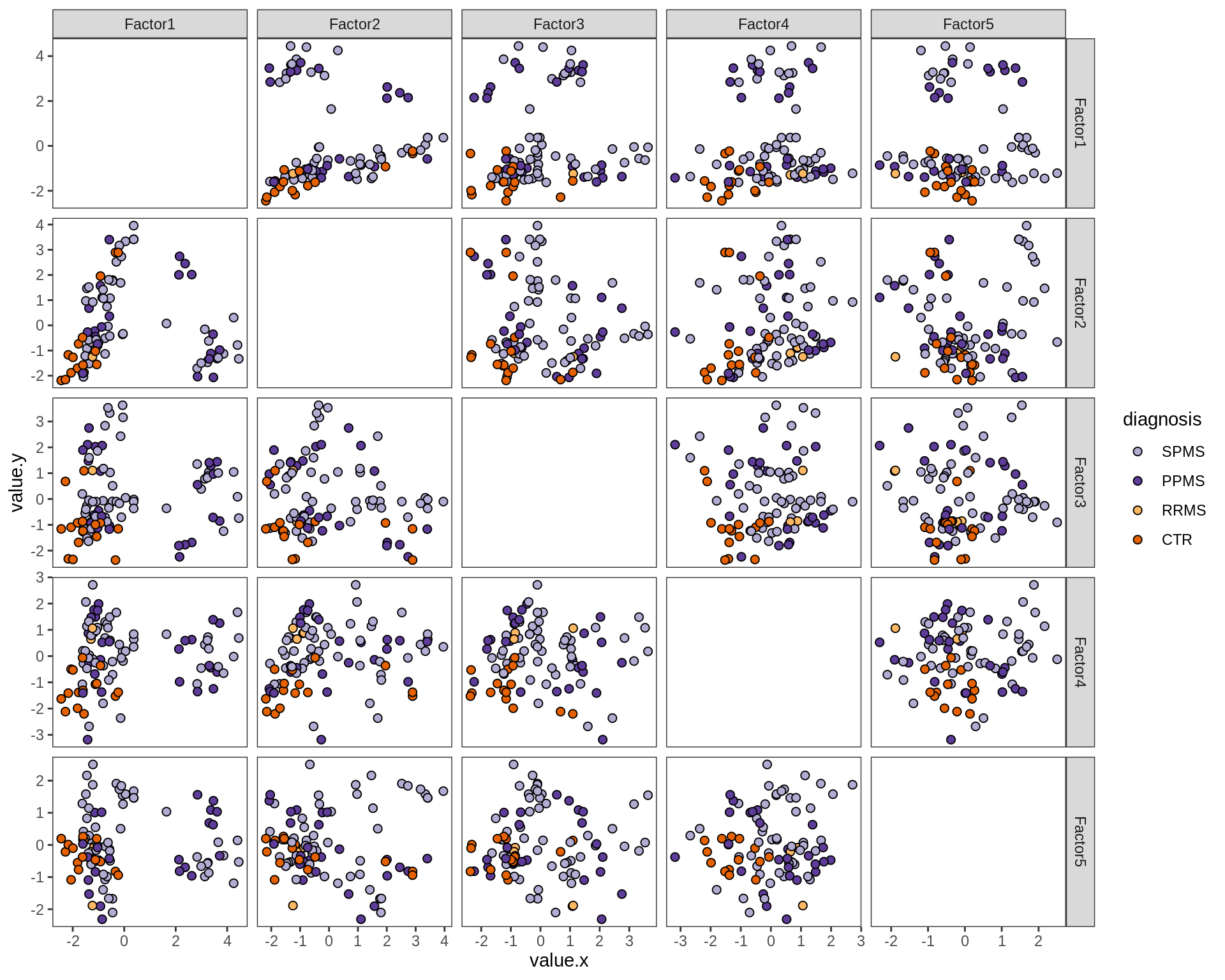
by sex
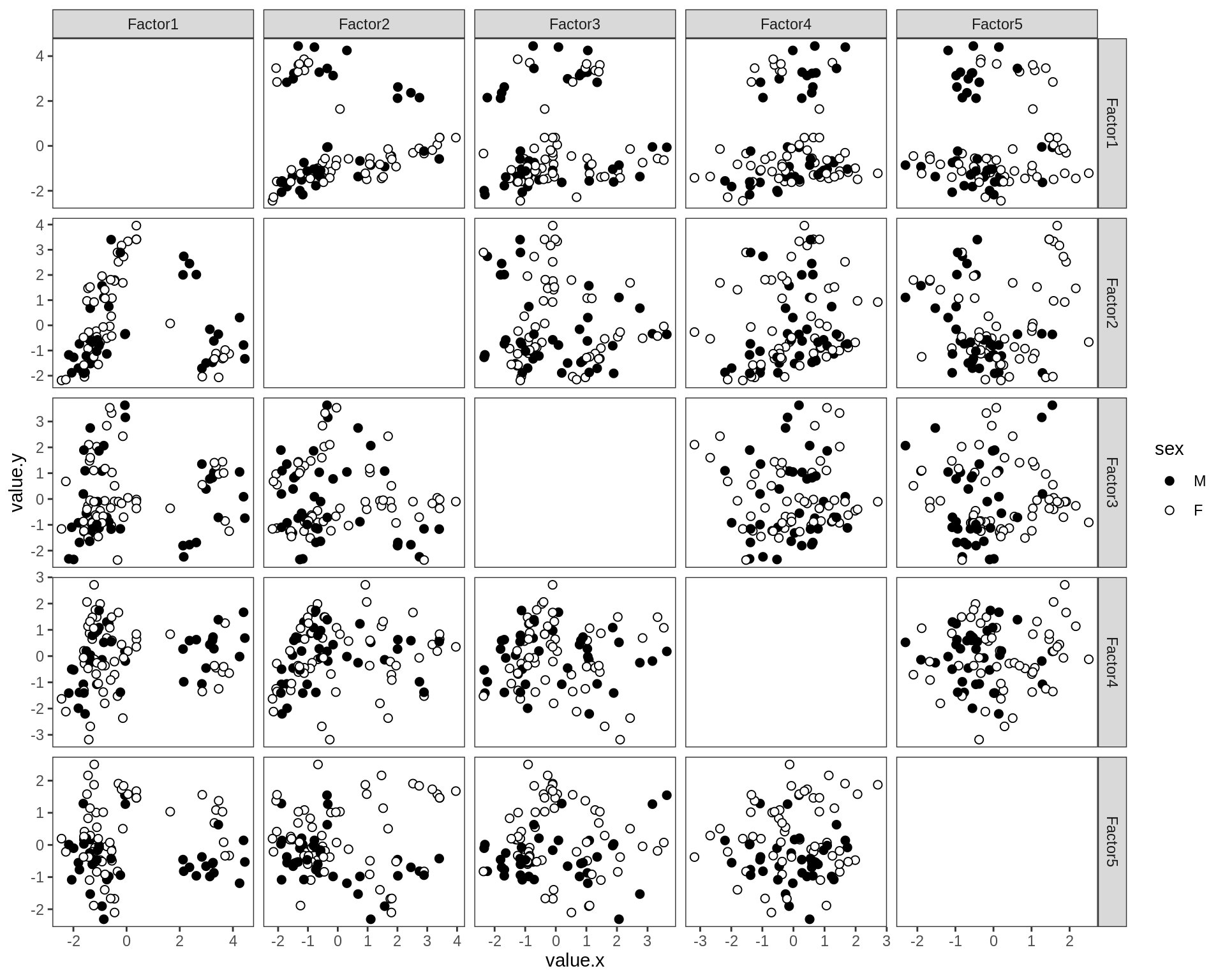
by sample_source
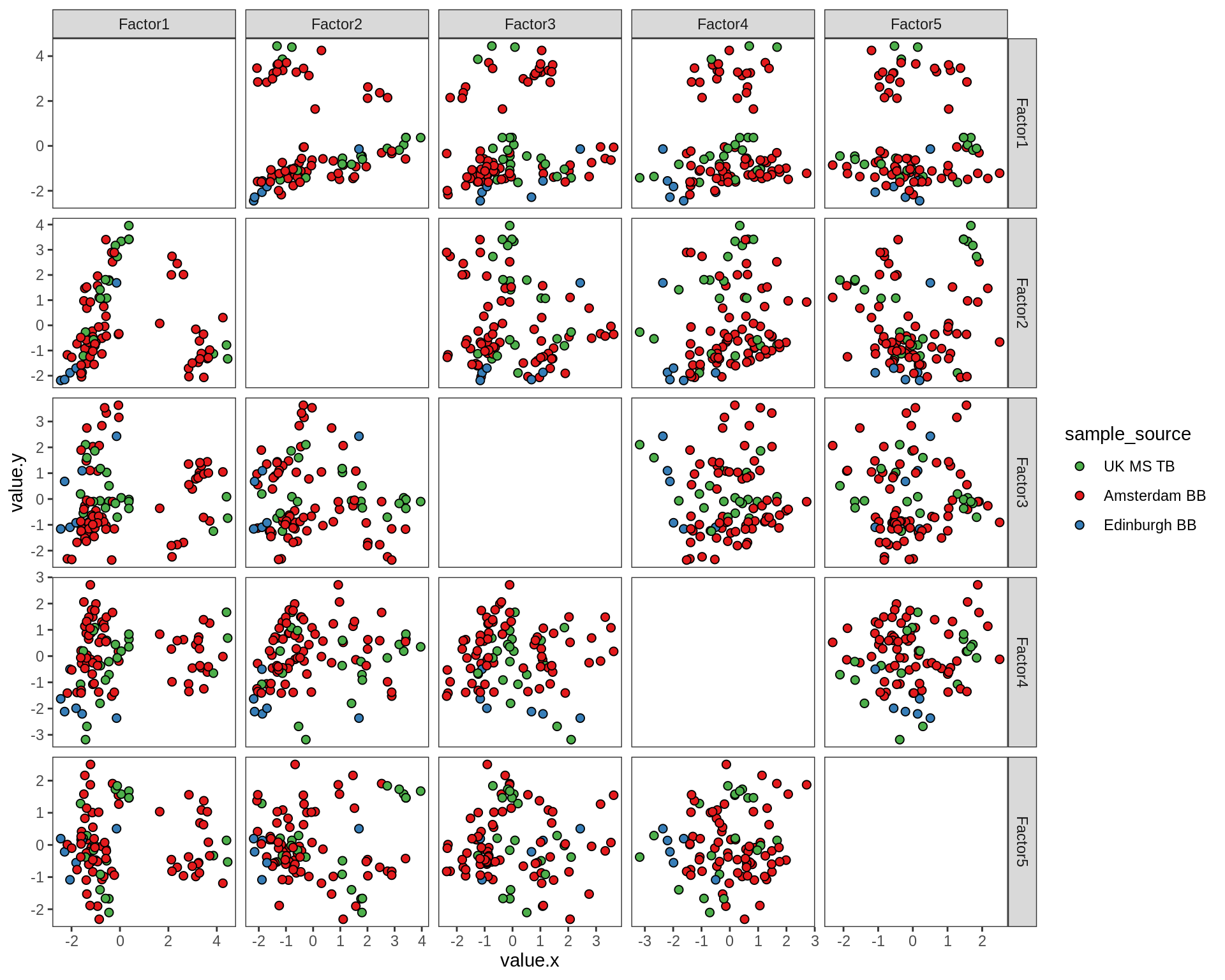
by smoker
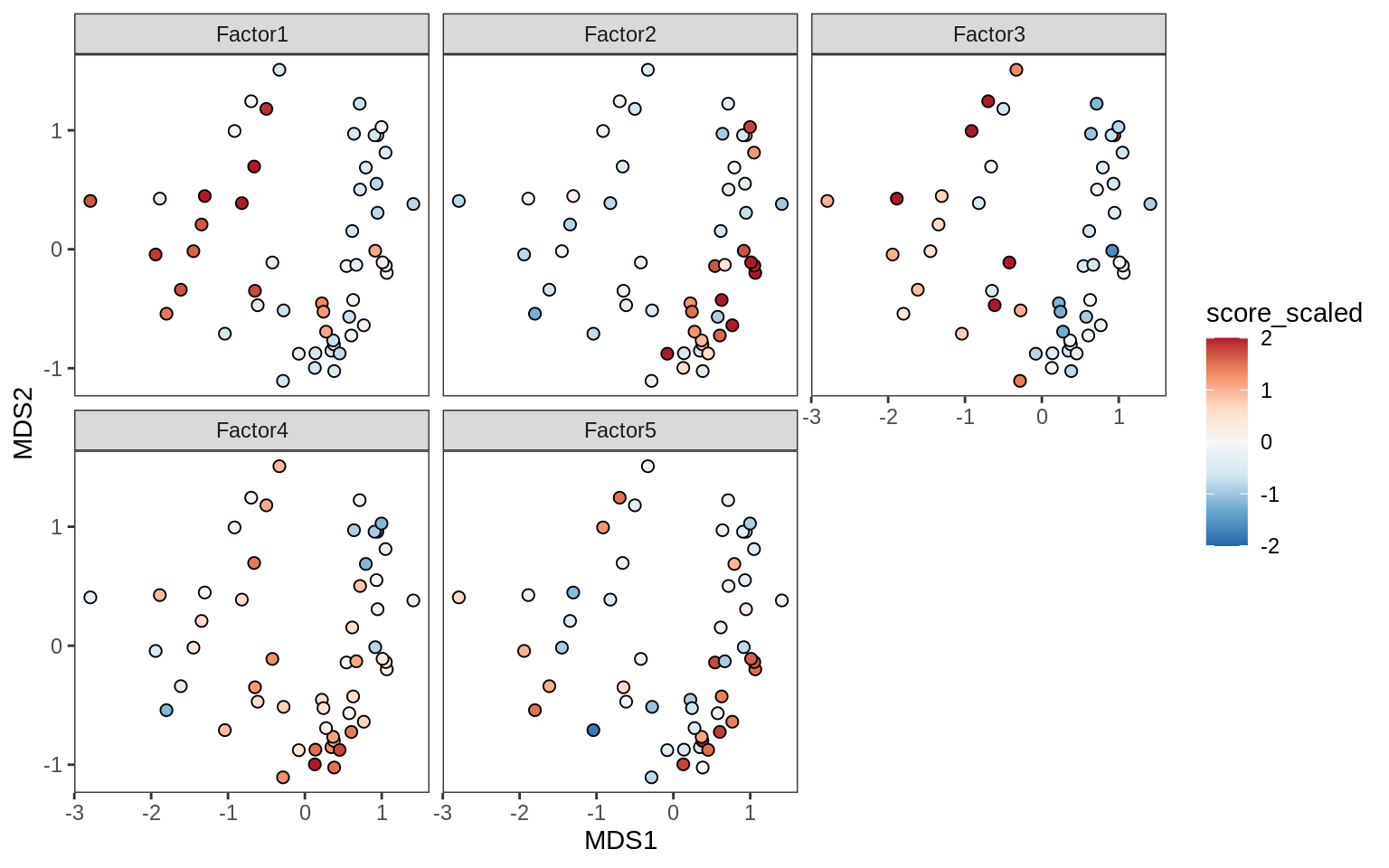
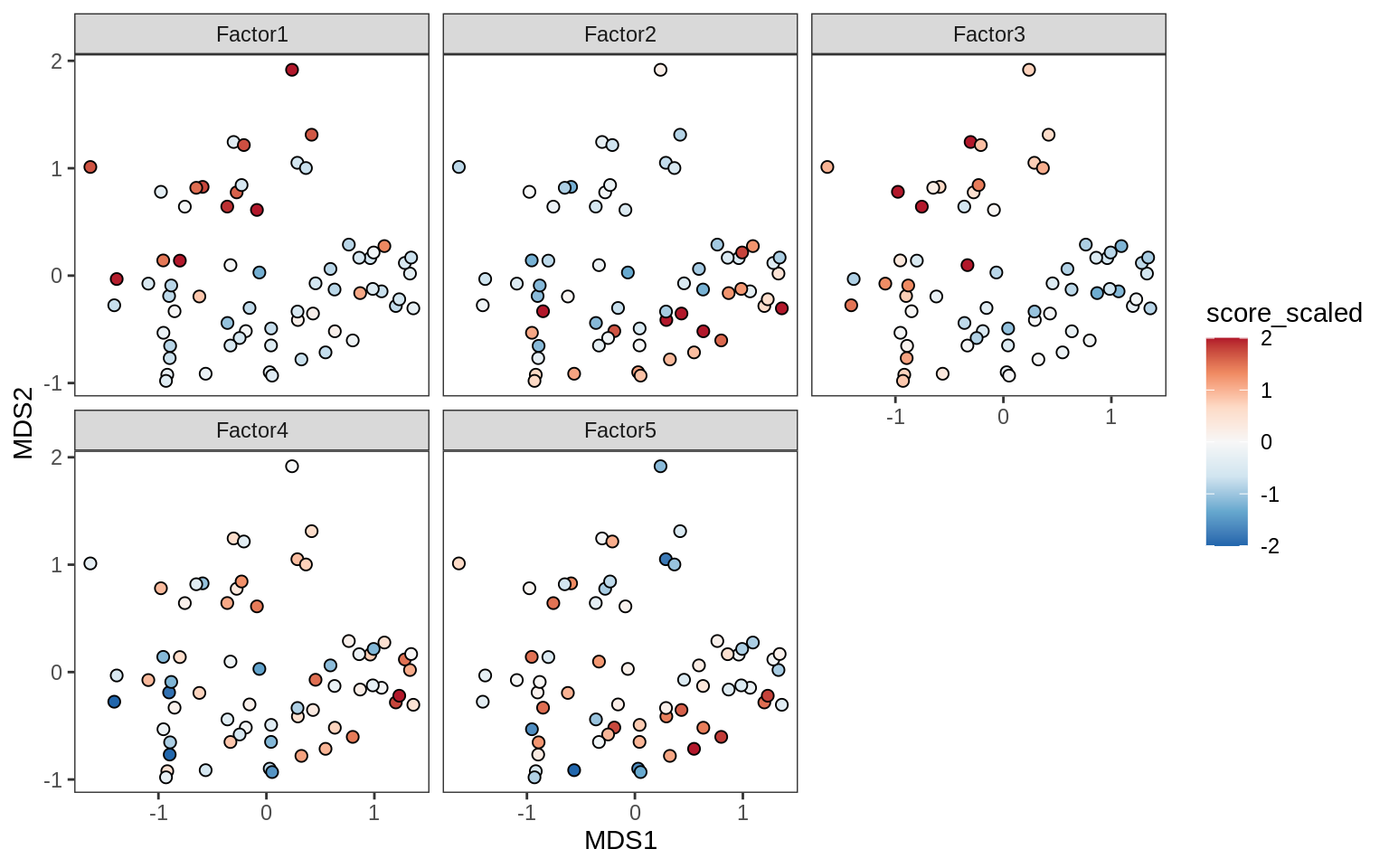
Factors over MDS layouts
for (cl in broad_ord) {
if (!(broad_short[[cl]] %in% views_names(model)))
next
cat('### ', cl, '\n', sep = '')
print(plot_factors_over_mds_samples(model, mds_sep_dt, cl = cl))
cat('\n\n')
}OPCs / COPs
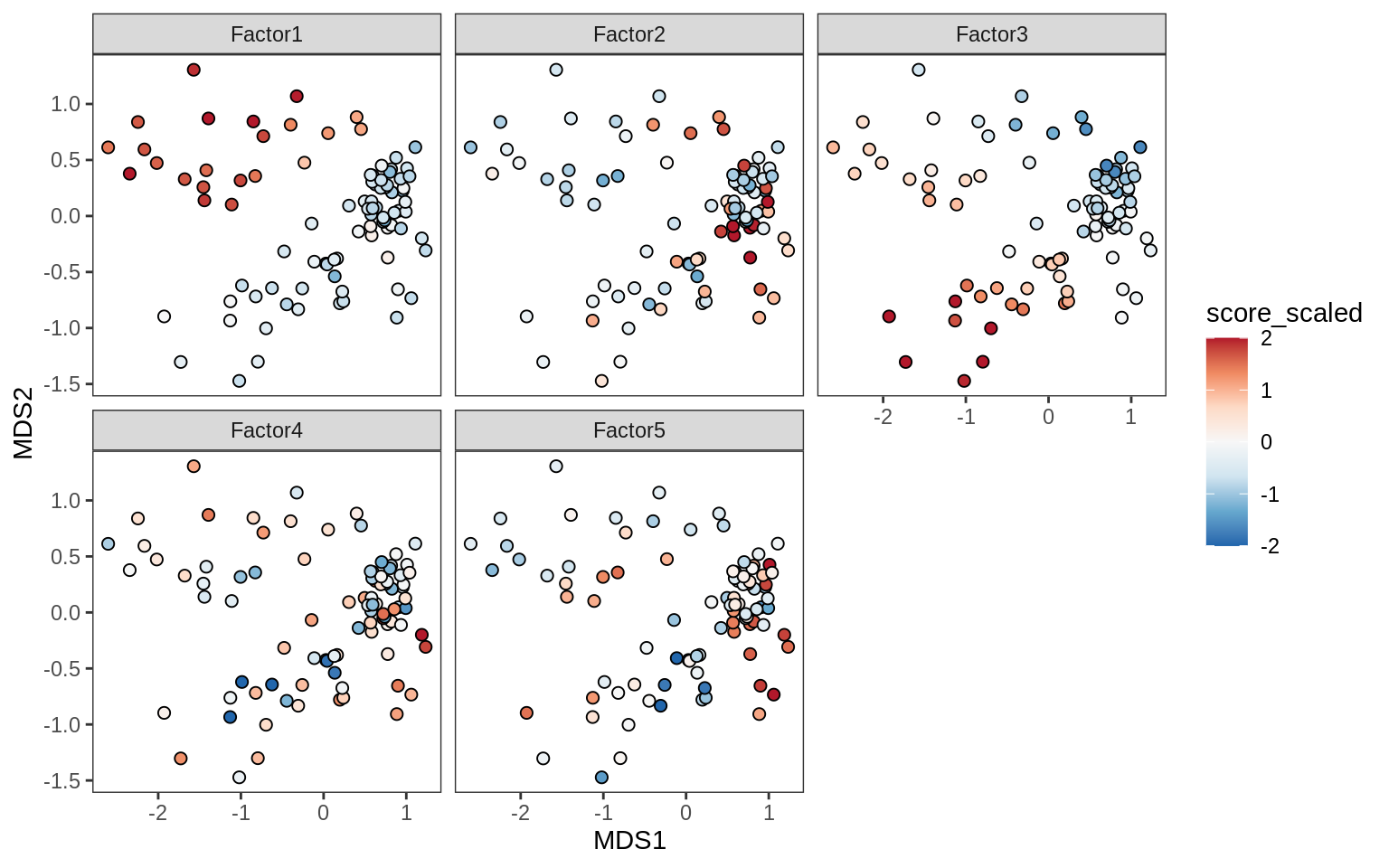
Oligodendrocytes
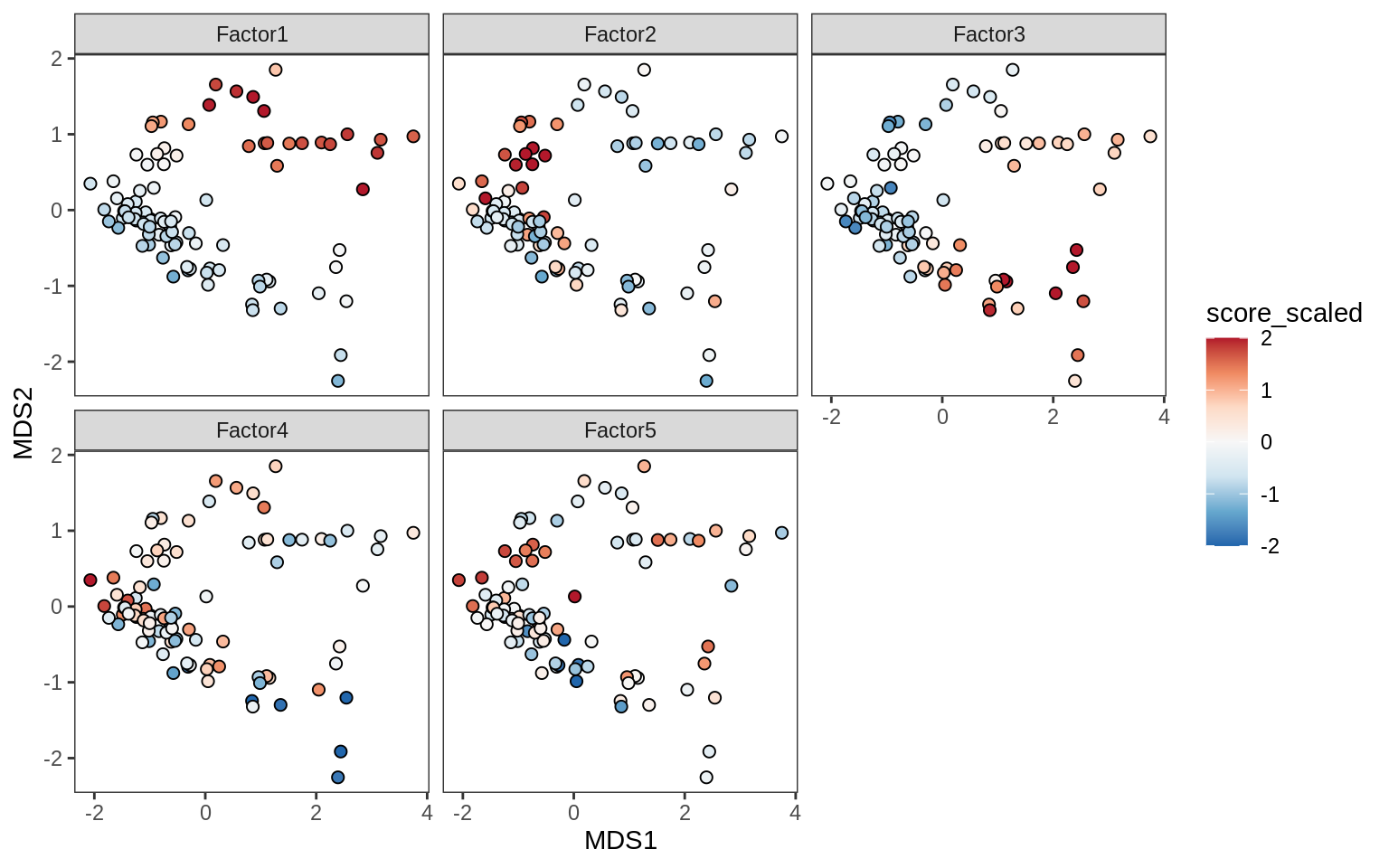
Astrocytes
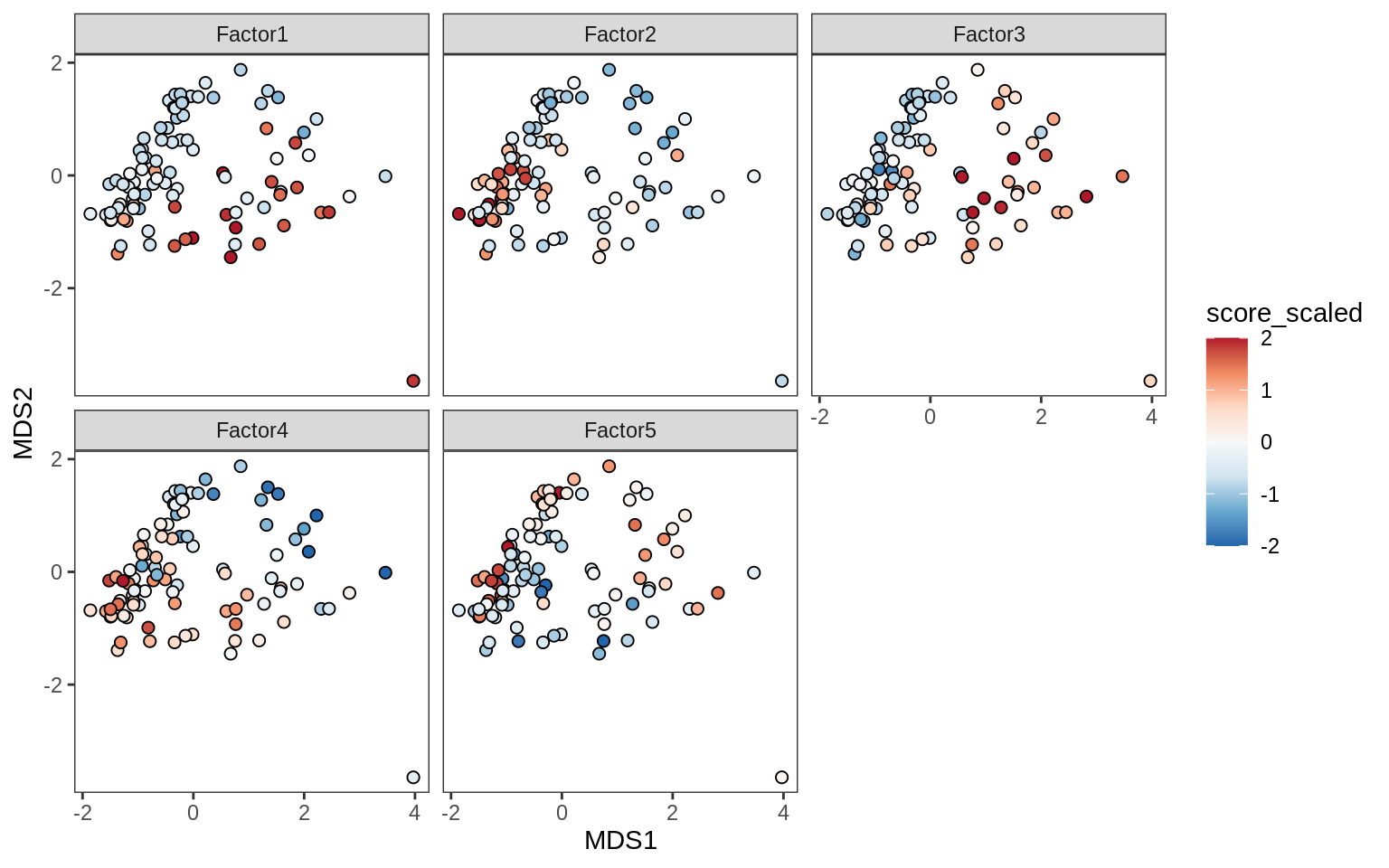
Microglia

Endothelial cells
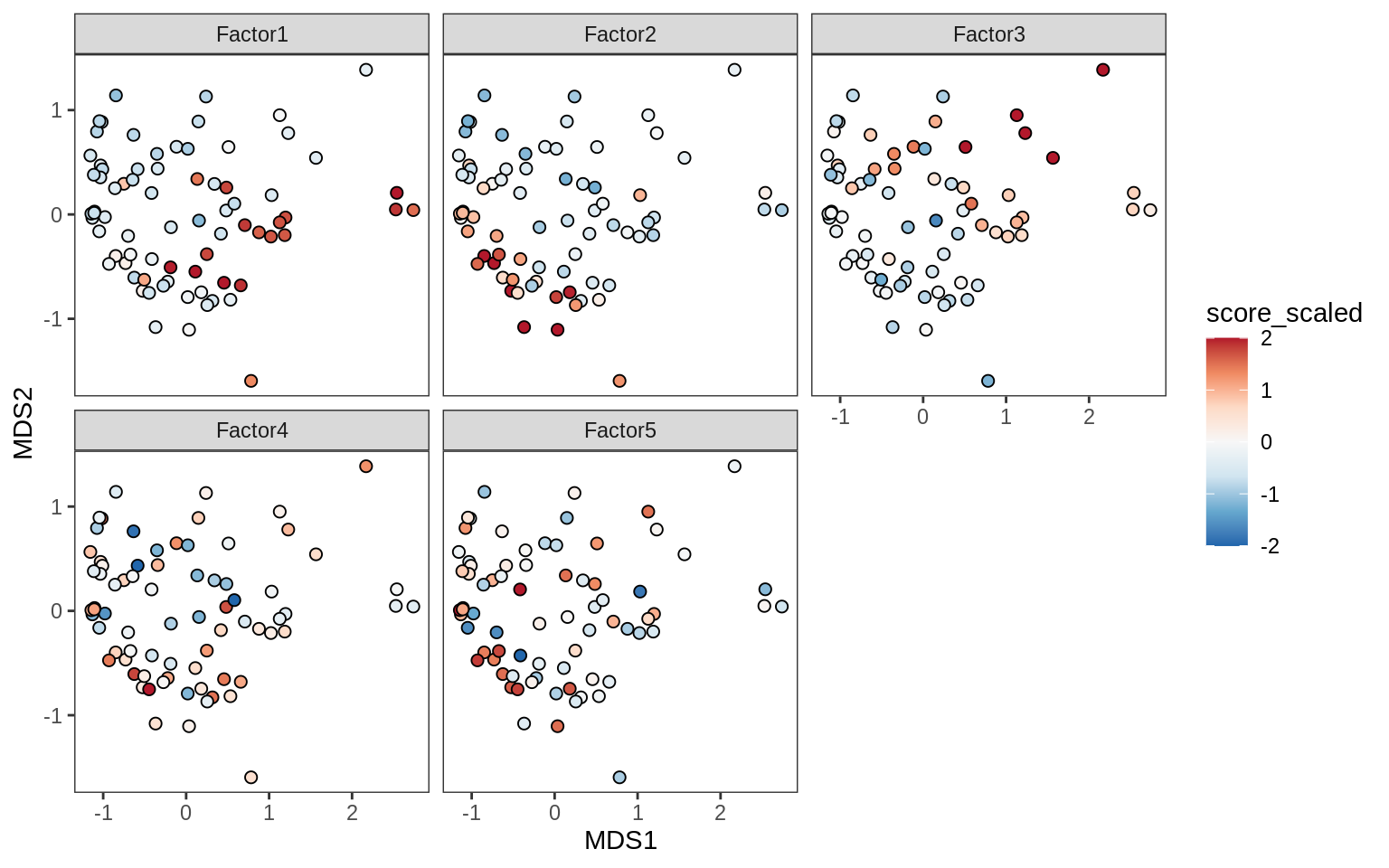
Pericytes
Oligo groupings over MDS layouts
olg_types = c("OPCs / COPs", "Oligodendrocytes")
(plot_oligo_grps_over_mds_samples(annots_dt, mds_sep_dt, olg_types))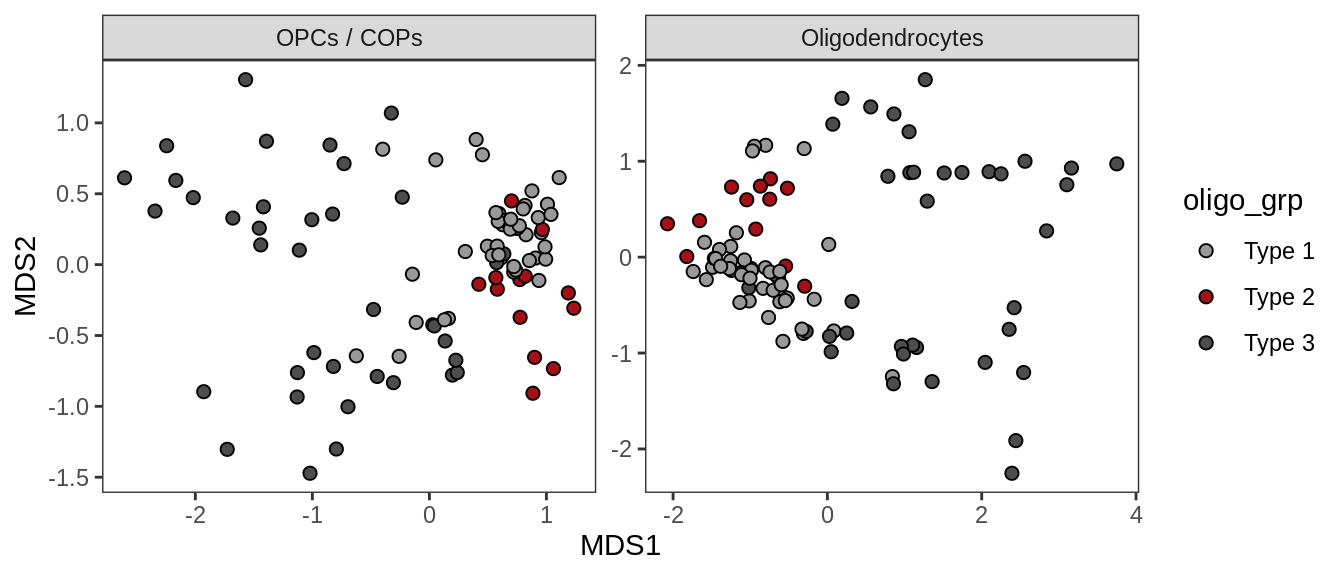
Factor distributions with patient annotations - few
for (v in c('score', 'score_scaled')) {
cat('### ', v, '\n', sep = '')
draw(plot_factors_heatmap(model, annots_dt, pb, what = 'few', plot_var = v))
cat('\n\n')
}score
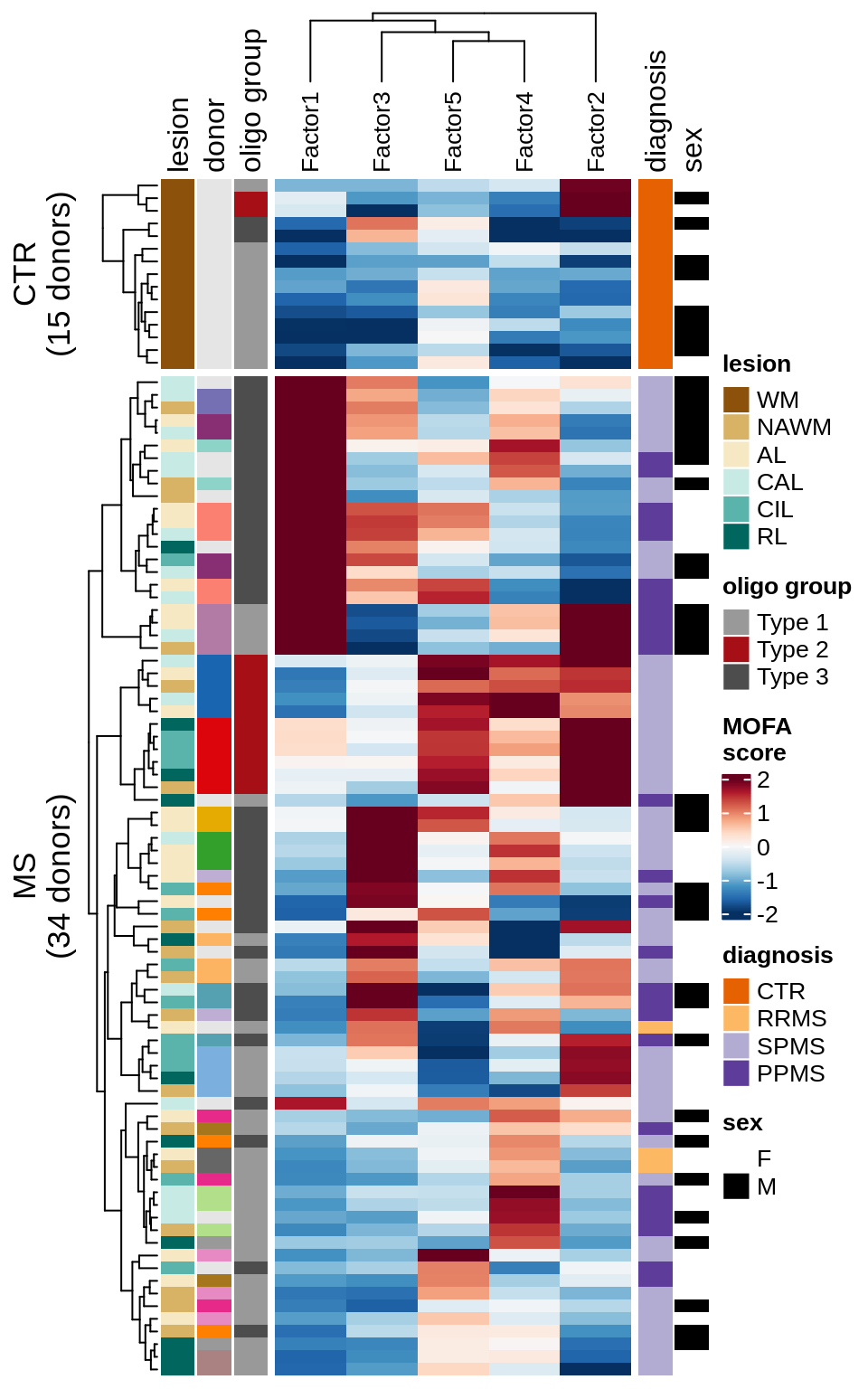
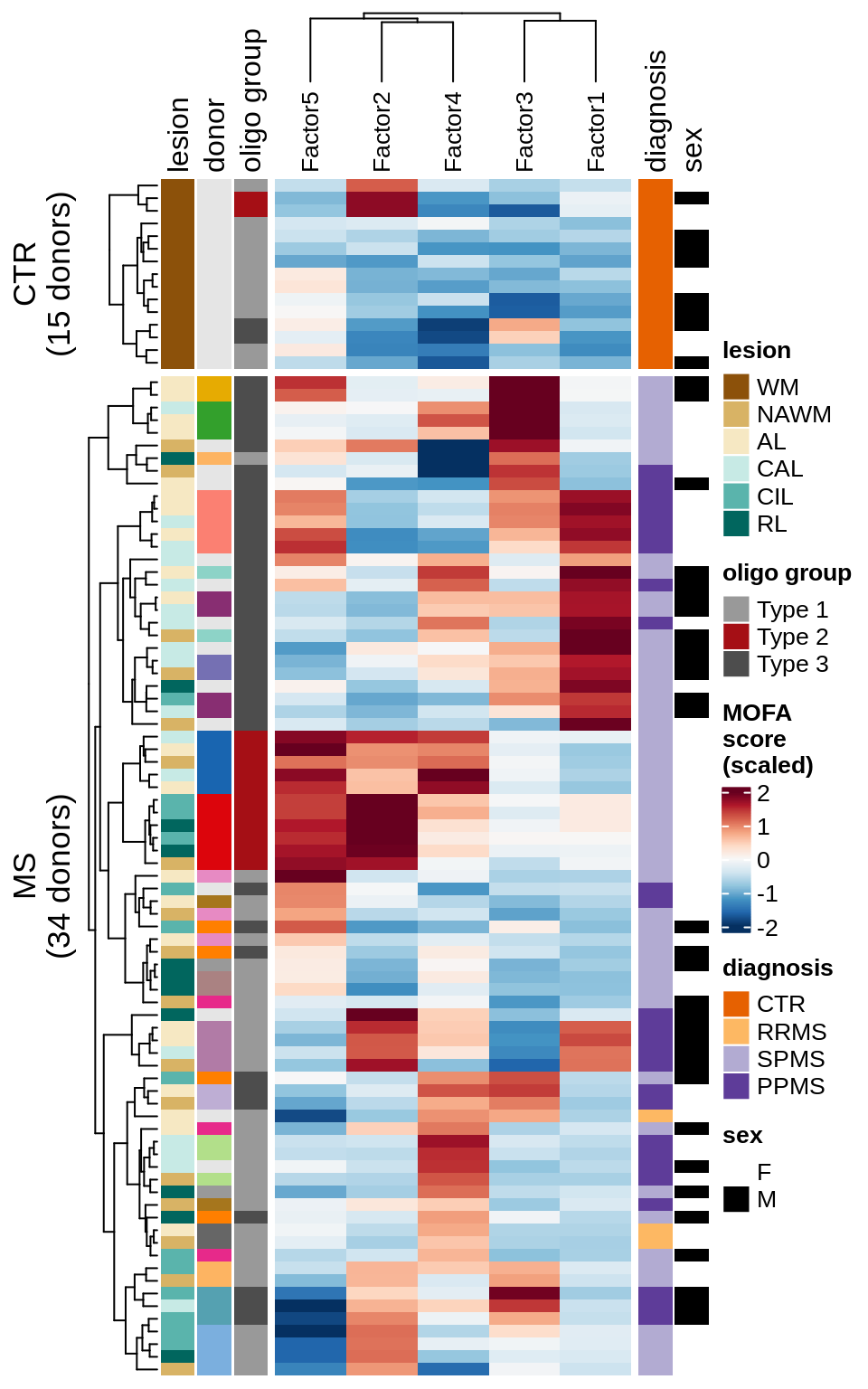
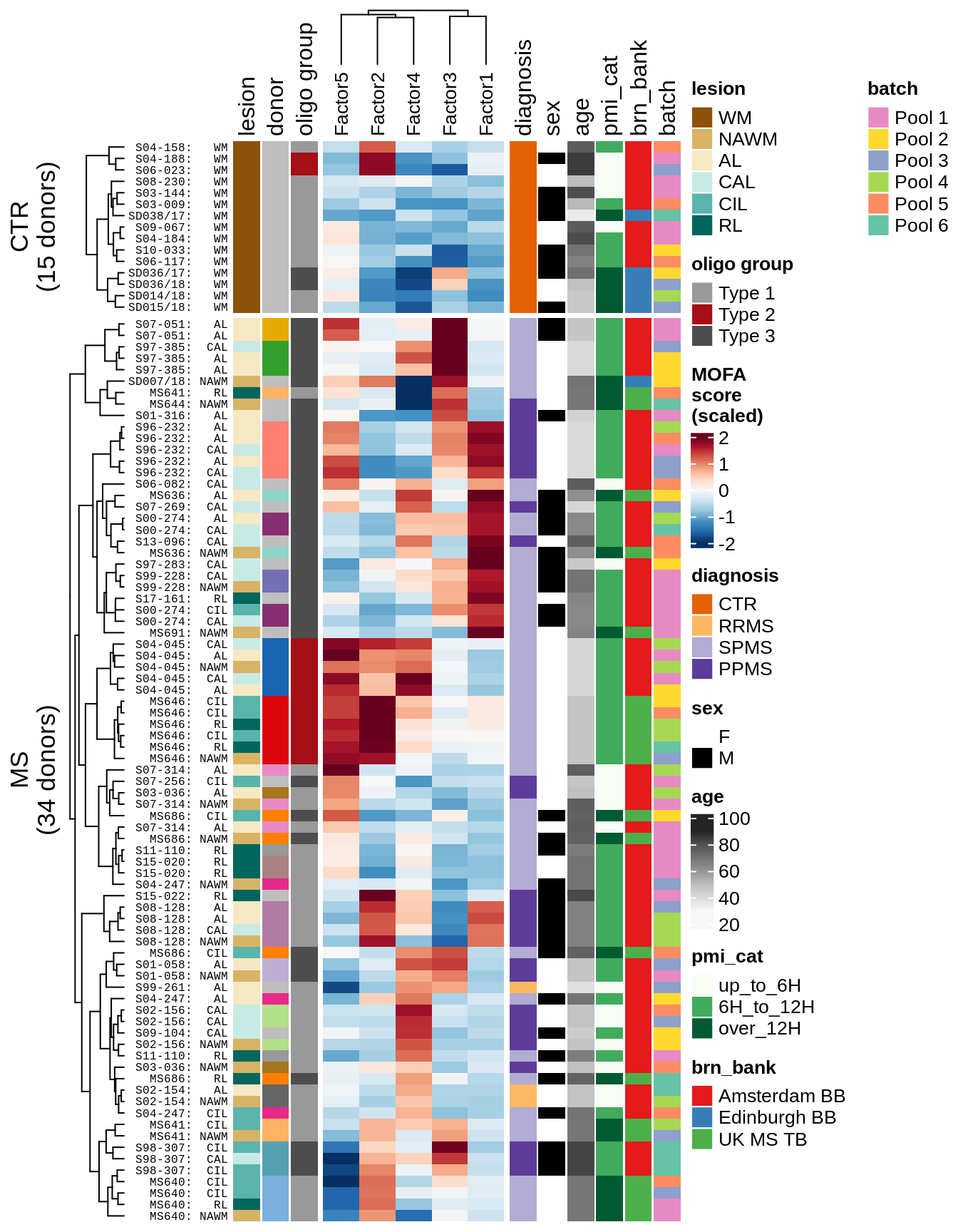
Factor distributions with patient annotations - all
for (v in c('score', 'score_scaled')) {
cat('### ', v, '\n', sep = '')
draw(plot_factors_heatmap(model, annots_dt, pb, what = 'all', plot_var = v),
merge_legend = TRUE)
cat('\n\n')
}score

Factor distributions with patient annotations - with QC
draw(plot_factors_heatmap_w_qc(model, annots_dt, pb, qc_stats))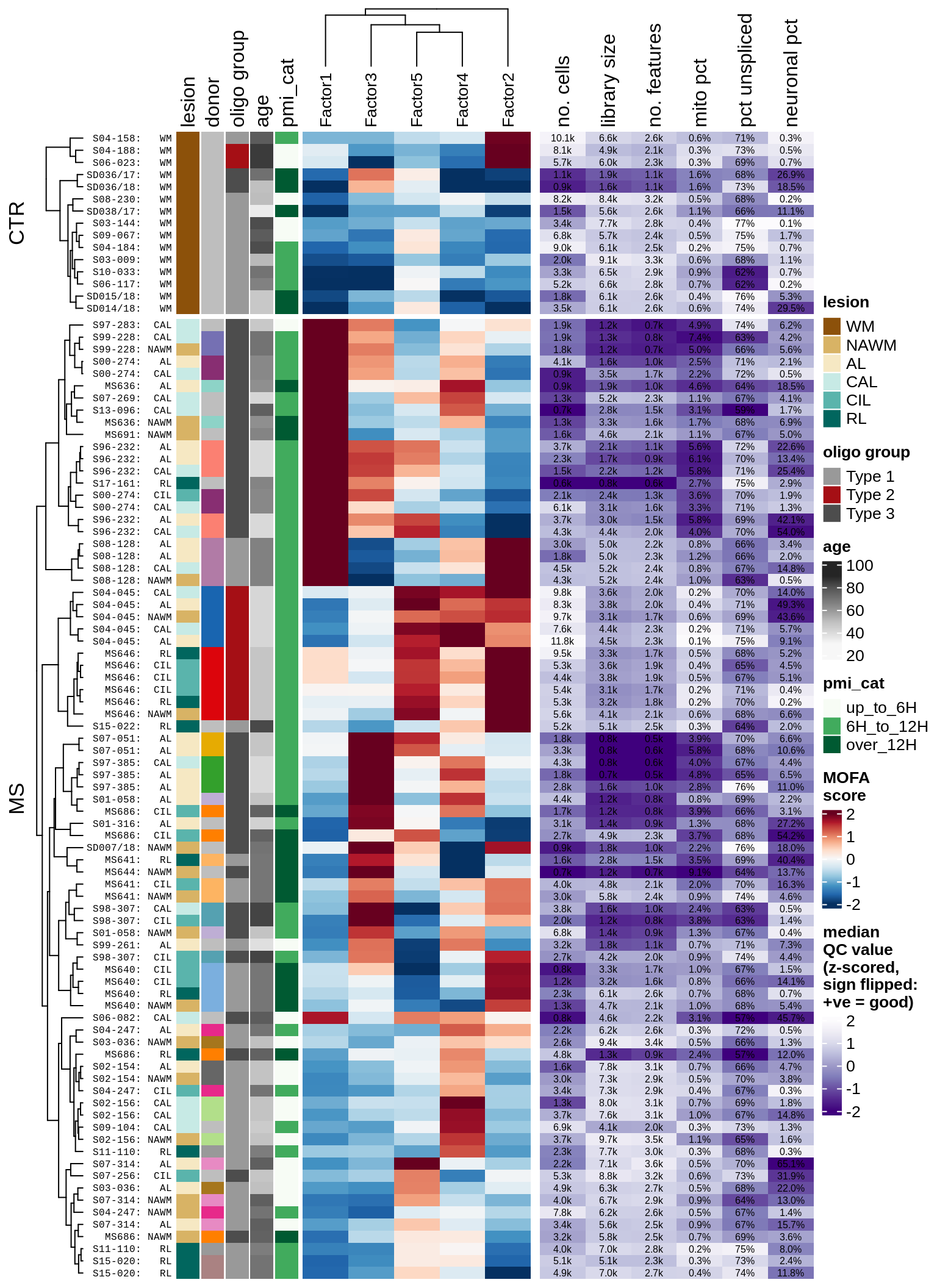
Factors vs QC metrics
(plot_factors_vs_qc(model, annots_dt, qc_stats))
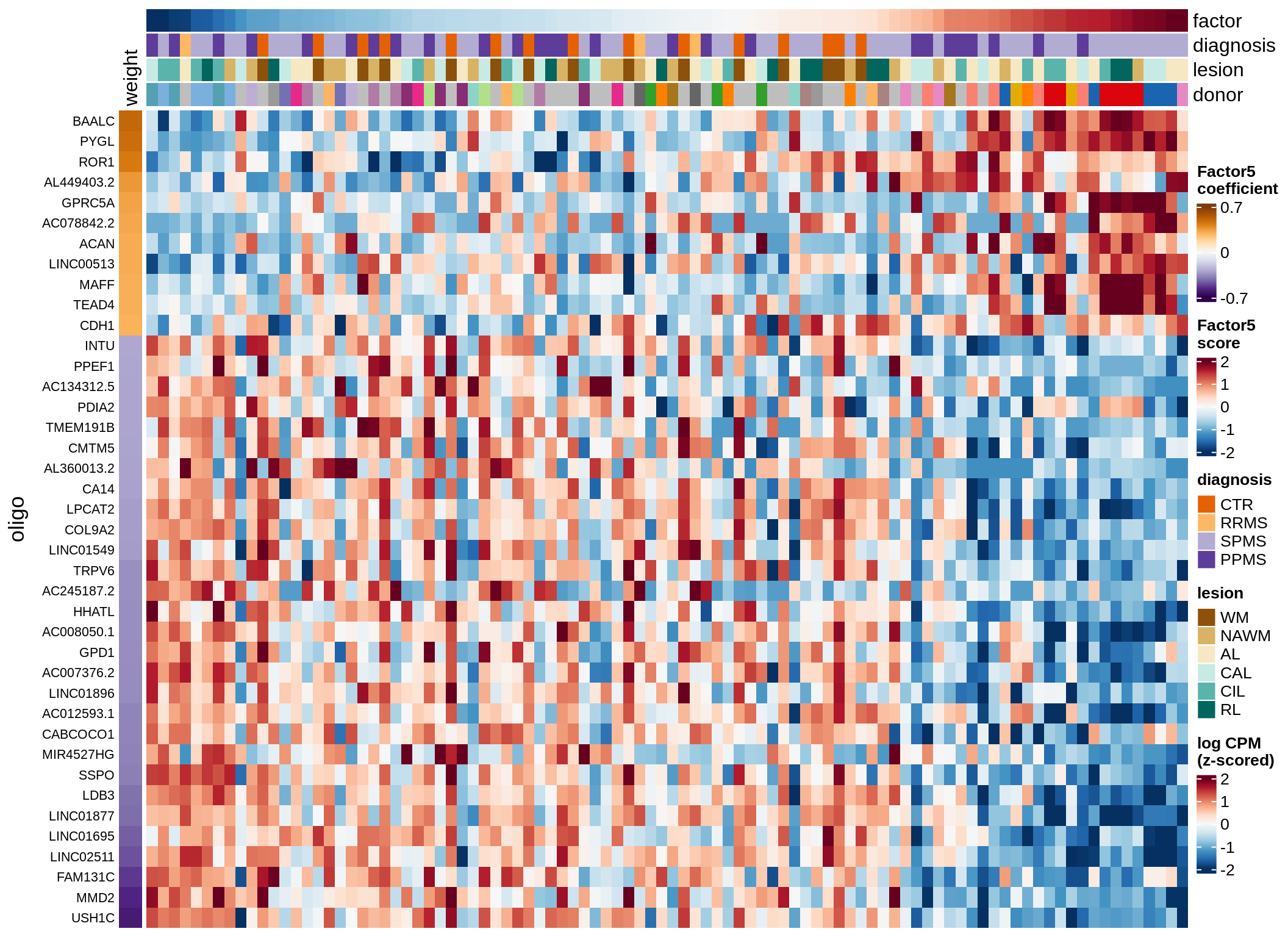
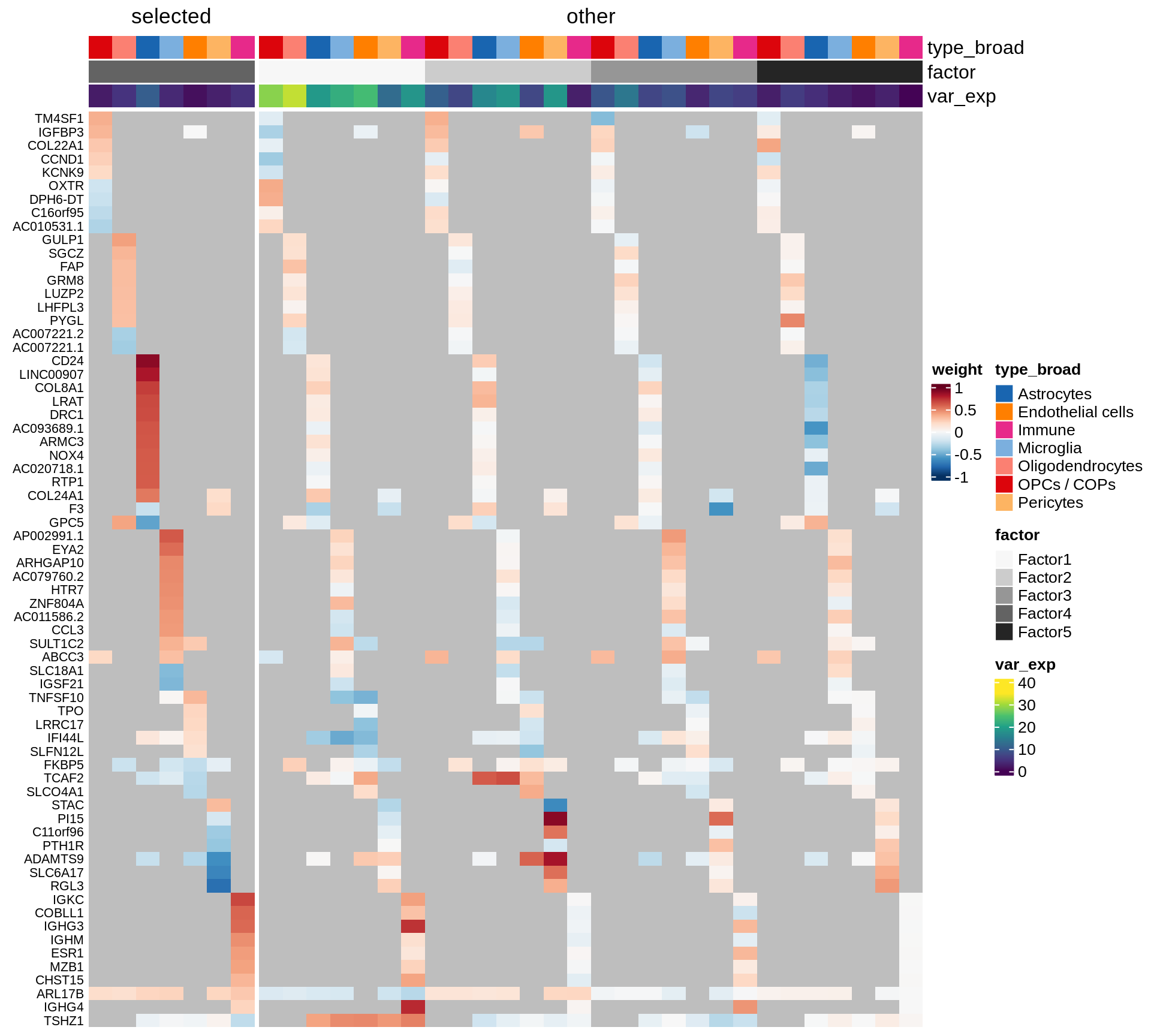
Coefficients of top genes (by factor)
for (f in factors_names(model)) {
cat('### ', f, '\n', sep = '')
draw(plot_top_weights_heatmap_by_factor(model, var_exp_dt, sel_f = f))
cat('\n\n')
}Factor1

Factor2

Factor3
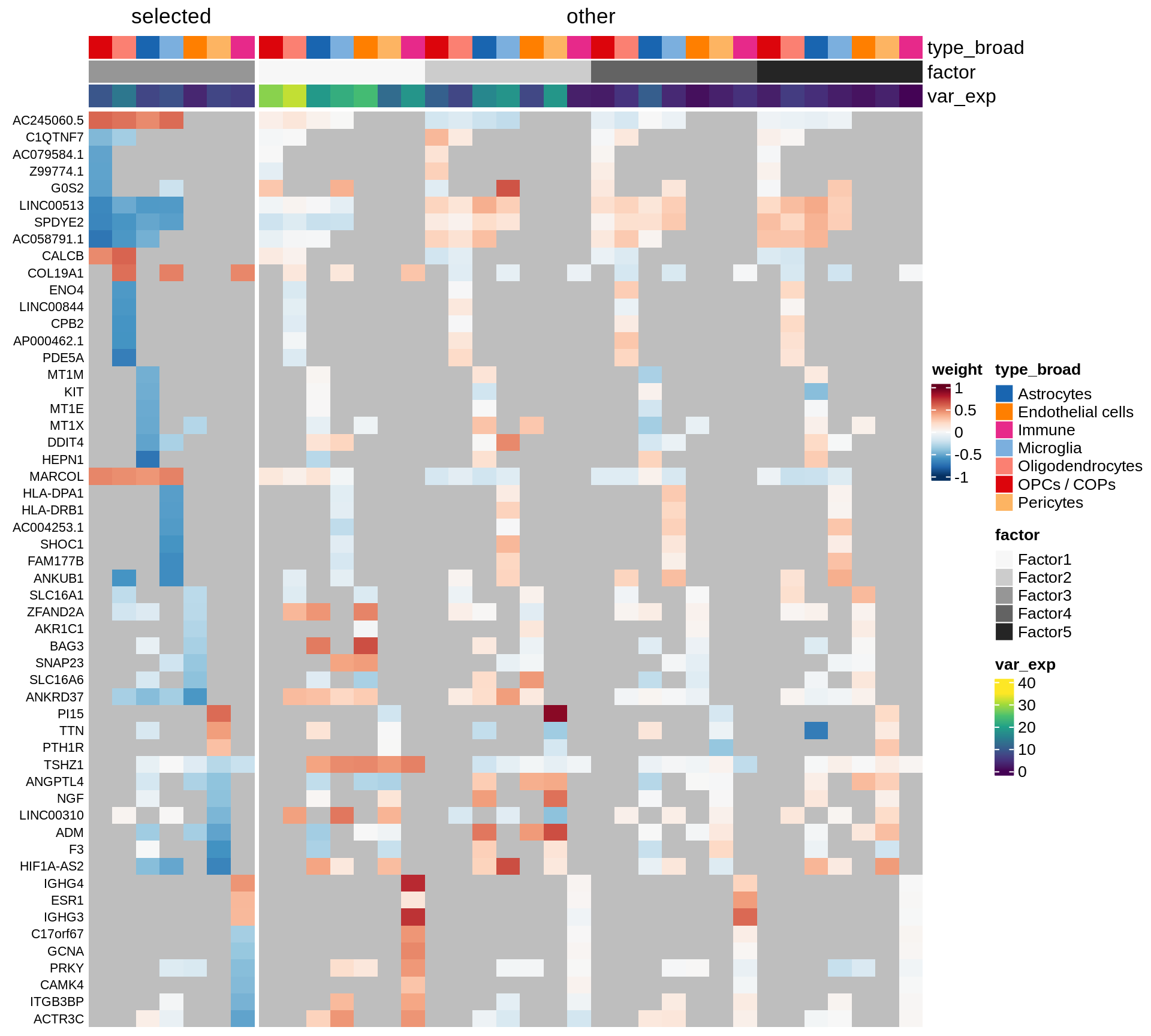
Factor4
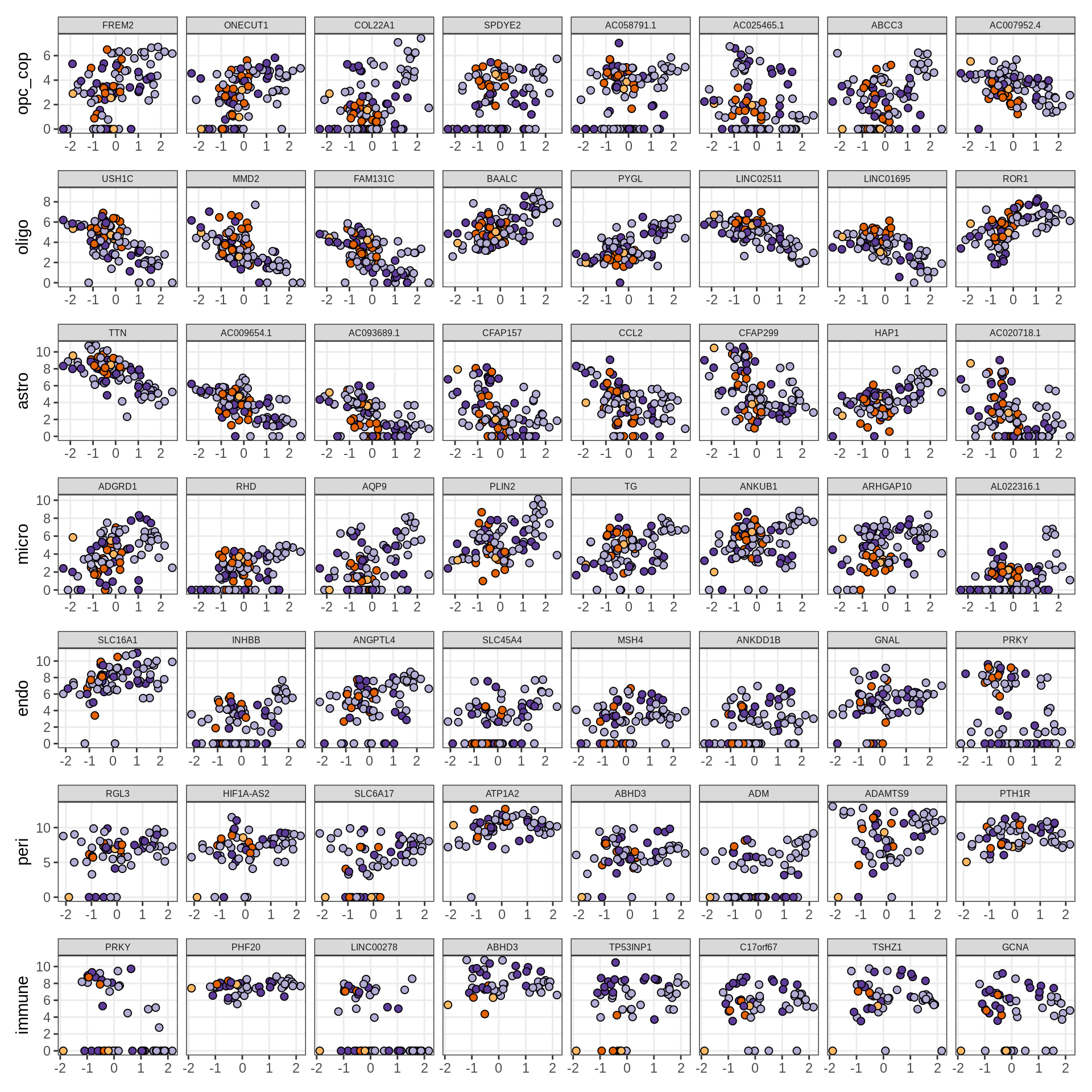
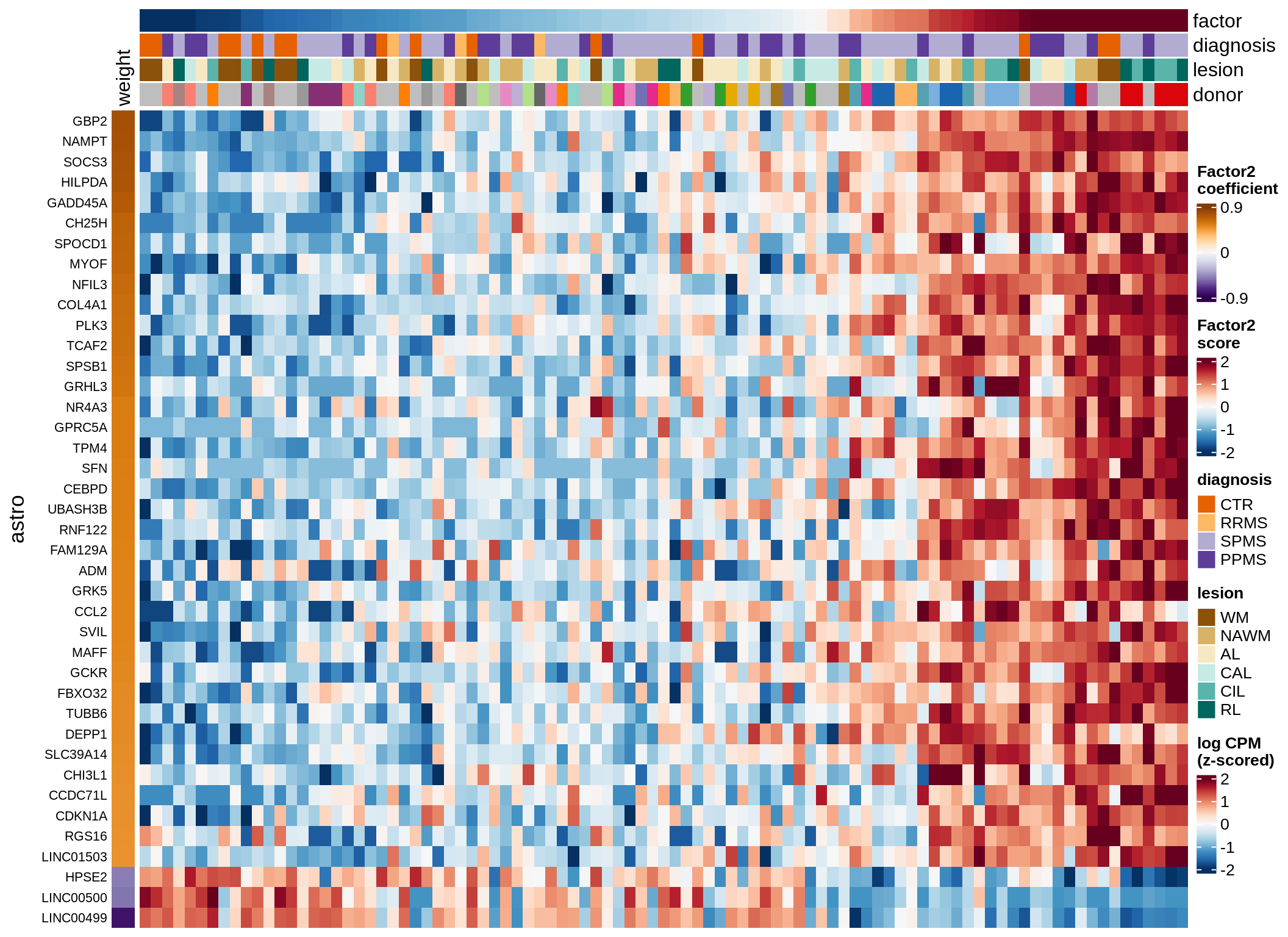
Expression of top genes per celltype
# iterate plots
for (i in seq.int(nrow(to_plot_dt))) {
# descriptor of what plotted
sel_v = as.character(to_plot_dt[i]$view)
sel_f = to_plot_dt[i]$factor
this_r2 = to_plot_dt[i]$var_exp
# plot!
cat('### ', sel_v, '-F', as.integer(sel_f),
' (', round(this_r2, 0), '%)', '\n', sep = '')
draw(plot_top_genes_expression(model, pb, annots_dt,
filter_dt, tfs_dt, to_plot_dt[i],
sel_f = sel_f, min_var = 5, min_w = 0.2, n_top = 40),
merge_legend = TRUE)
cat('\n\n')
}oligo-F1 (32%)

opc_cop-F1 (28%)
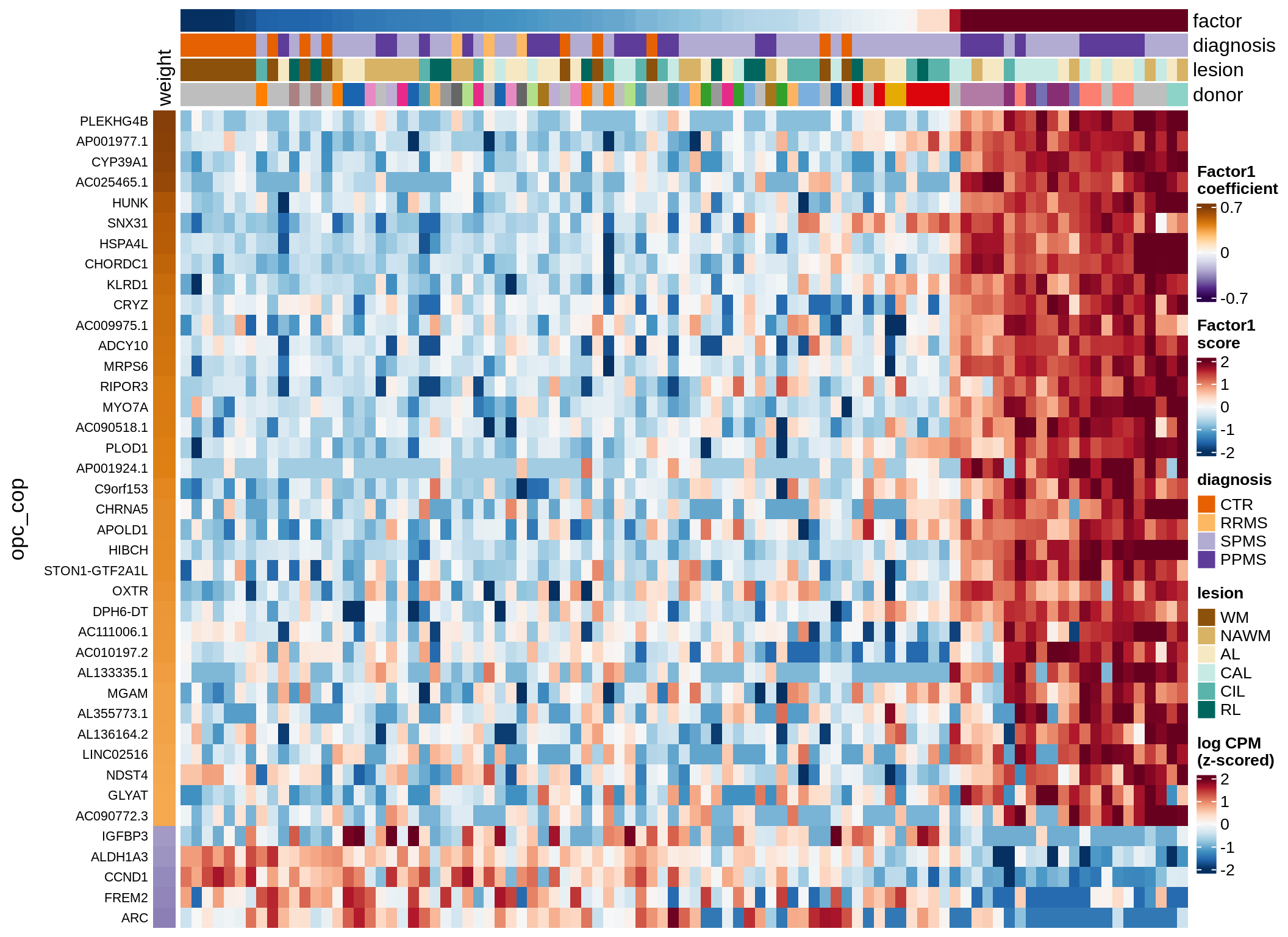
endo-F1 (24%)

micro-F1 (22%)
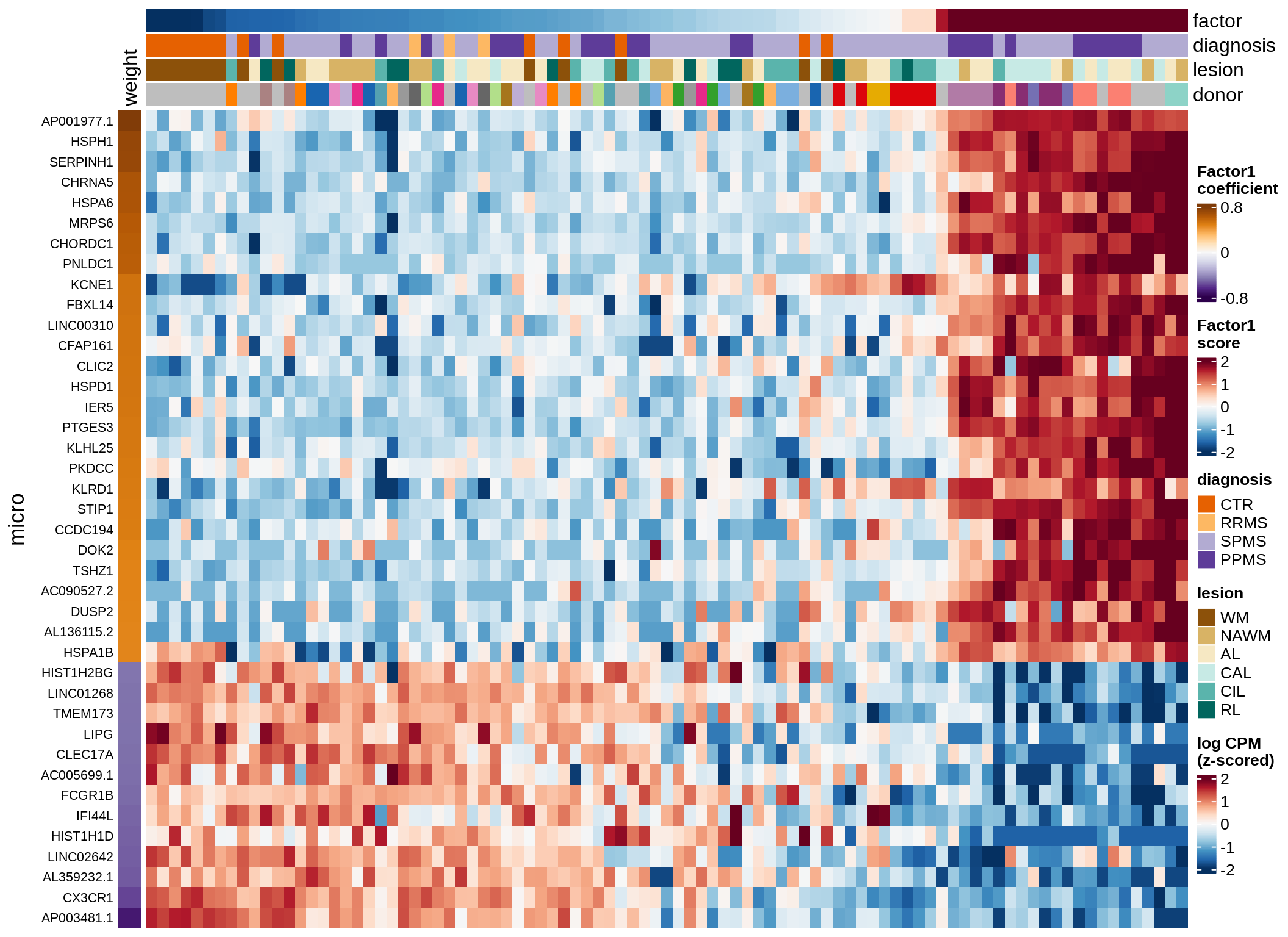
astro-F1 (19%)
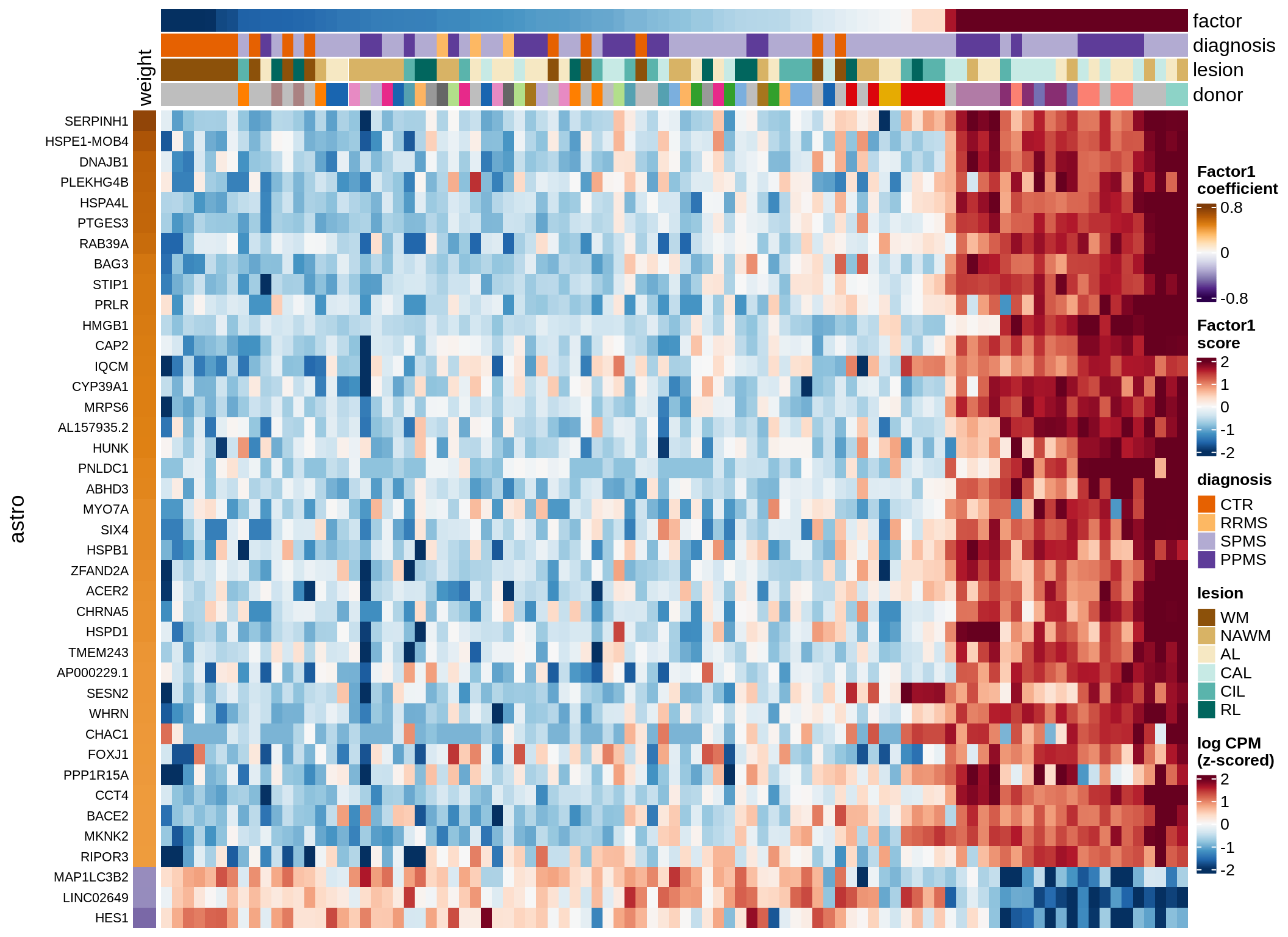
immune-F1 (18%)
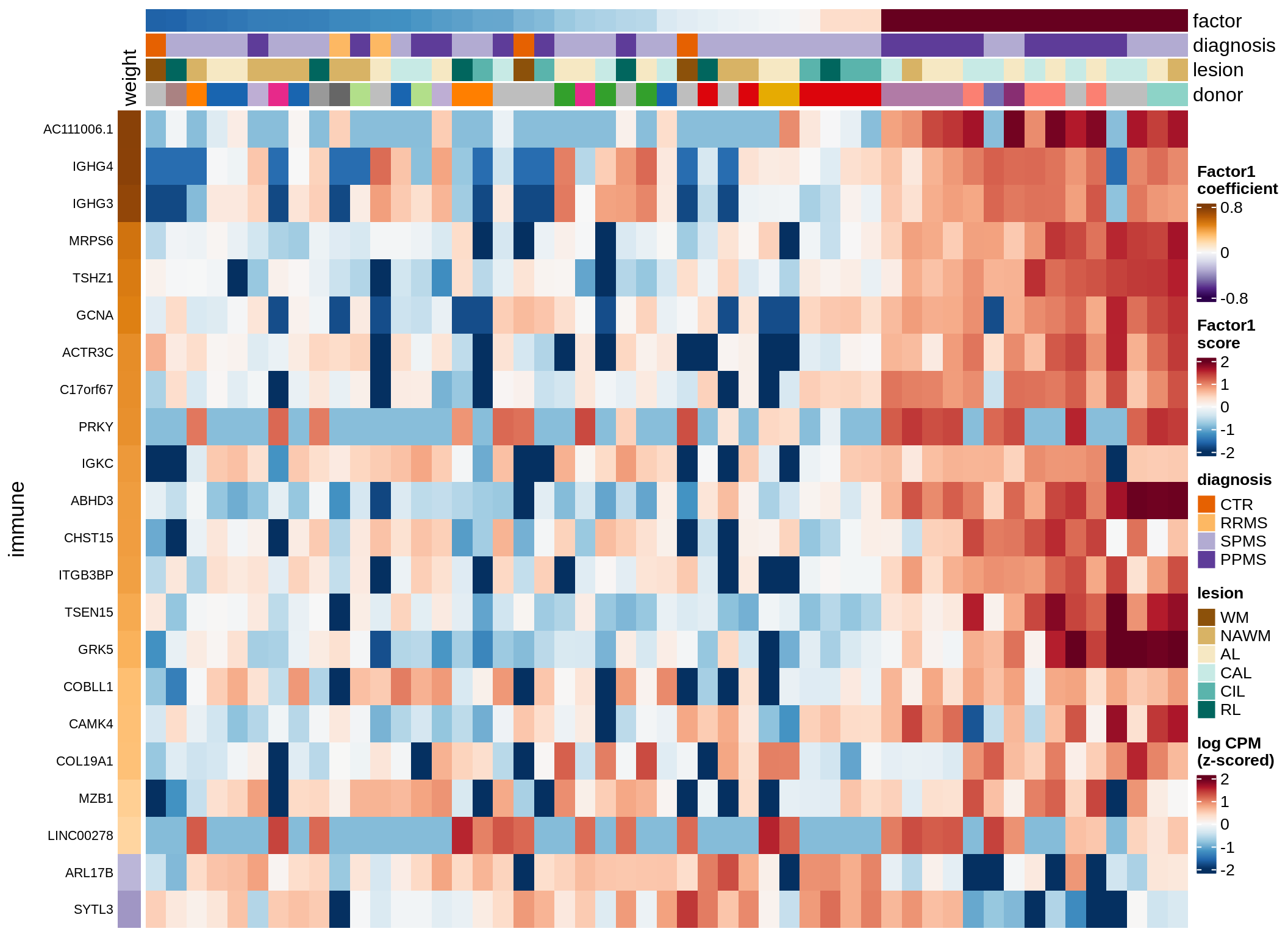
peri-F1 (12%)
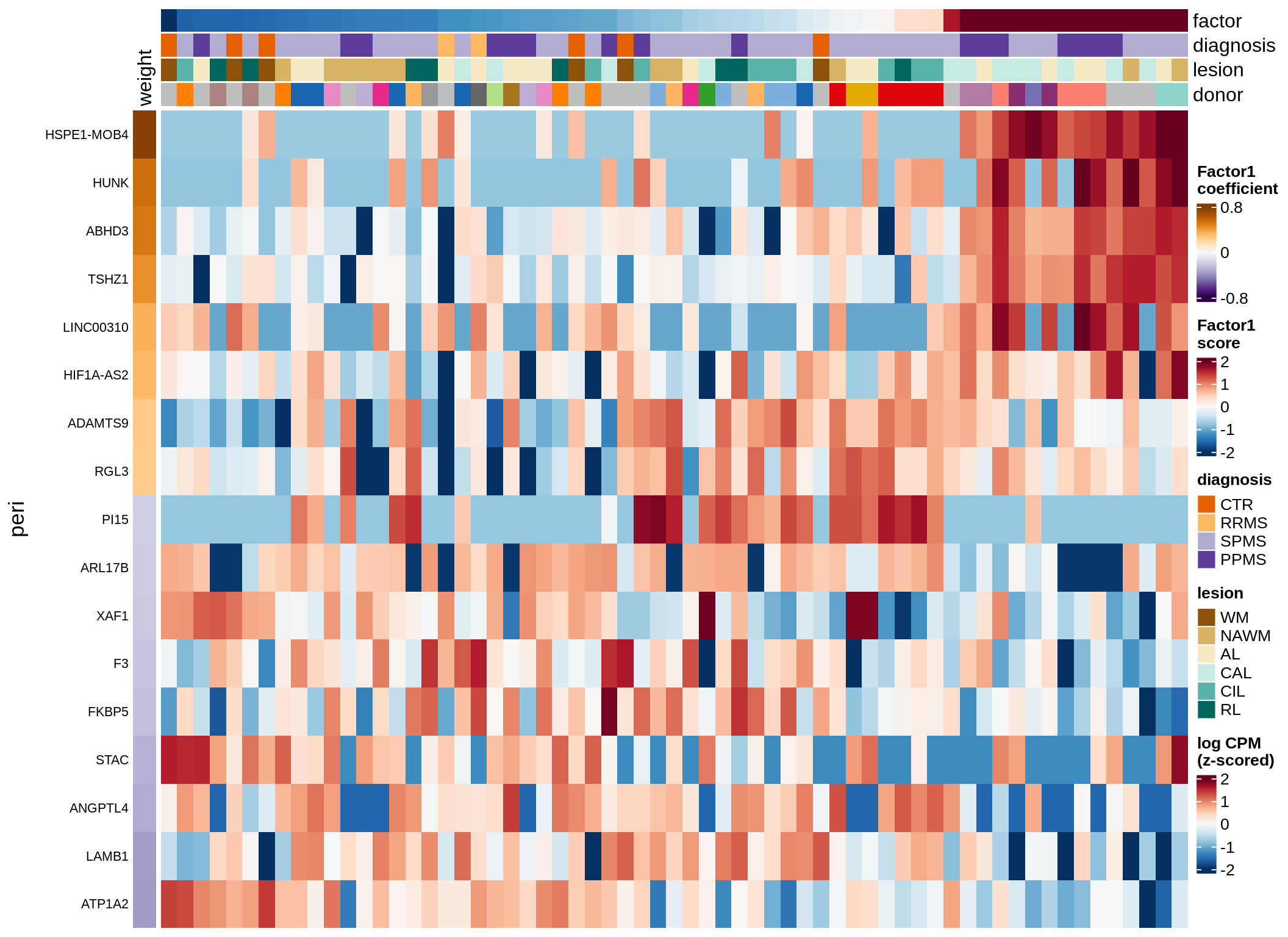
peri-F2 (18%)
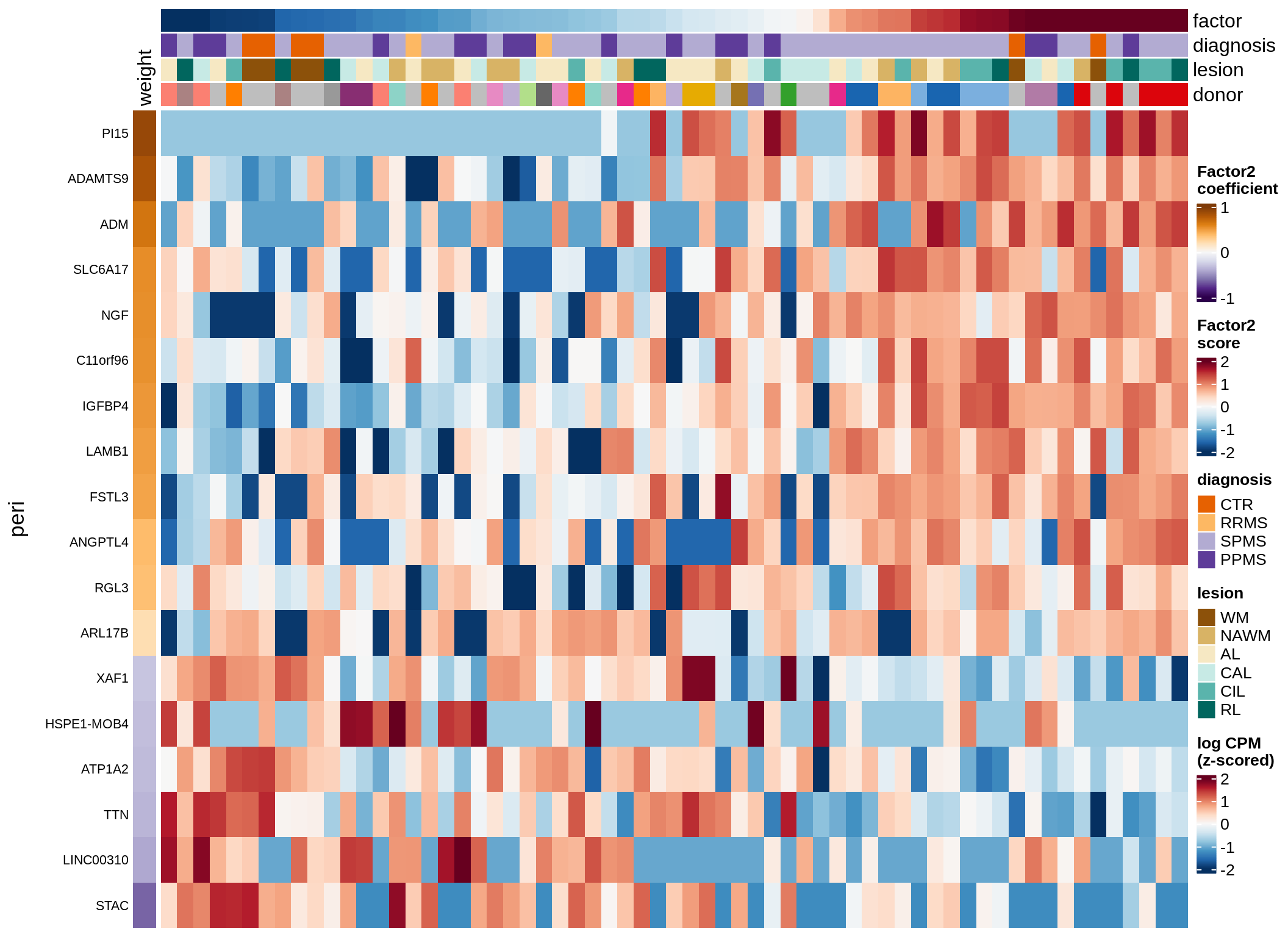
micro-F2 (18%)

astro-F2 (16%)
opc_cop-F2 (11%)

endo-F2 (7%)
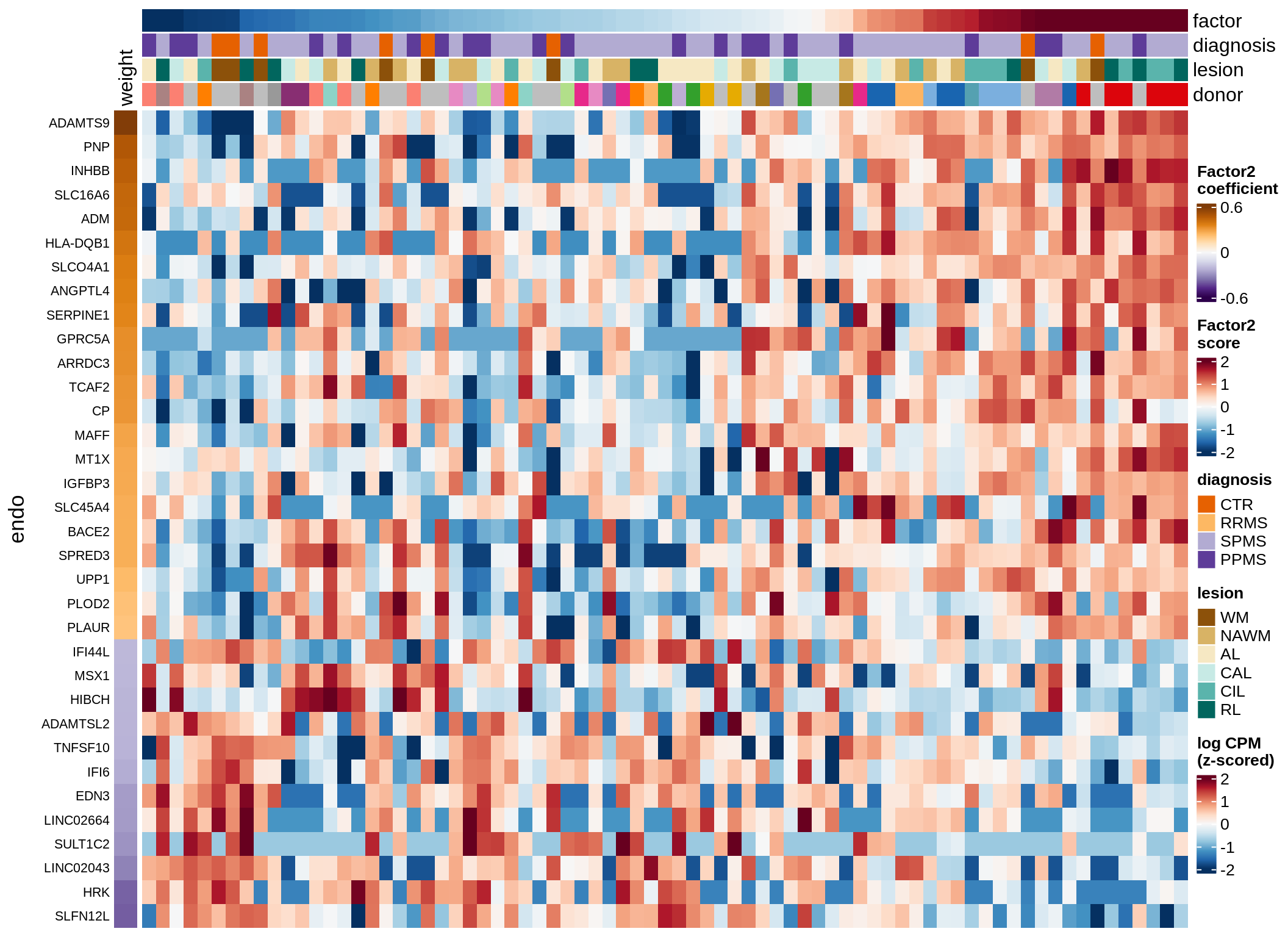
oligo-F2 (7%)
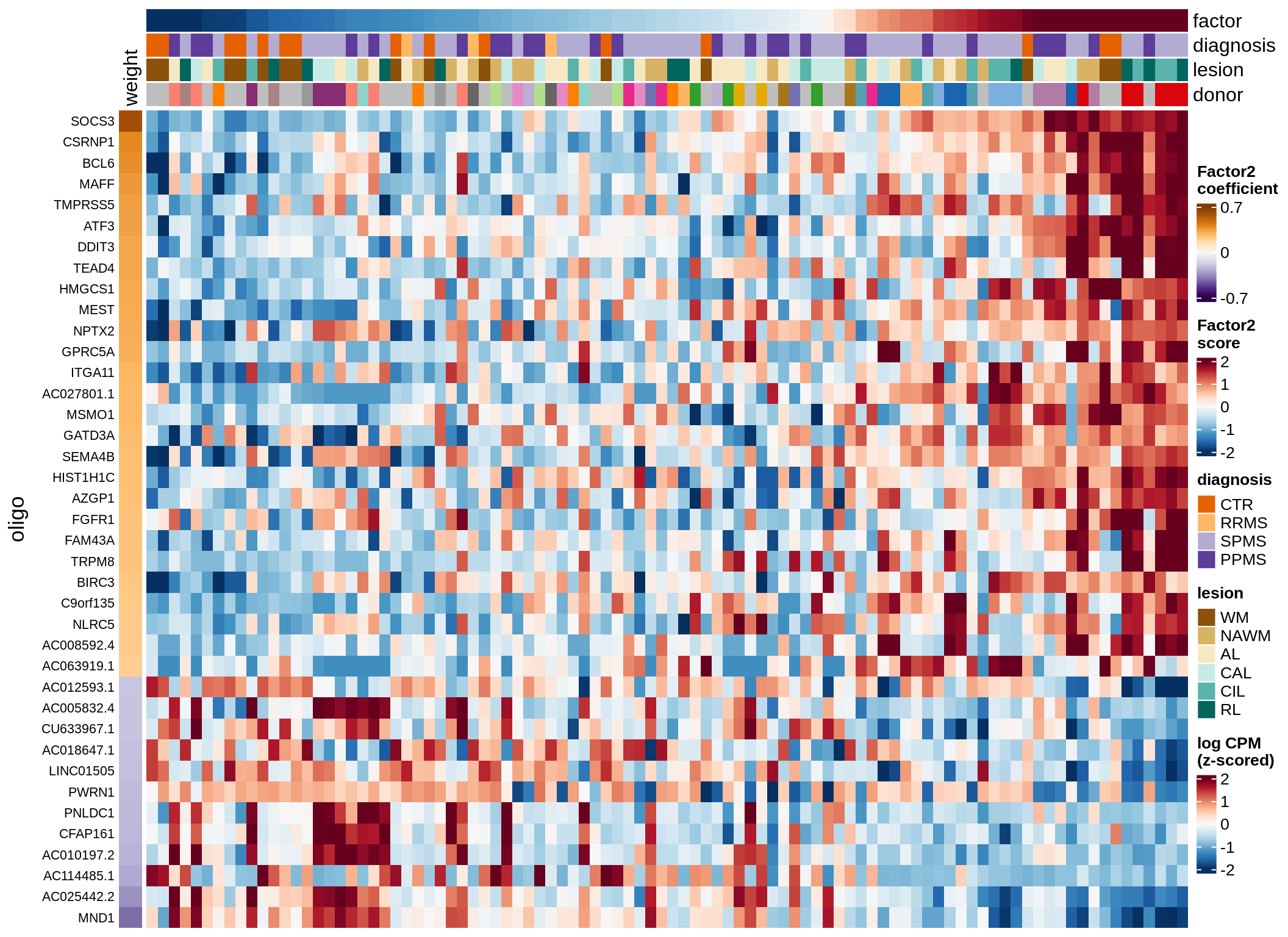
oligo-F3 (14%)
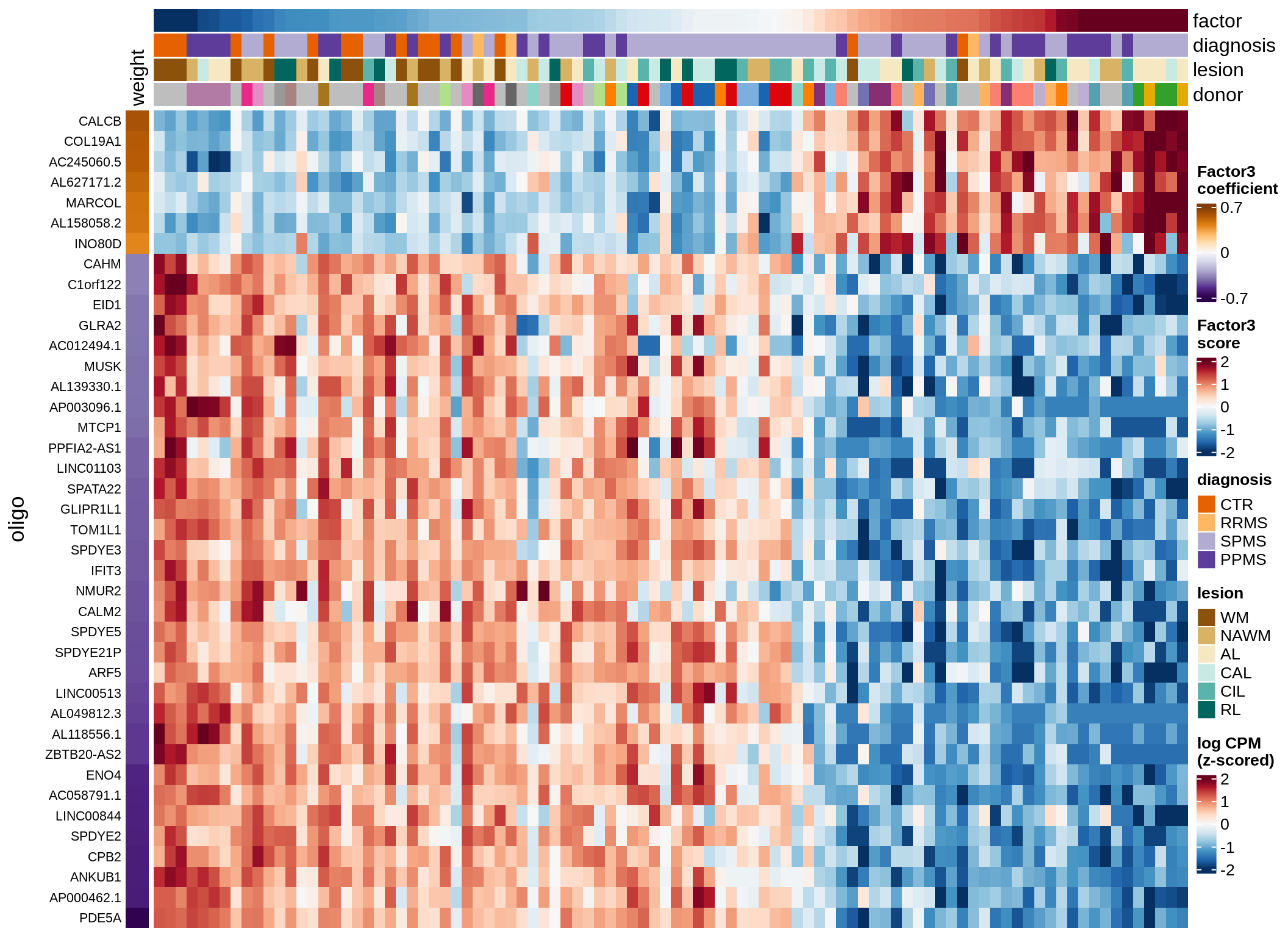
opc_cop-F3 (9%)
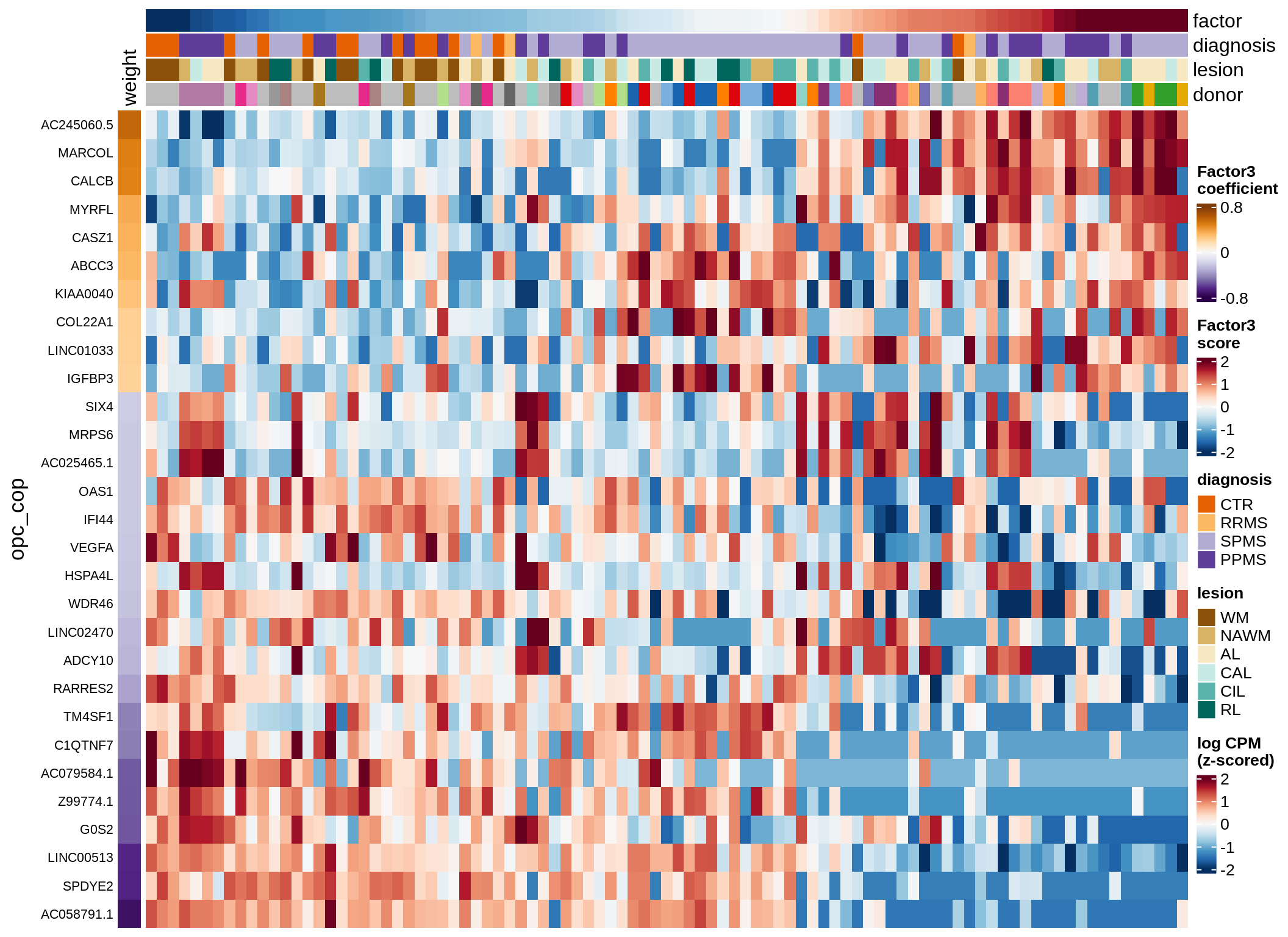
micro-F3 (9%)

peri-F3 (7%)

astro-F3 (7%)
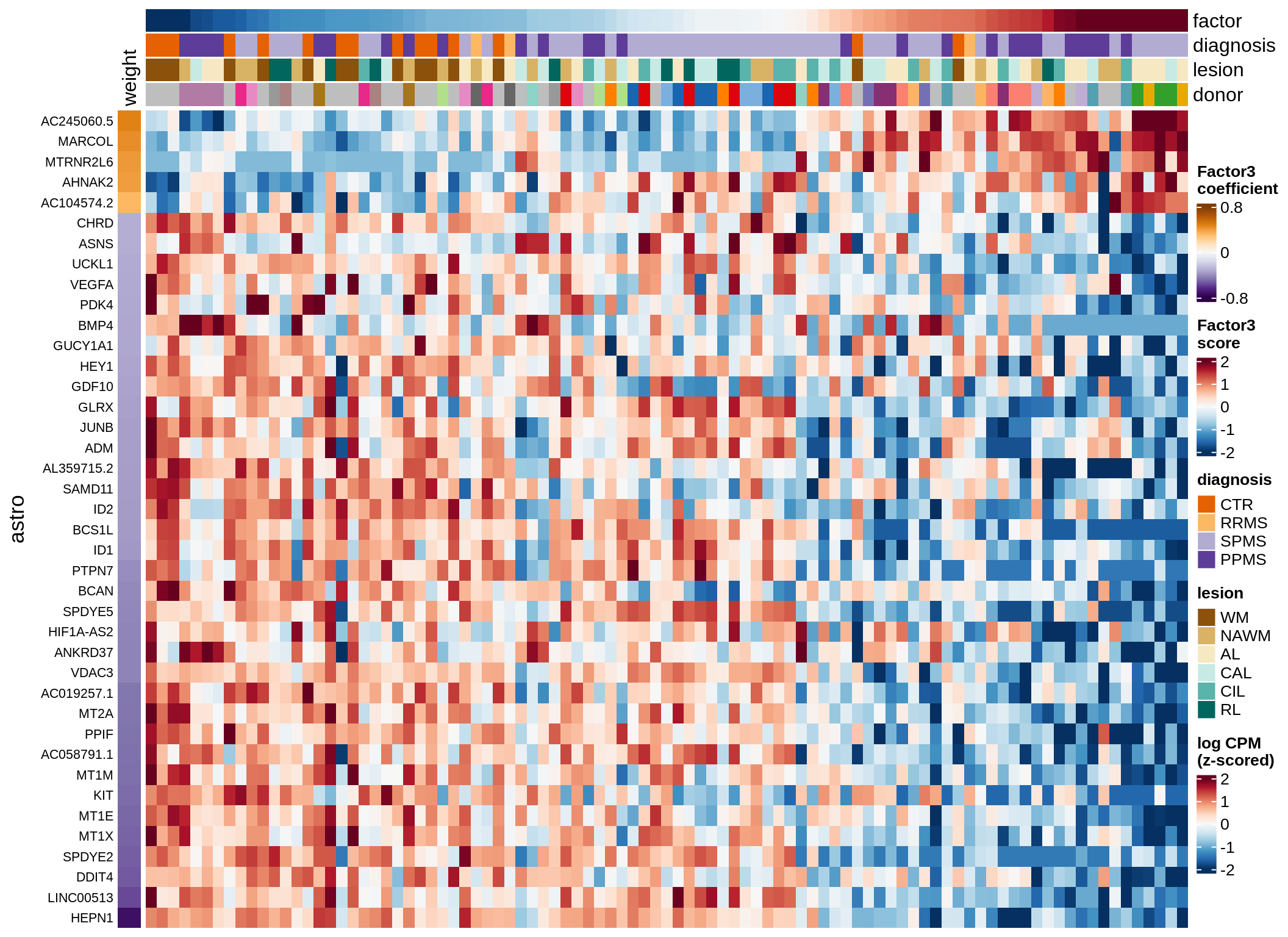
immune-F3 (6%)
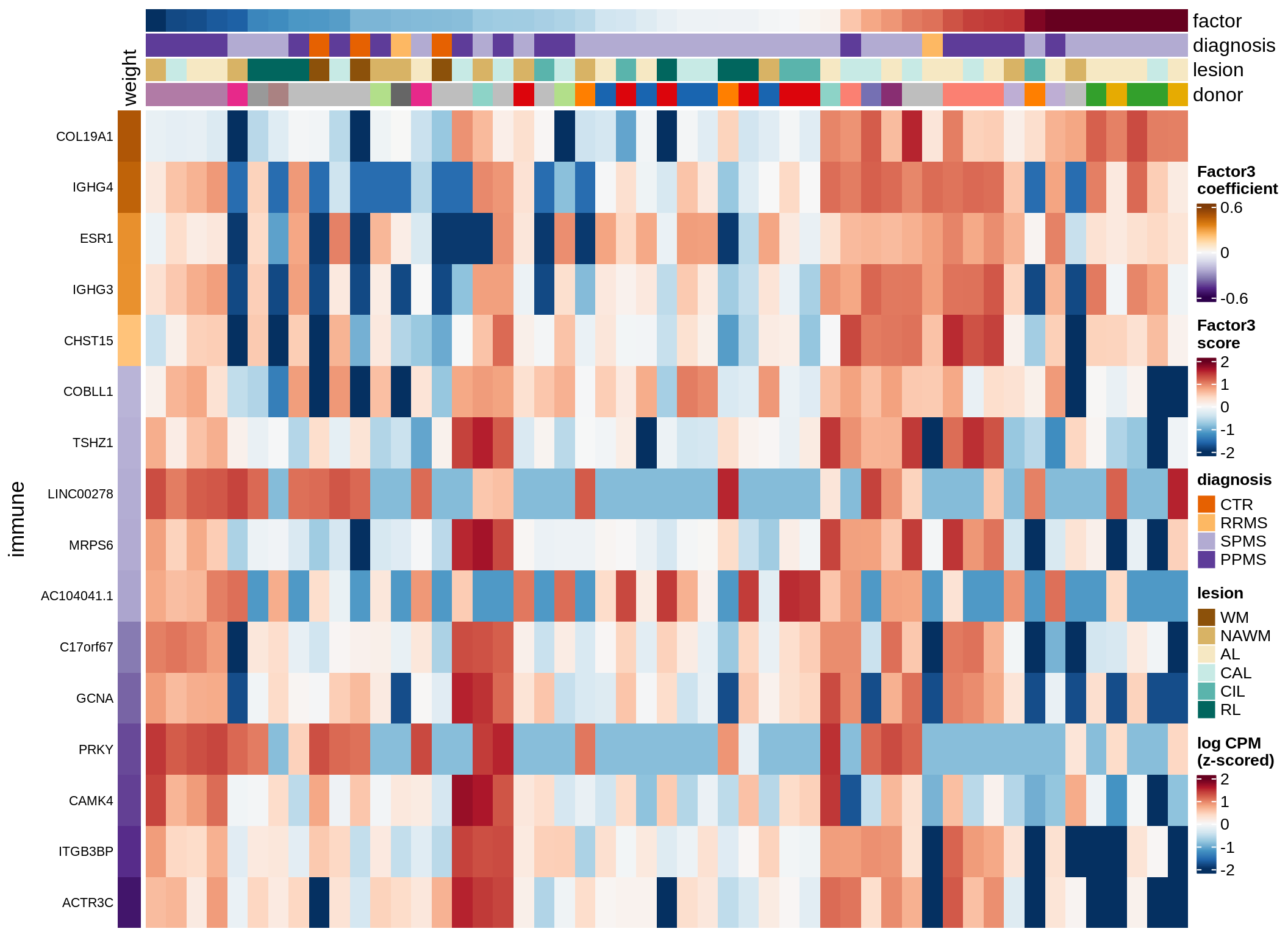
astro-F4 (10%)

oligo-F4 (5%)
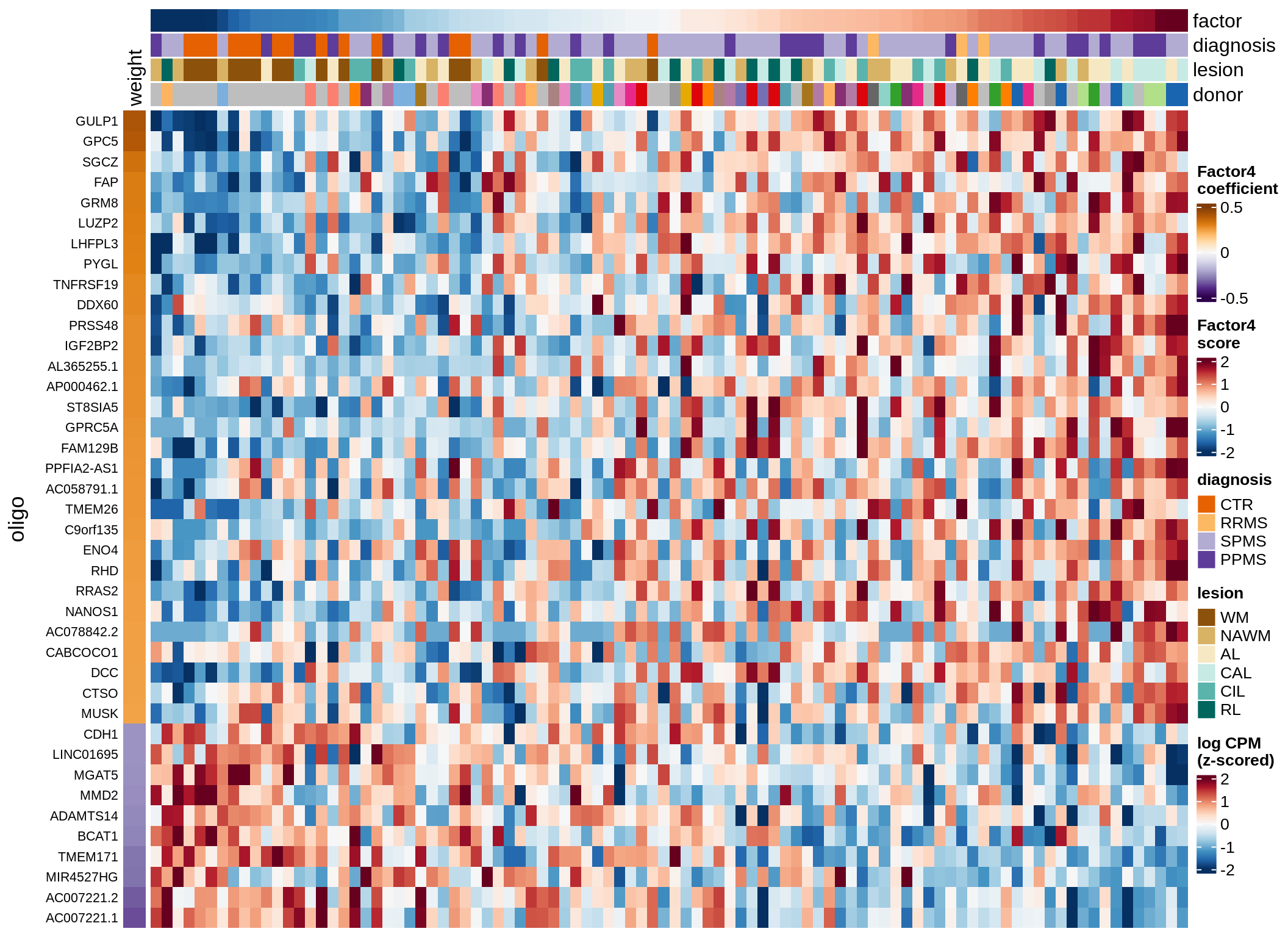
Expression of top genes across all factors per celltype
# iterate plots
for (sel_v in broad_short[sel_cl]) {
cat('### ', sel_v, '\n', sep = '')
draw(plot_top_genes_expression_all_factors(model, pb, annots_dt, filter_dt,
tfs_dt, var_exp_dt, sel_v = sel_v, n_top = 10, min_var), merge_legend = TRUE )
cat('\n\n')
}opc_cop
oligo
astro

micro
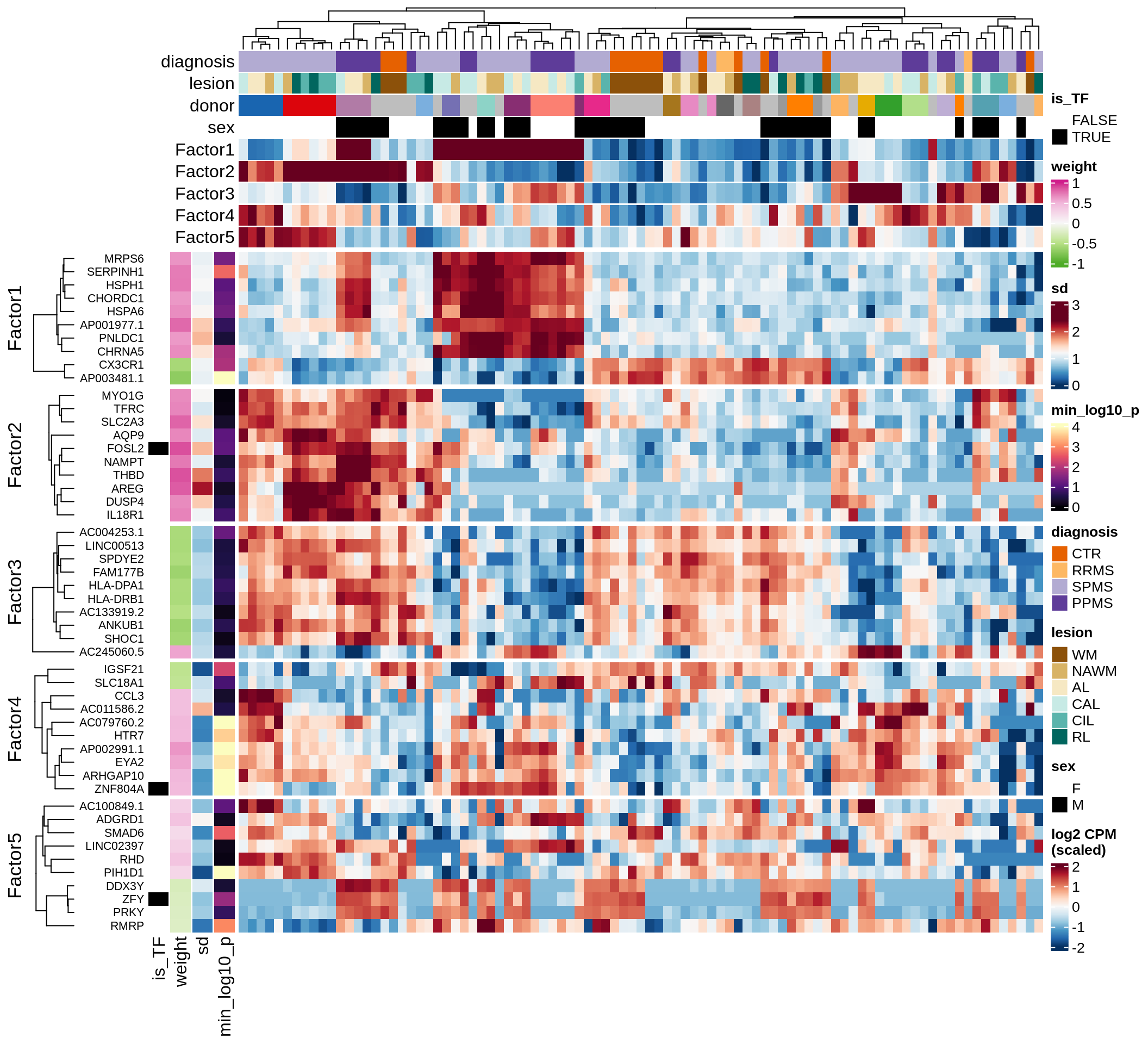
endo

peri

Factors vs number of cells
for (f in factors_names(model) ) {
cat('### ', f, '\n', sep = '')
print(plot_mofa_vs_n_cells(model, n_cells_dt, sel_f = f))
cat('\n\n')
}Factor1

Factor2

Factor3

Factor4

Factors vs top genes
for (f in factors_names(model)) {
cat('### ', f, '\n', sep = '')
print(plot_mofa_vs_logcpm(model, annots_dt, sel_f = f))
cat('\n\n')
}Factor1

Factor2

Factor3
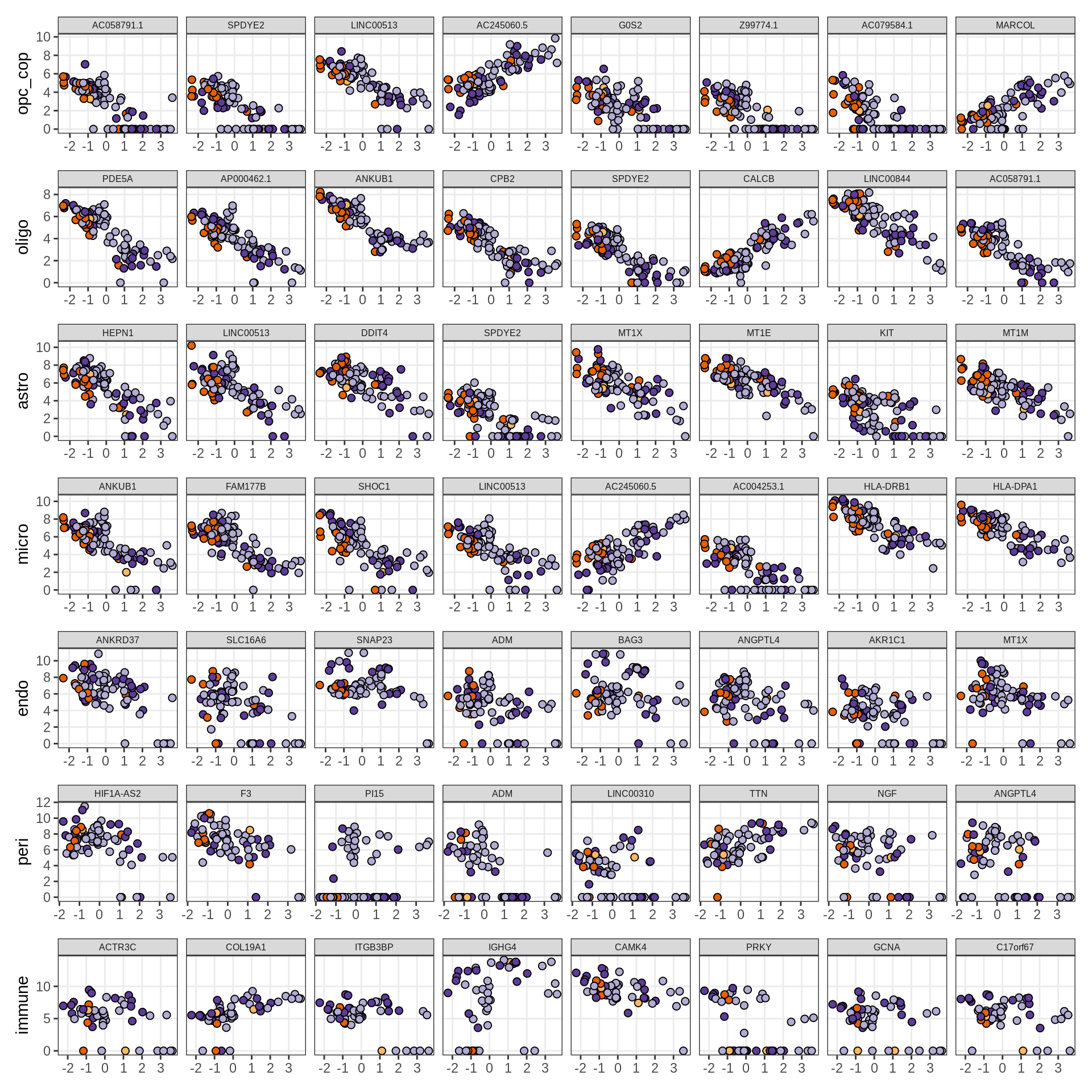
Factor4

Factors vs top genes - soup
for (f in factors_names(model) ) {
cat('### ', f, '\n', sep = '')
print(plot_mofa_vs_soup_logcpm(model, annots_dt, soup_dt,
sel_f = f, trans = 'linear'))
cat('\n\n')
}Factor1
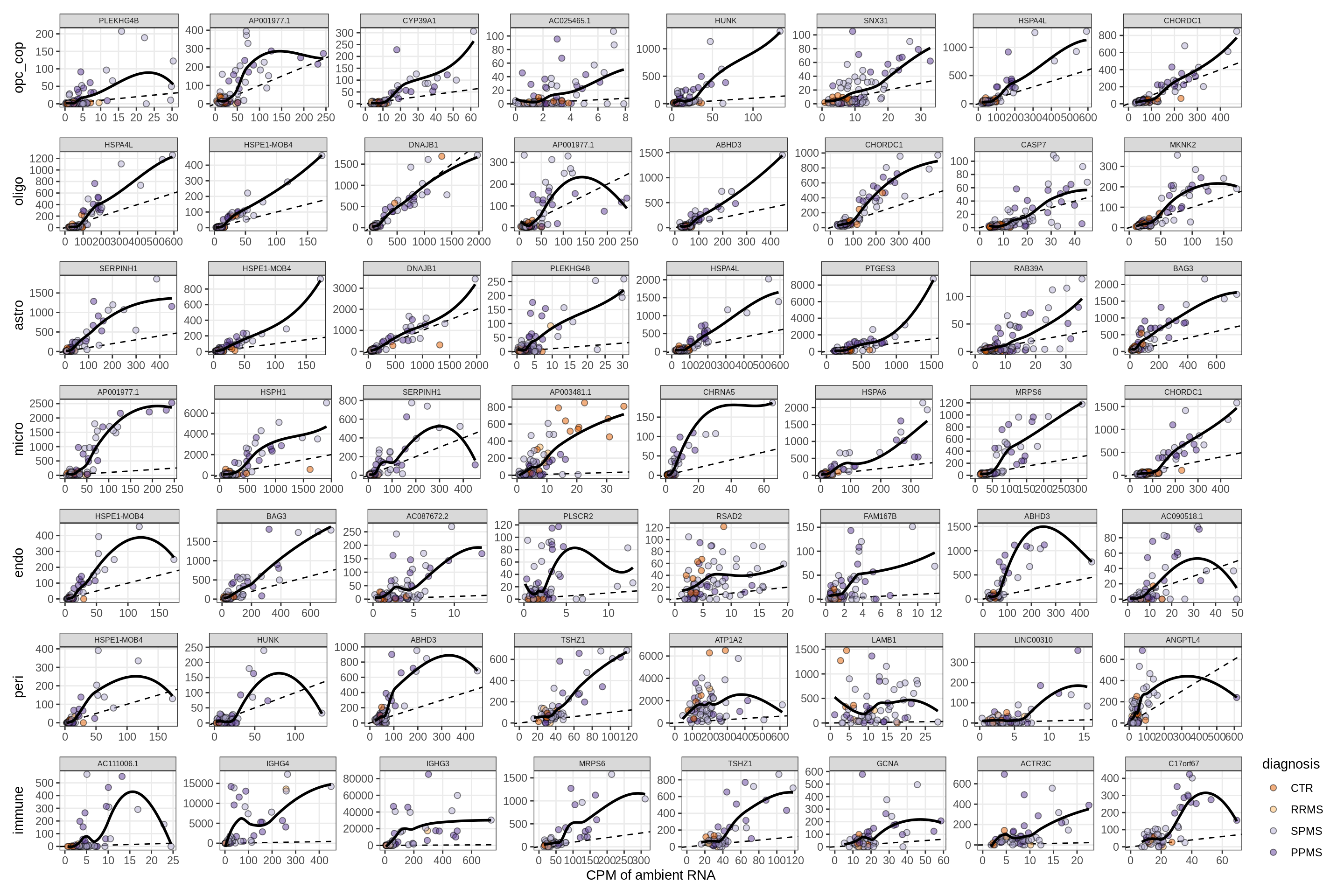
Factor2
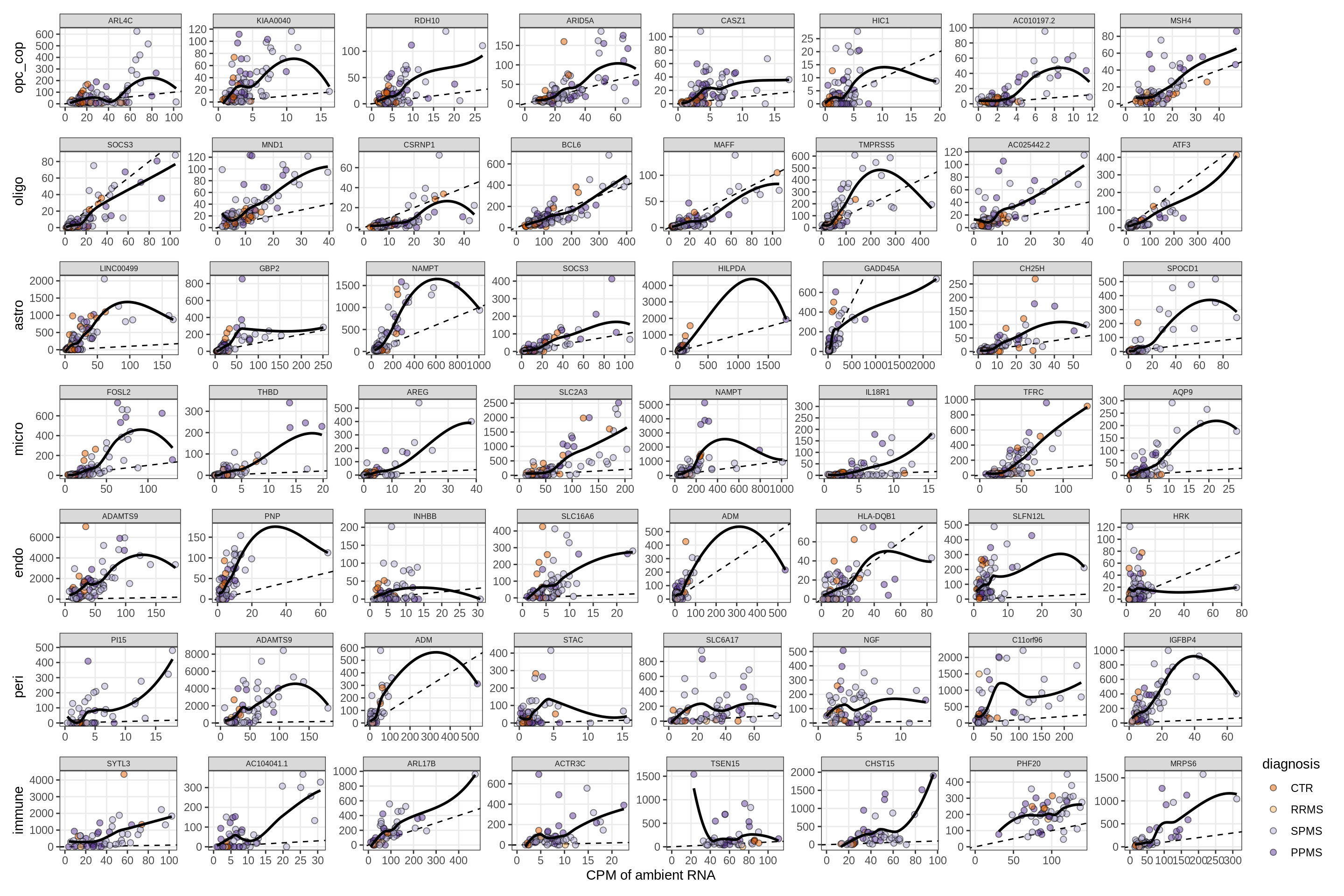
Factor3
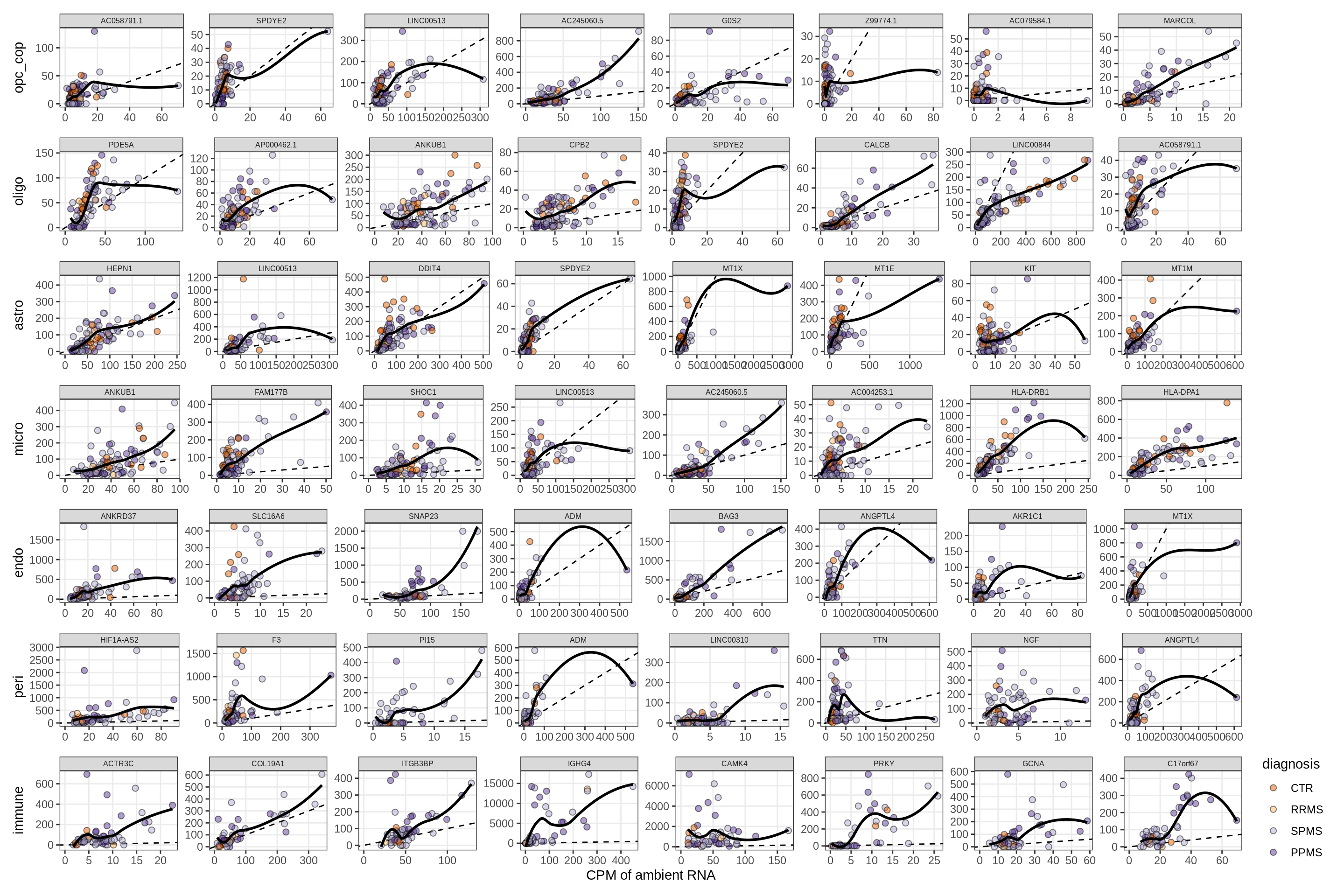
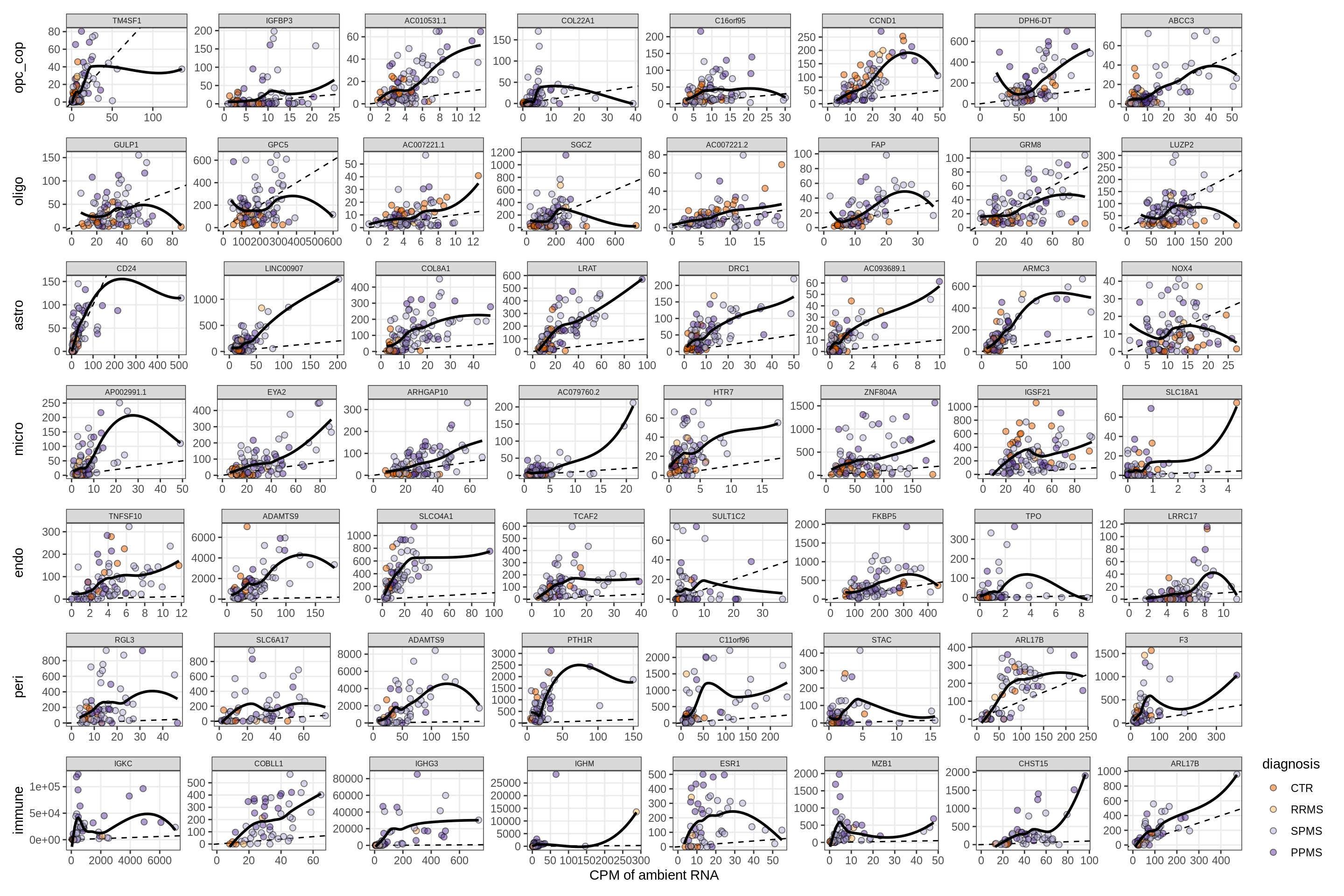
Factor4
Distributions of factor weights
(plot_mofa_weights(model))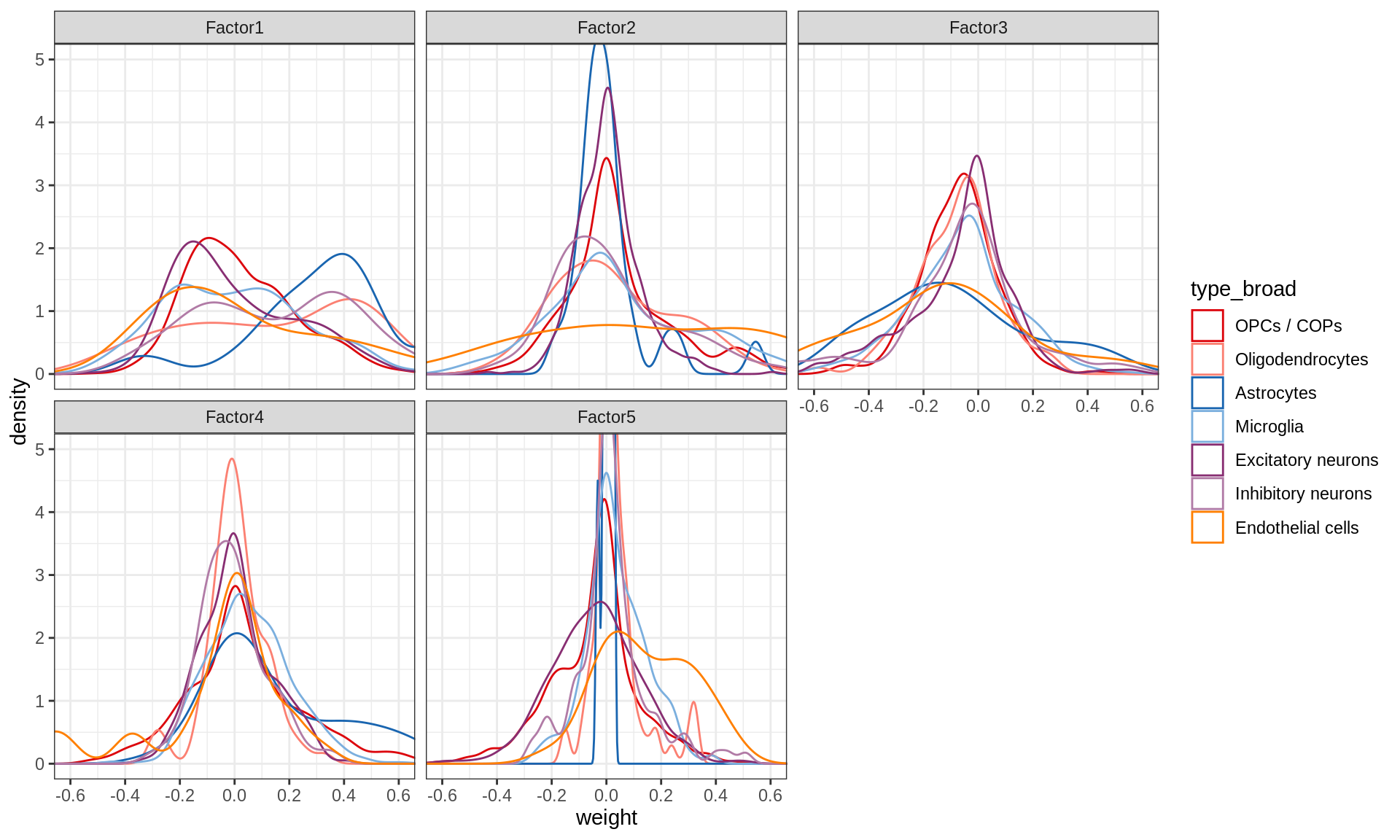
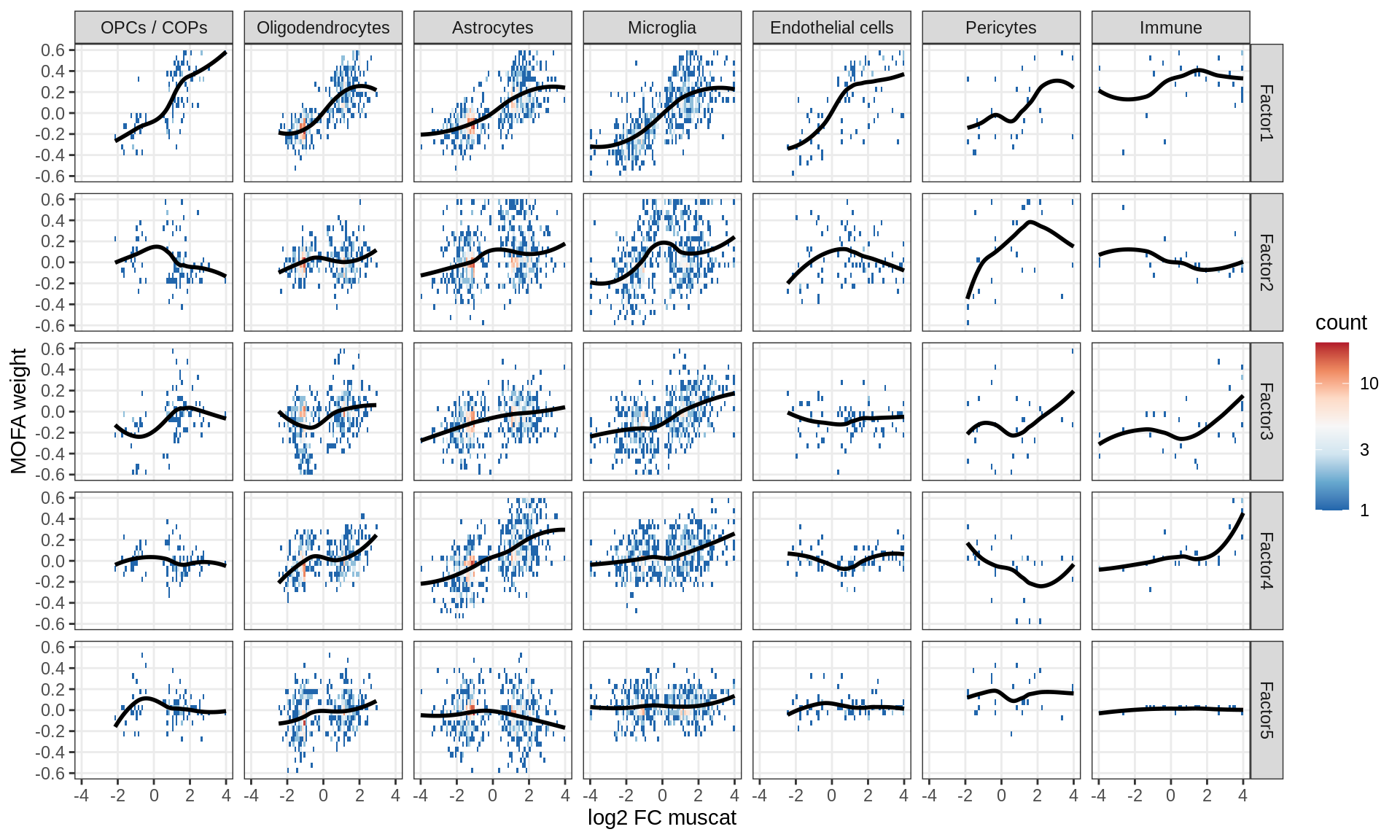
Factor weights vs muscat results
for (what in c('log10_padj', 'log2FC')) {
cat('### ', what, '\n', sep = '')
print(plot_muscat_vs_mofa(model, filter_dt, what = what))
cat('\n\n')
}log10_padj
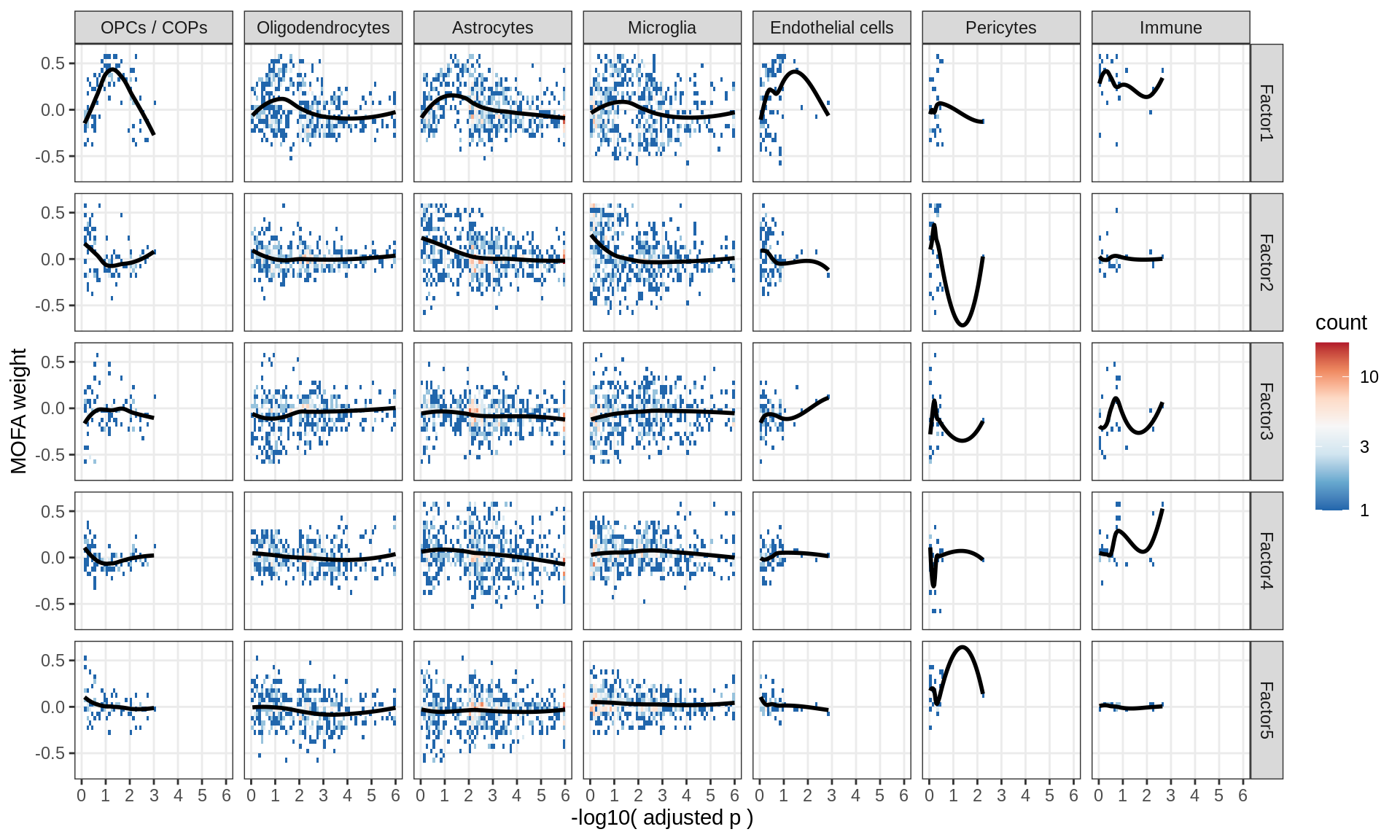
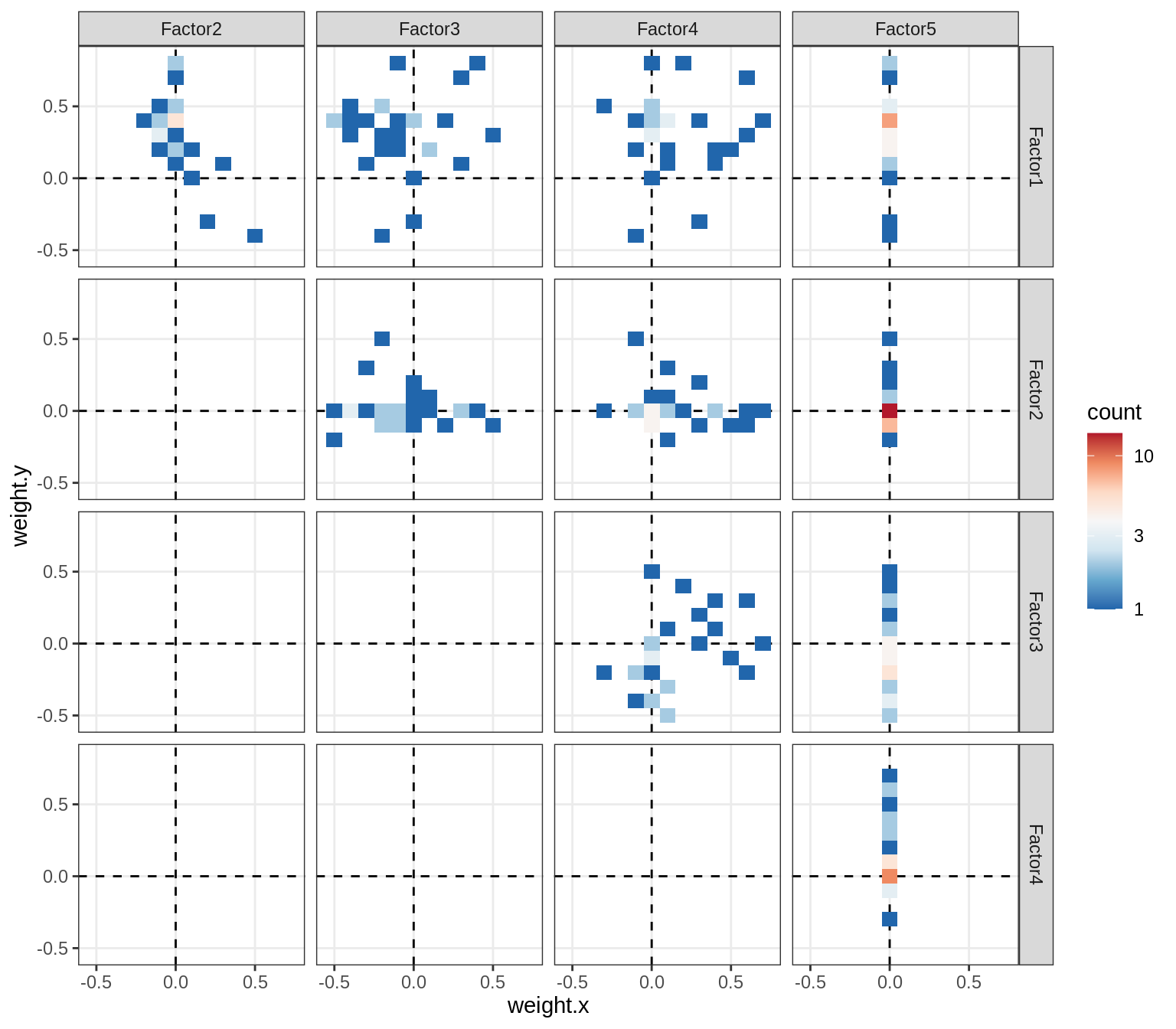
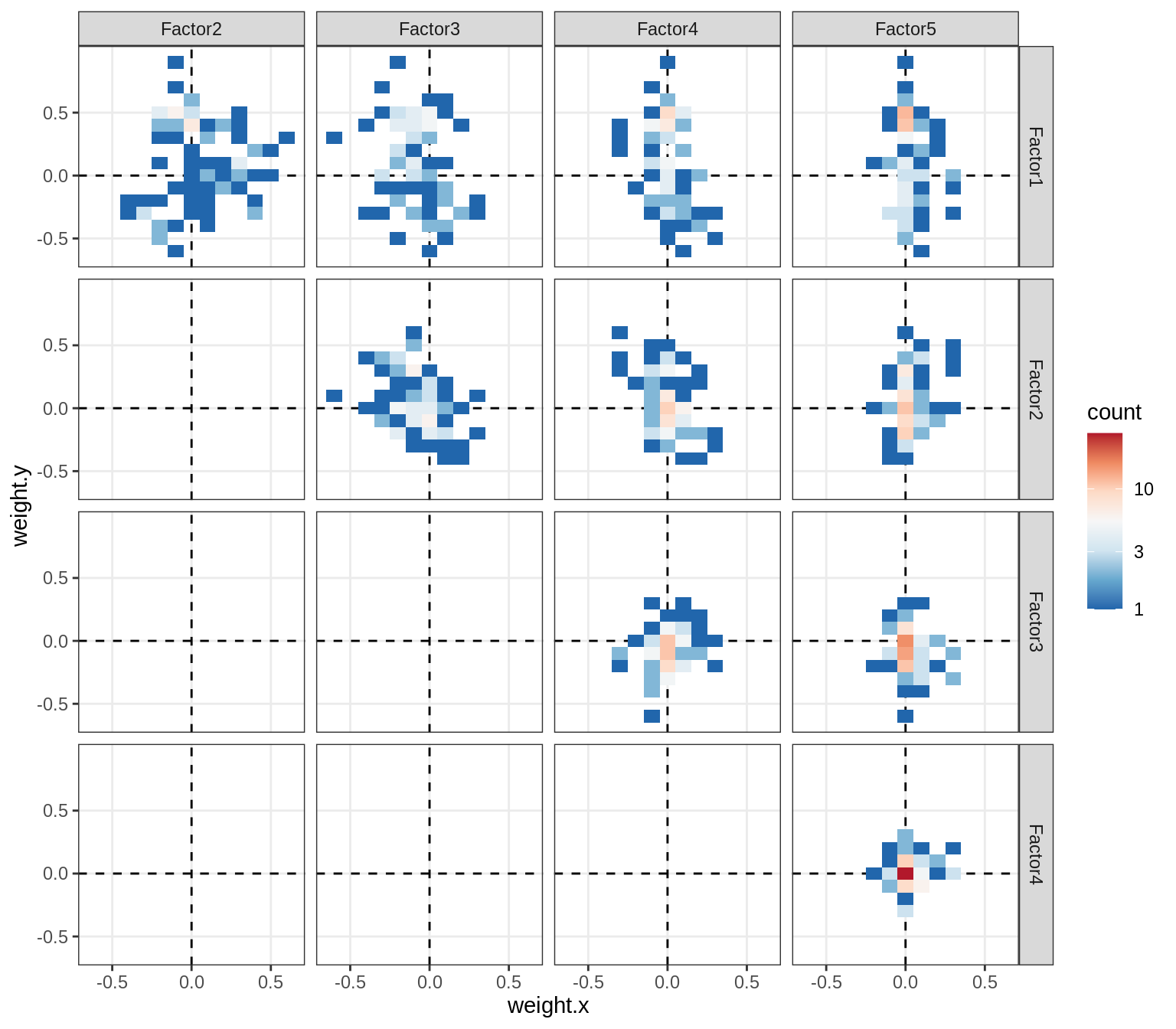
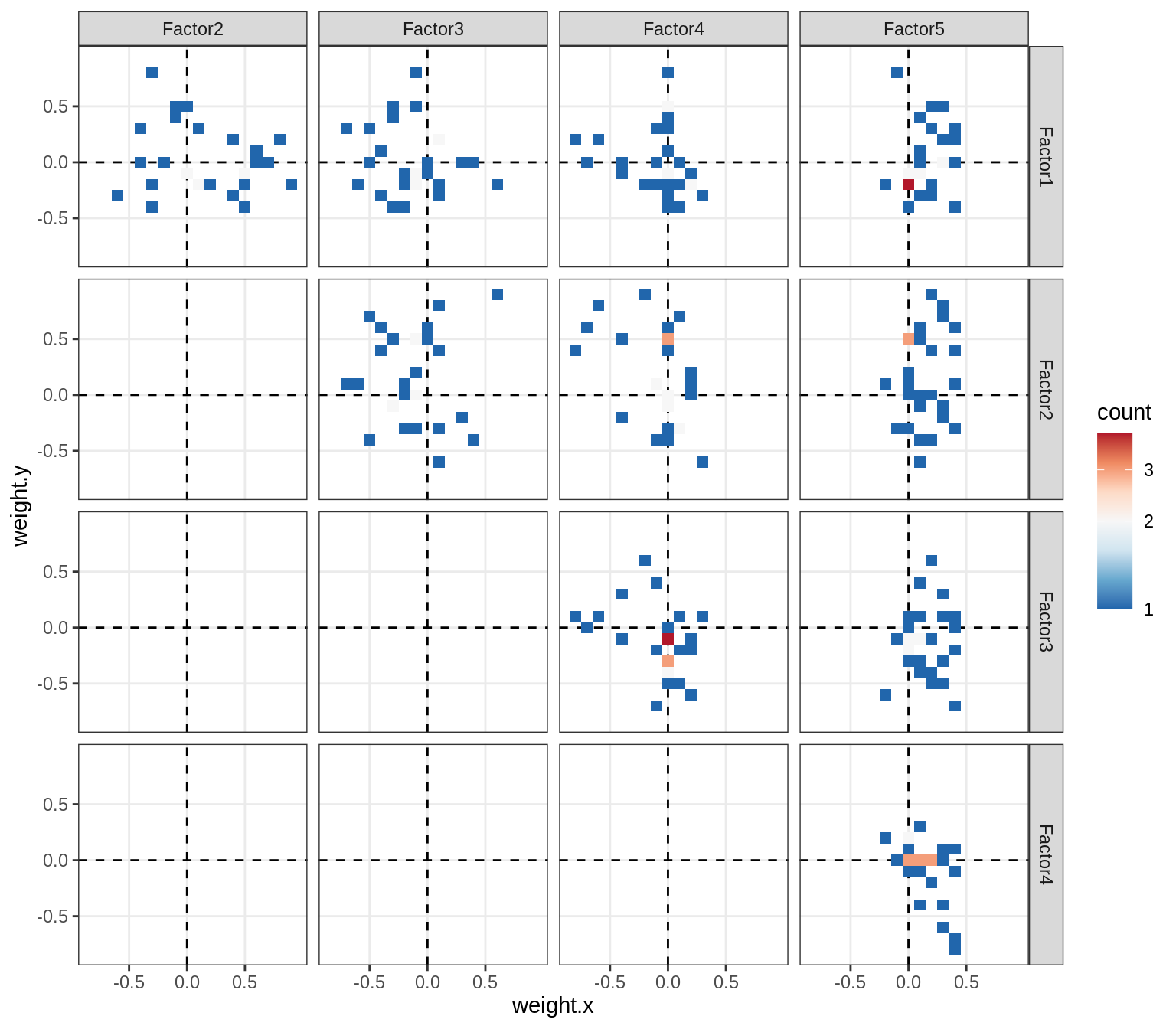
Correlations between factor weights - split by celltype
for (v in broad_short[sel_cl]) {
cat('### ', v, '\n', sep = '')
print(plot_factor_weight_corrs(model, v, by = 'type', how = 'bin'))
cat('\n\n')
}opc_cop
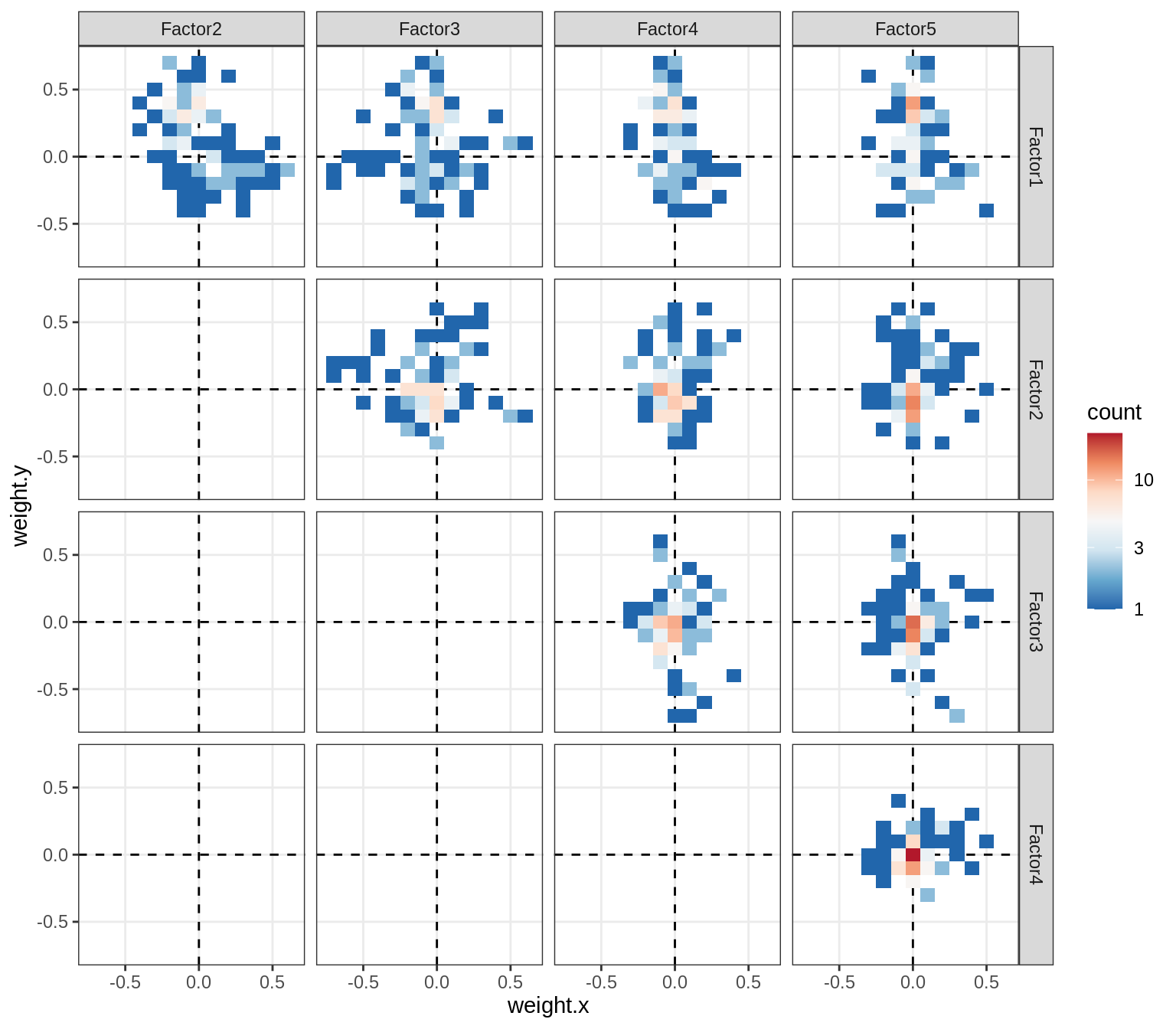
oligo
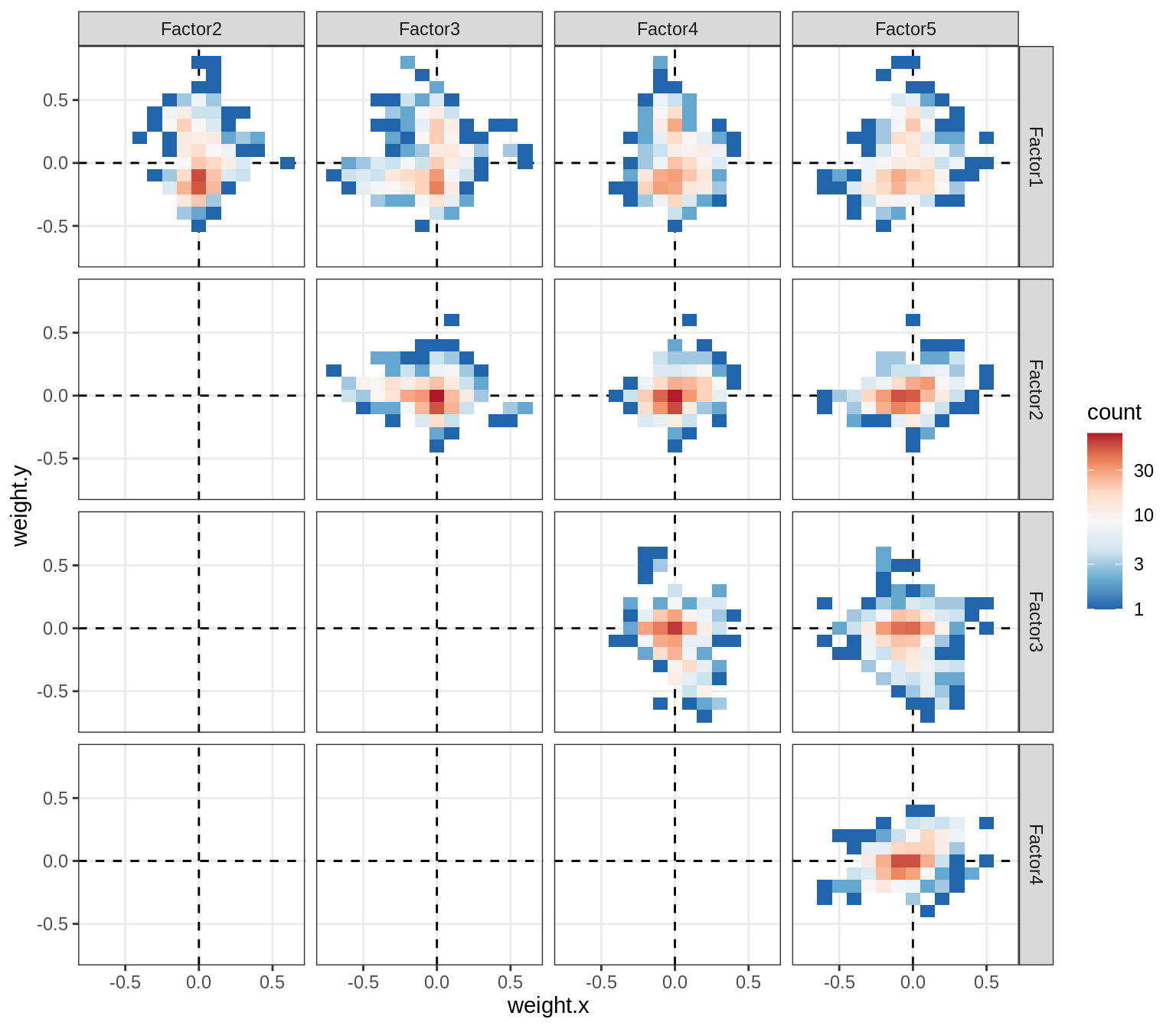
astro

micro

endo
peri
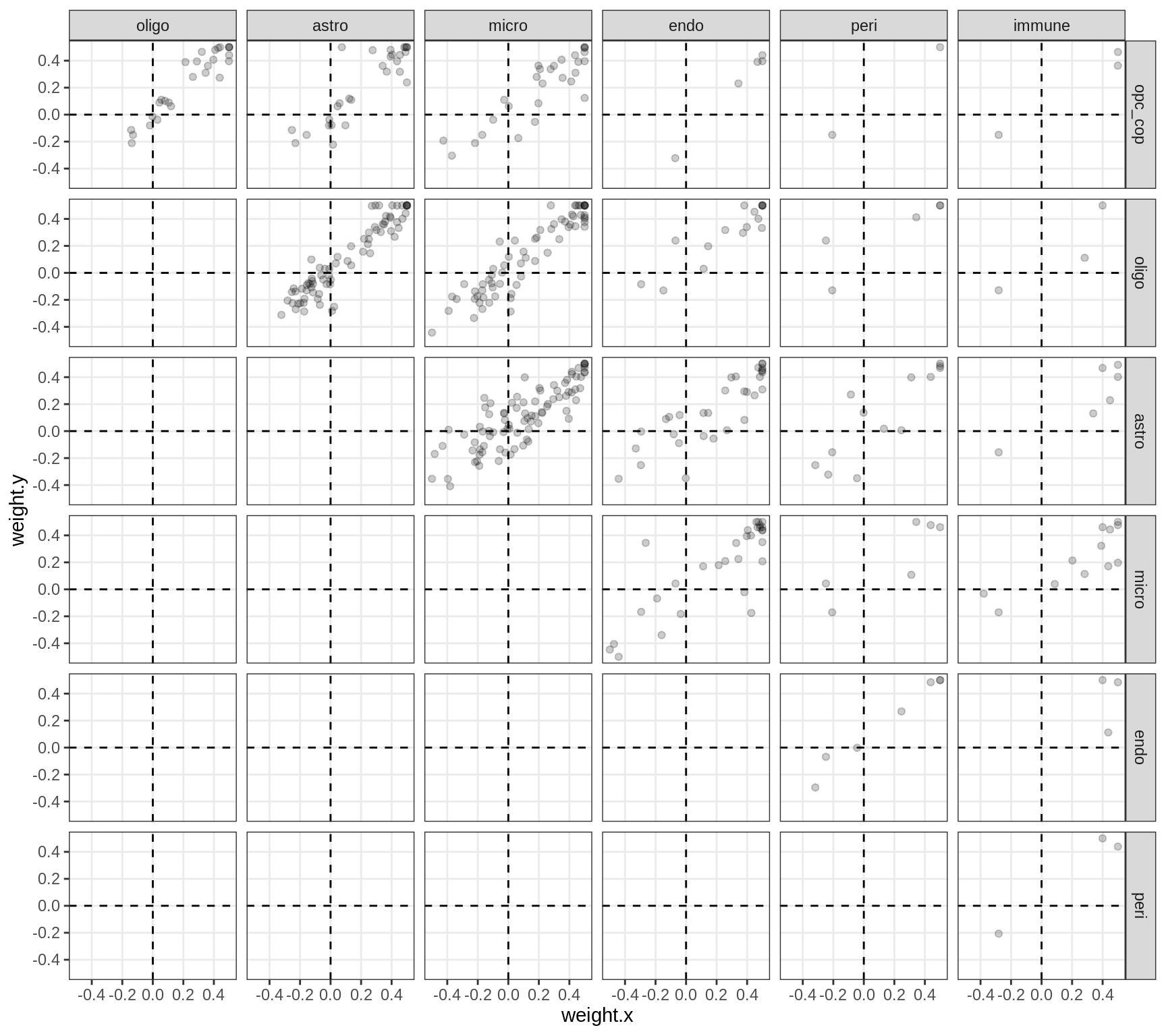
Correlations between factor weights - split by factor
for (f in factors_names(model) ) {
cat('### ', f, '\n', sep = '')
print(plot_factor_weight_corrs(model, f, by = 'factor', how = 'point'))
cat('\n\n')
}Factor1
Factor2
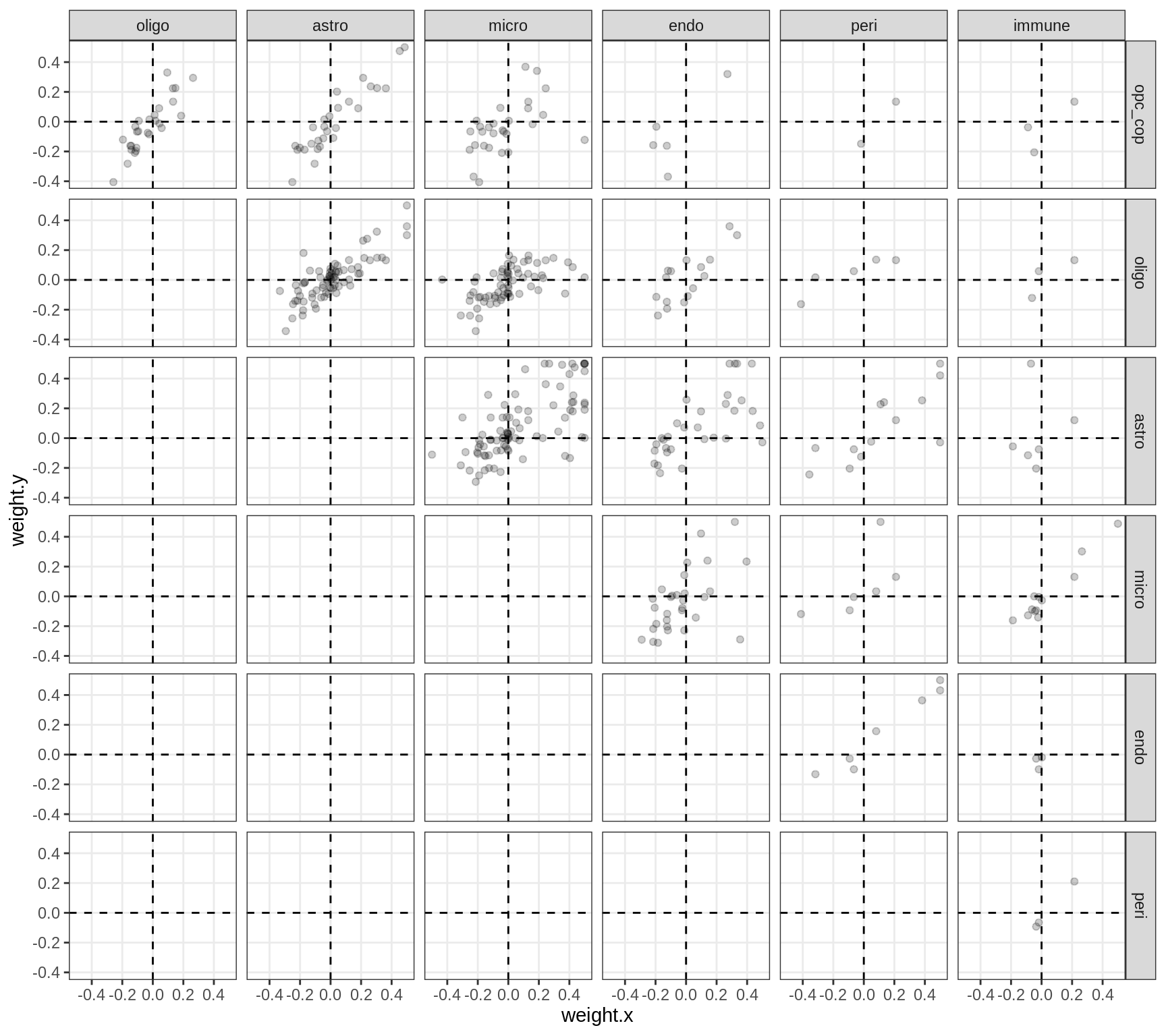
Factor3

Factor4

Variance explained
(plot_var_exp(var_exp_dt))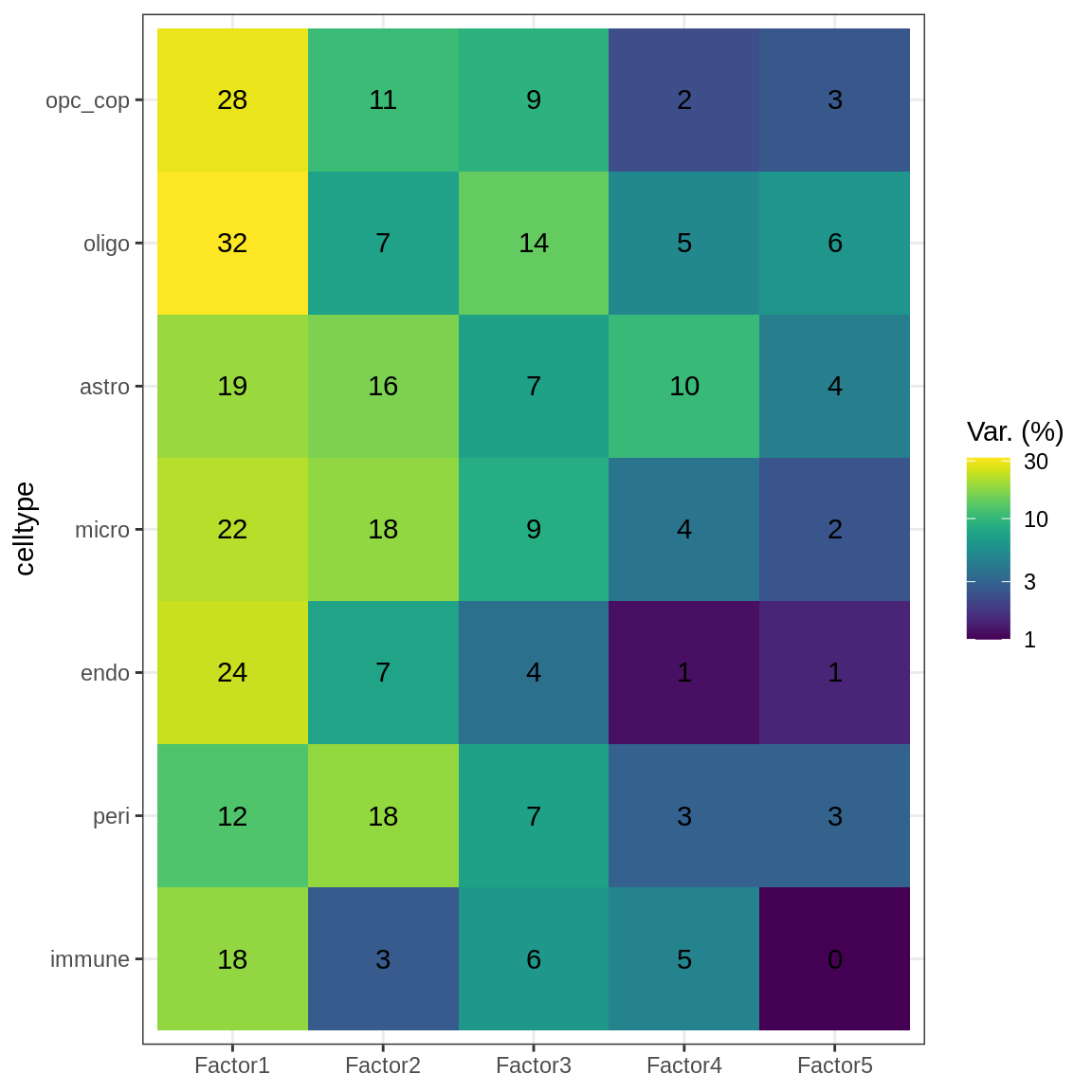
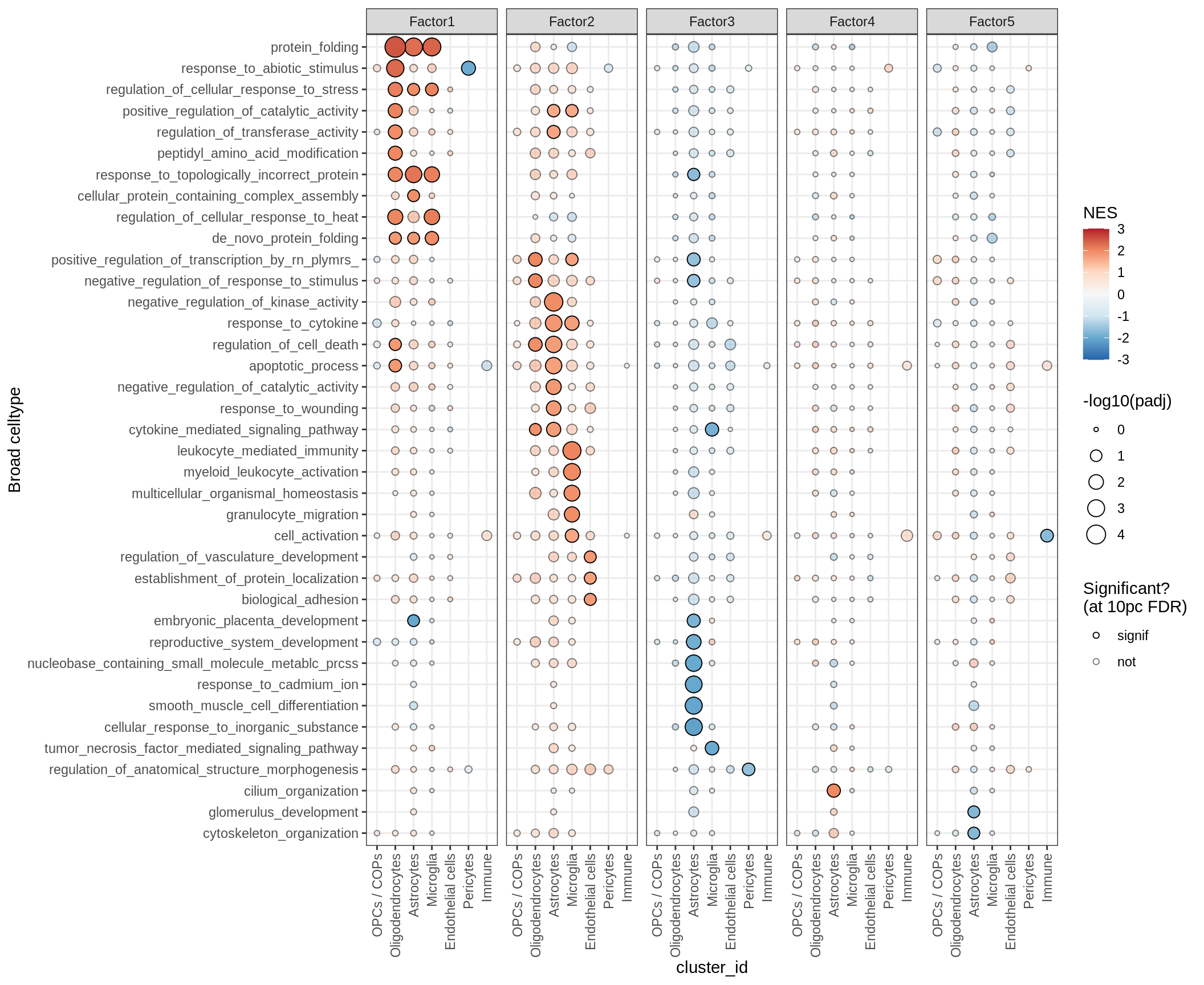
GSEA for factors
for (p in names(gsea_list)) {
# restrict to relevant GO terms
cat('### ', p, '\n', sep='')
dt = gsea_list[[p]]
if (nrow(dt[ main_path == TRUE ]) == 0)
next
# plot
print(plot_mofa_gsea_dotplot(dt, labels_dt,
fgsea_cut = fgsea_cut, n_total = 60))
cat('\n\n')
}go_bp
go_cc
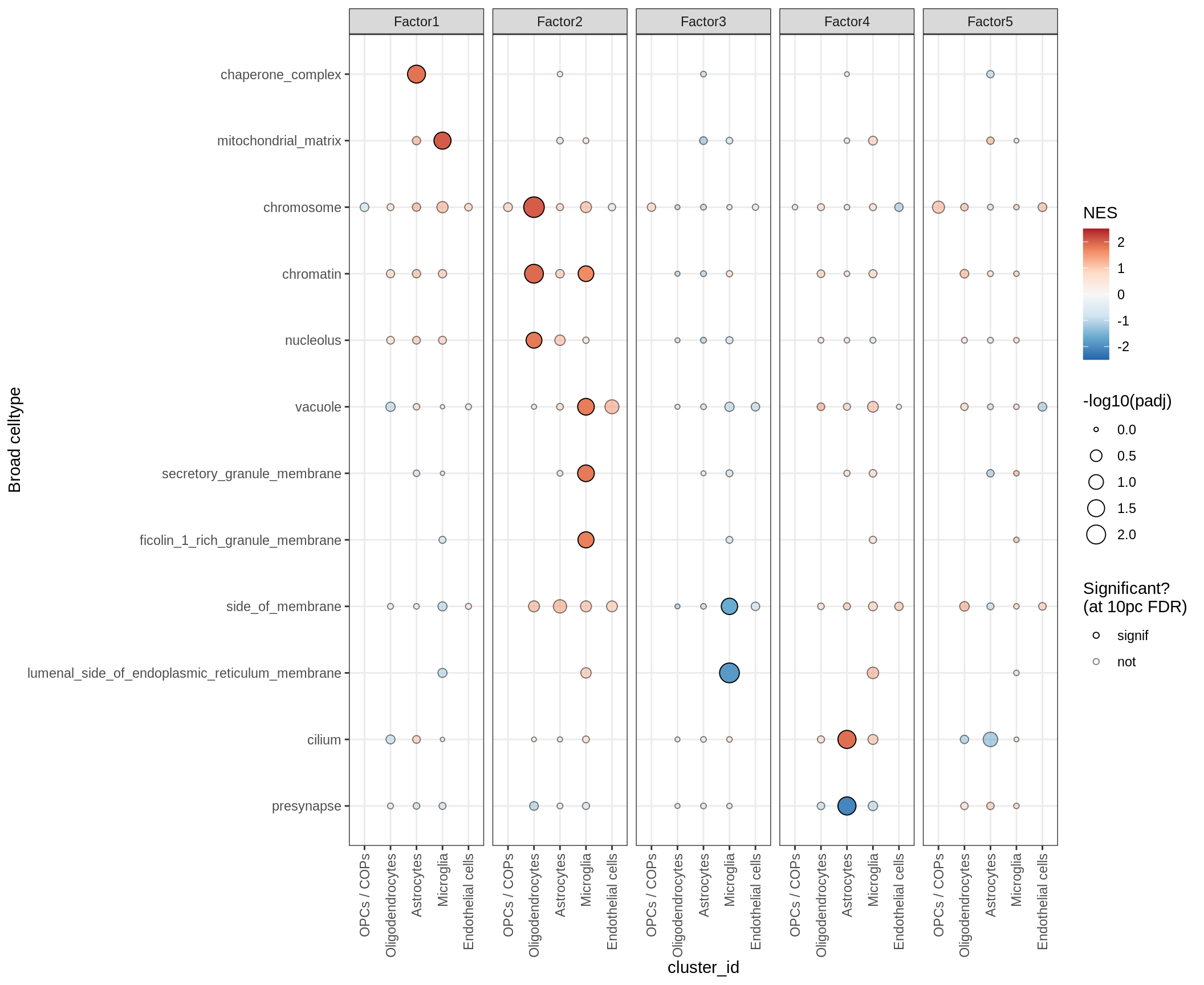
go_mf

hallmark
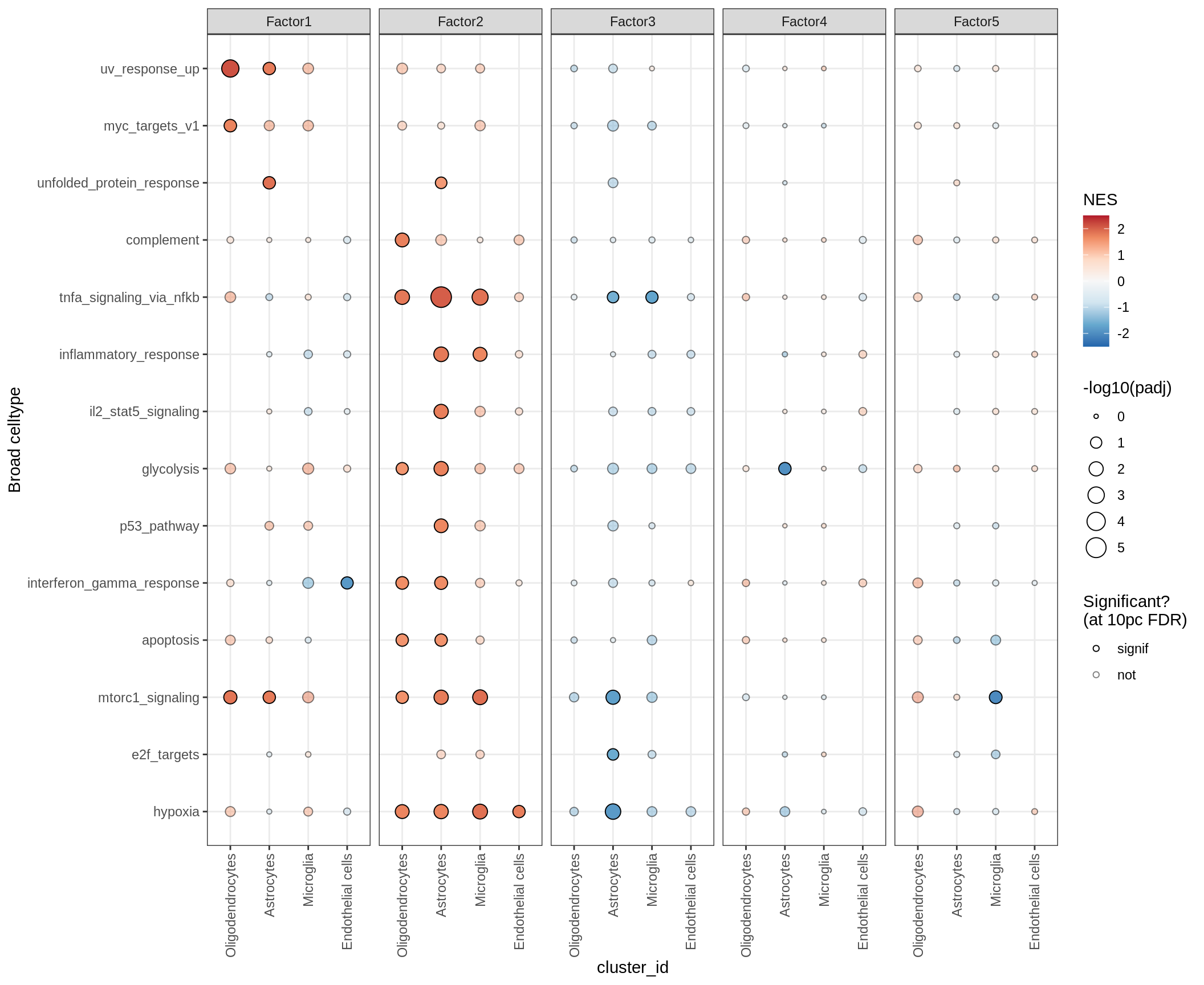
Outputs
Top filter genes
# merge filtered and weights
xls_dt = calc_xls_dt(model, filtered_dt)
# save outputs
write_xlsx(list(mofa_weights = xls_dt), path = interesting_f)Figures
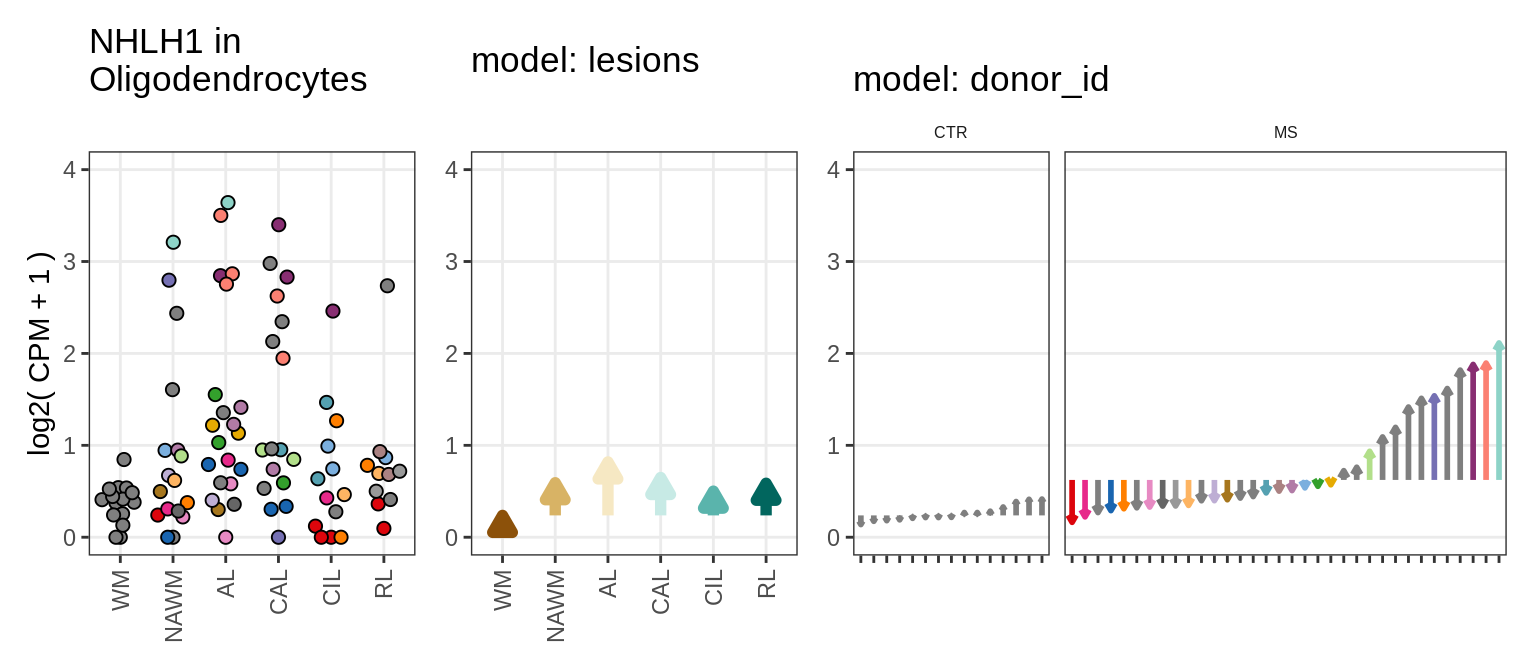
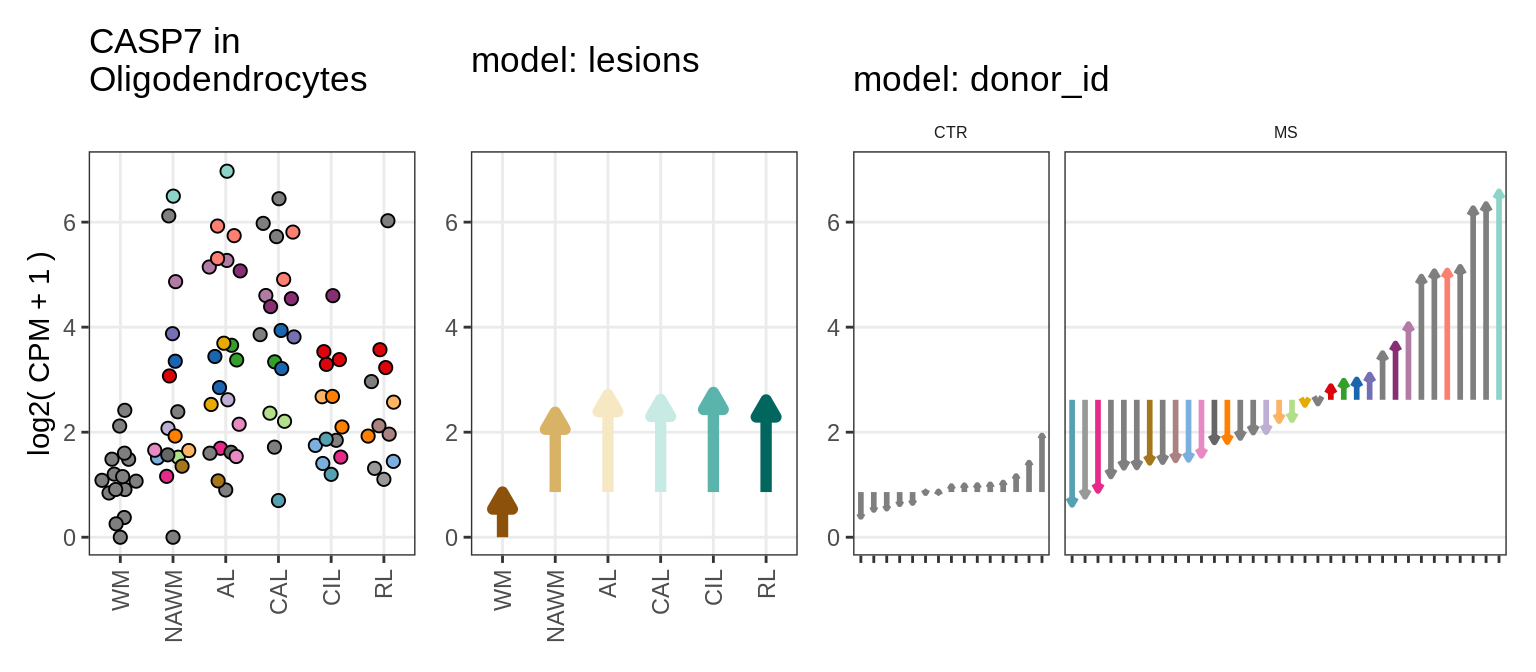
Illustrative example
for (g in example_gs) {
cat('### ', str_extract(g, '^[^_]+'), '\n', sep = '')
suppressWarnings(print(plot_ranef_example(pb, example_cl, g)))
cat('\n\n')
}NHLH1
CASP7
RELN
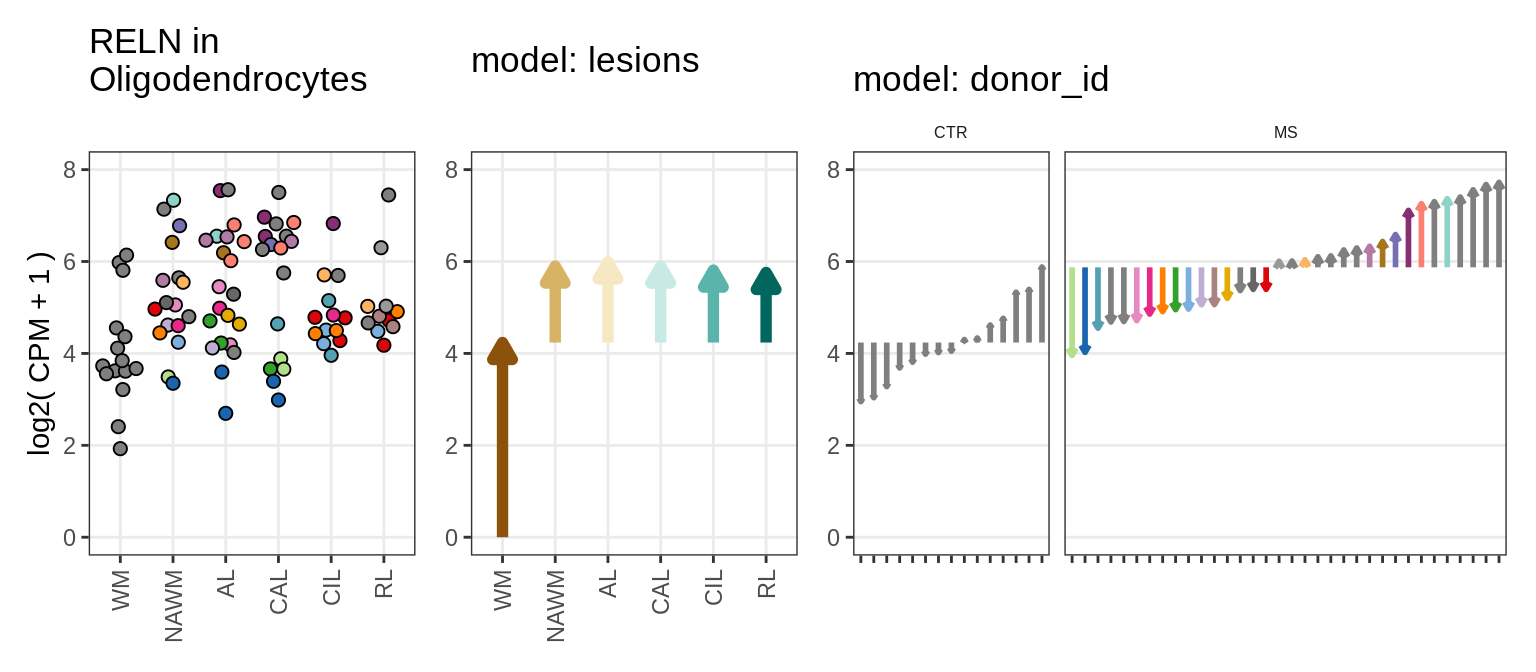
KLB
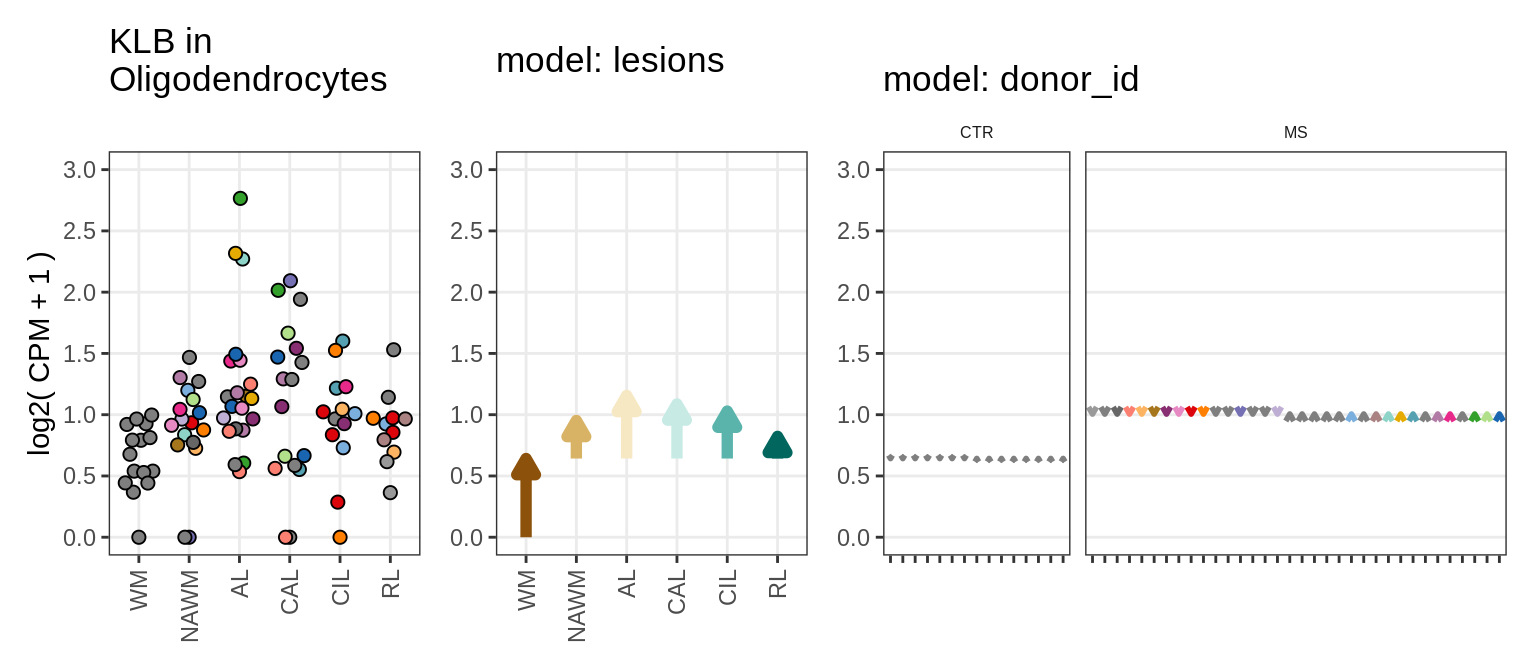
NRTN
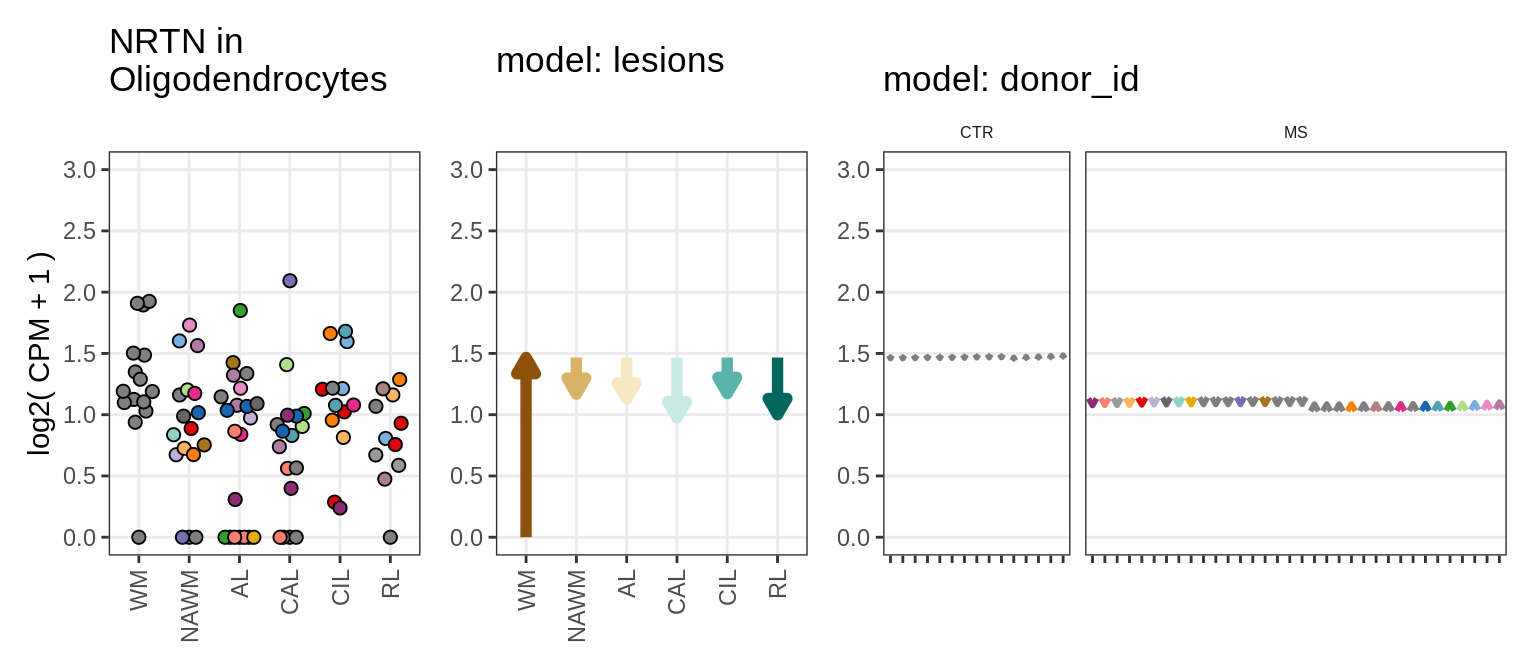
EVI5L
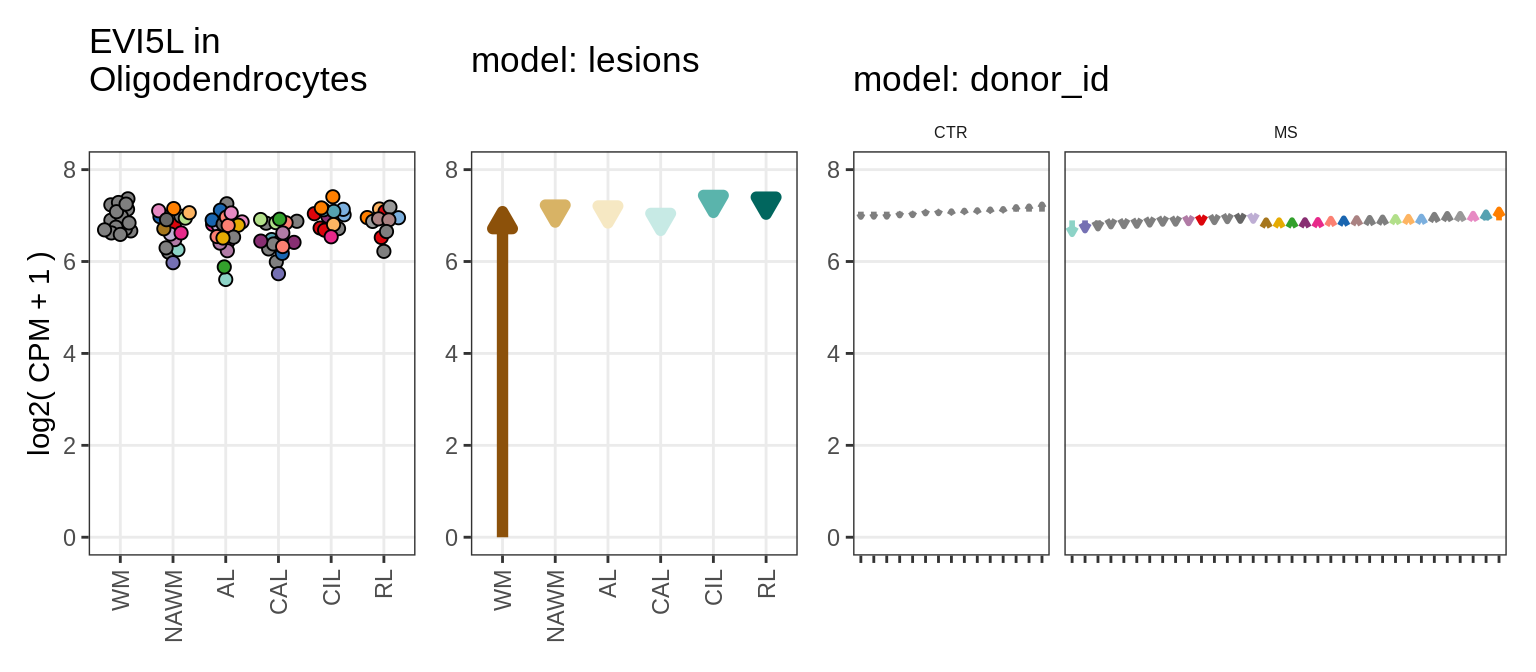
PWP2
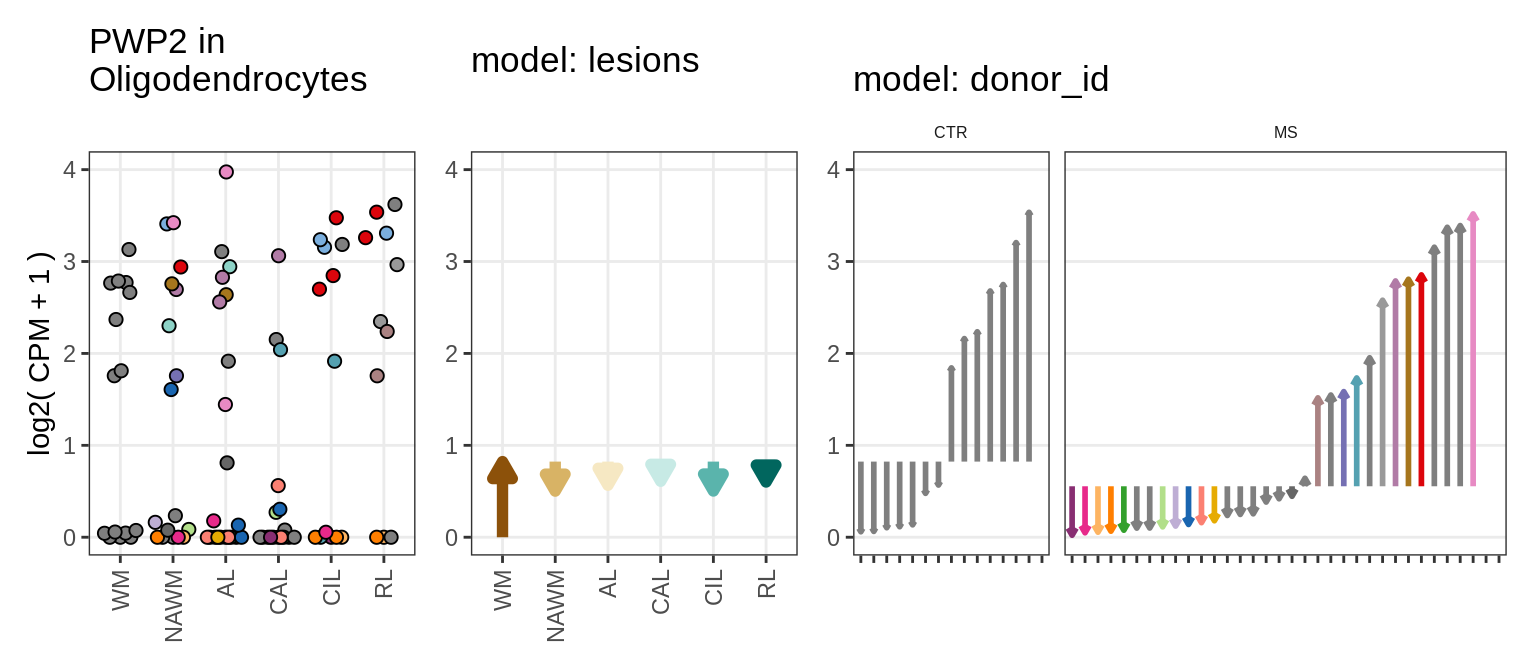
GRID2
Selection of interesting genes
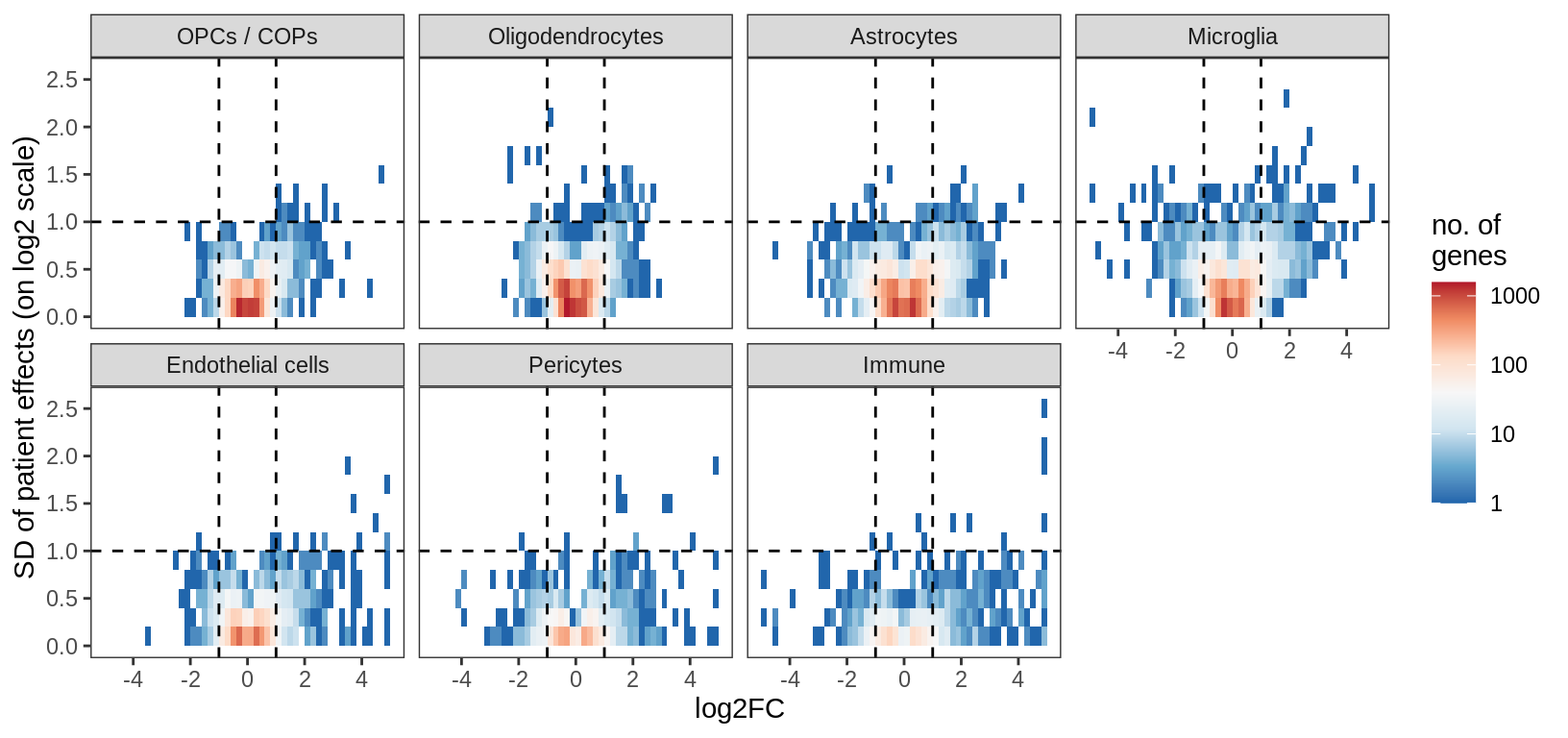
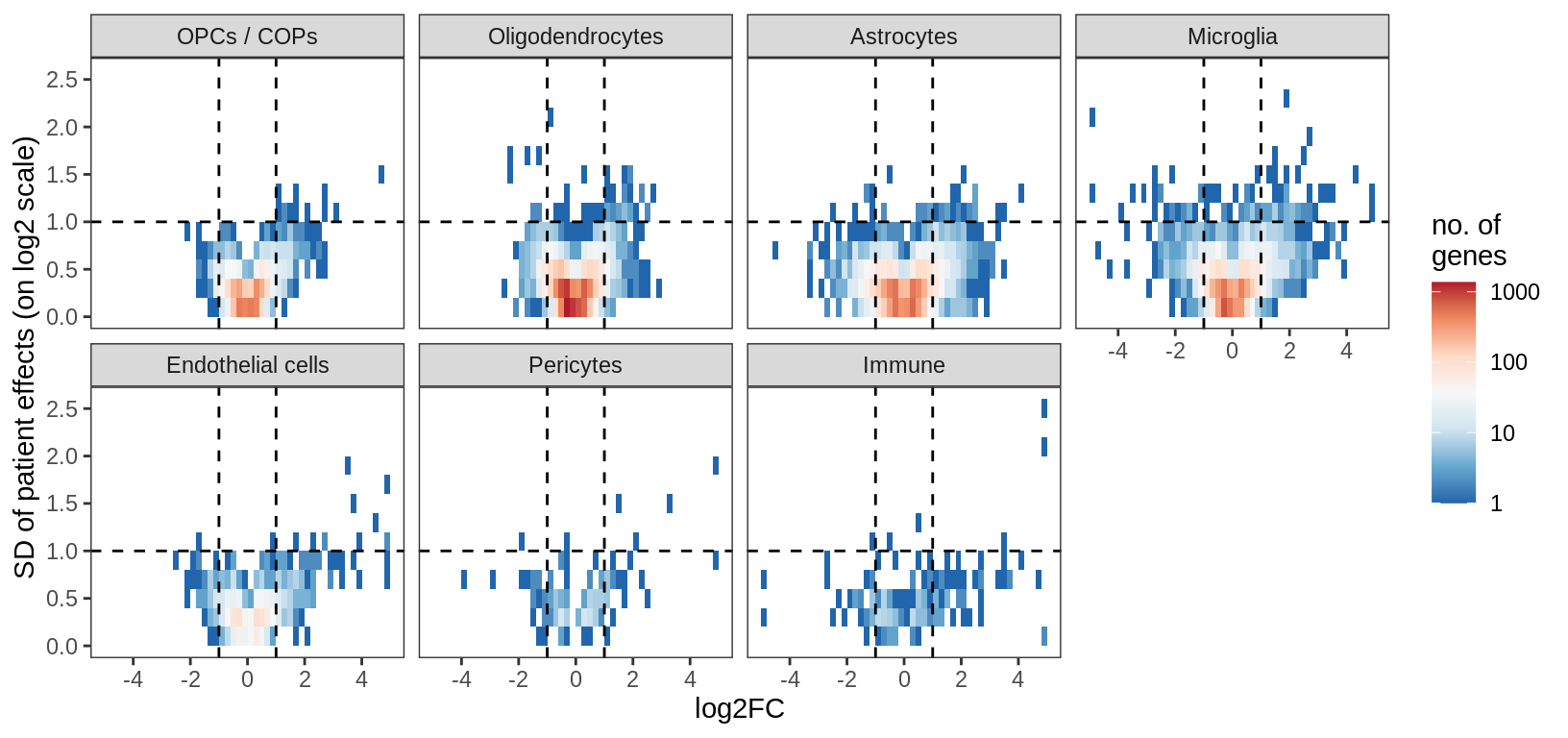
for (what in c('fc_vs_sd_all', 'fc_vs_sd_signif', 'ms_p_vs_lrt_p')) {
cat("### ", what, "\n")
print(plot_ms_vs_random(filter_dt, sel_cl, max_p, min_fc, min_sd, what = what))
cat("\n\n")
}fc_vs_sd_all
fc_vs_sd_signif
muscat results vs SD, MAGMA hits only
magma_hits = magma_dt[ p_magma_adj < 0.1 ]$gene_id
(plot_muscat_vs_sd_min(res_dt[ gene_id %in% magma_hits ], sd_dt,
sel_cl, min_sd, max_p))muscat results vs LoFs
(plot_muscat_and_sd_vs_lof(res_dt, sd_dt, lof_dt, sel_cl))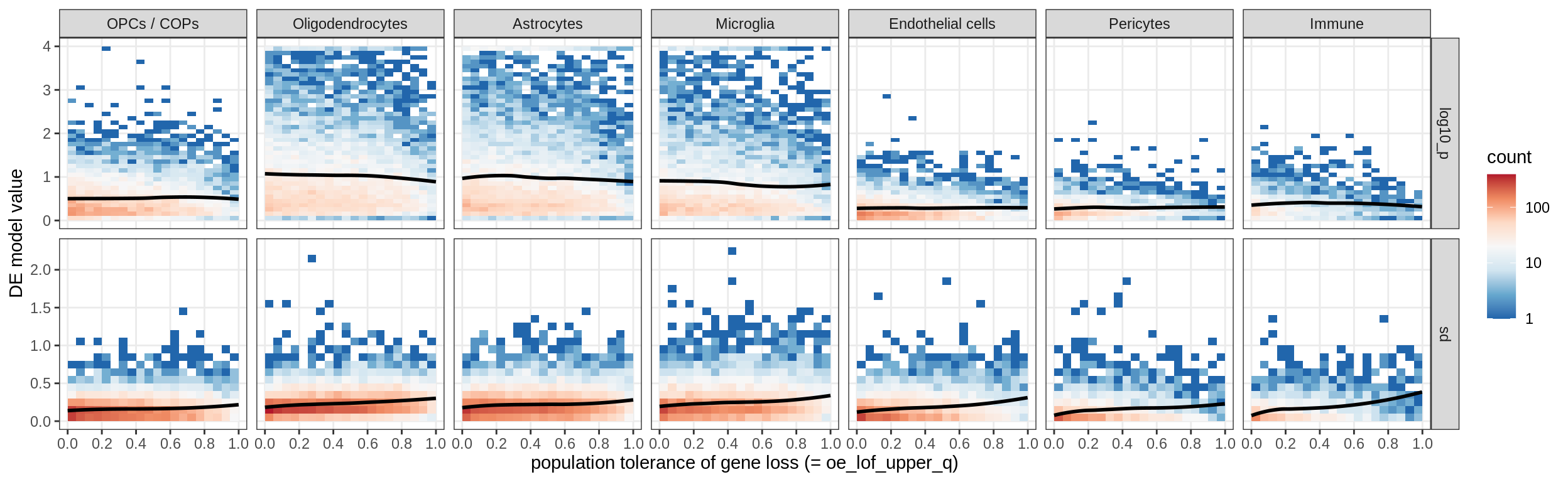
Expression heatmaps
Some notes:
- pca has both rows and columns ordered in a sensible data-driven way.
- clustered has the rows clustered by hierarchical clustering, and the columns the same as pca.
- three_per_patient is the same as clustered but only showing patients where we have >=3 samples.
- by_lesion has the rows ordered by lesion type, and the columns ordered by MS logFC (hopefully this shows the horseshoe a bit).
- FactorX has the rows ordered by each sample’s factor score, and the columns ordered by each gene’s factor weight; I also exclude genes with small weights for that factor.
- is_shared on top of the heatmap indicates whether a gene is unique to the celltype, or was also selected for another celltype.
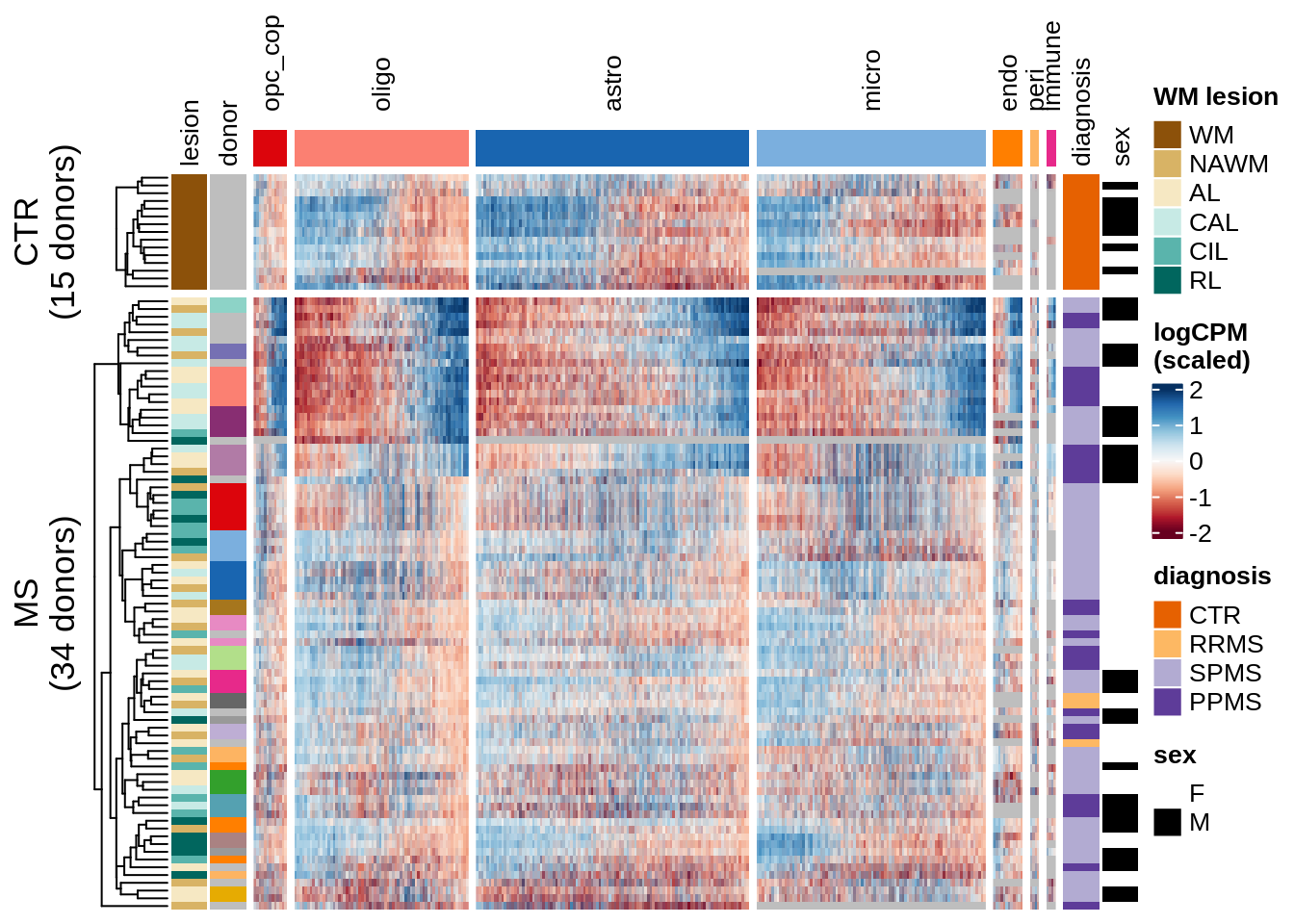
for (o in c("pca", "clustered", "three_per_patient", "by_lesion", factors_names(model))) {
cat("### ", o, "\n")
draw(plot_expression_heatmap_samples(pb, filtered_dt, annots_dt, sel_cl,
model, ordering = o)
, merge_legend = TRUE)
cat("\n\n")
}pca

clustered
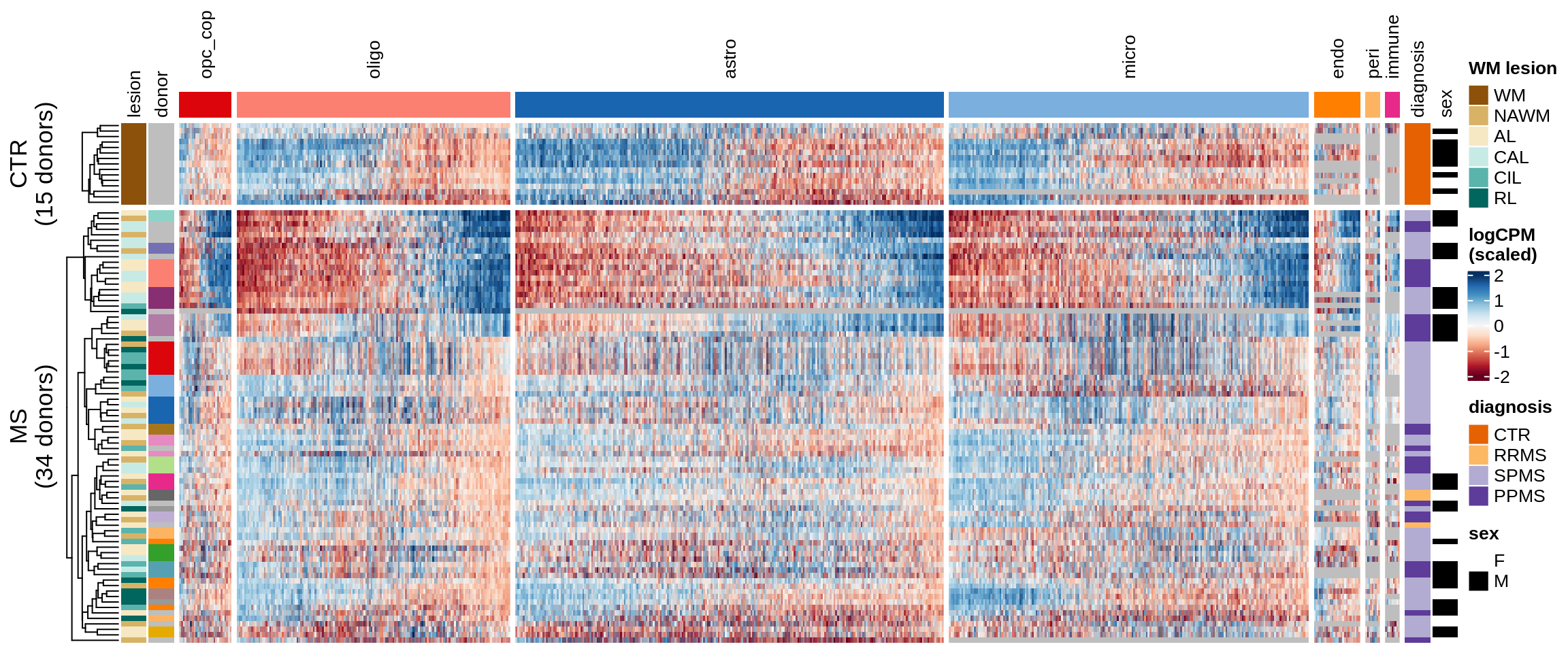
three_per_patient
by_lesion
Warning: Unknown levels in `f`: GM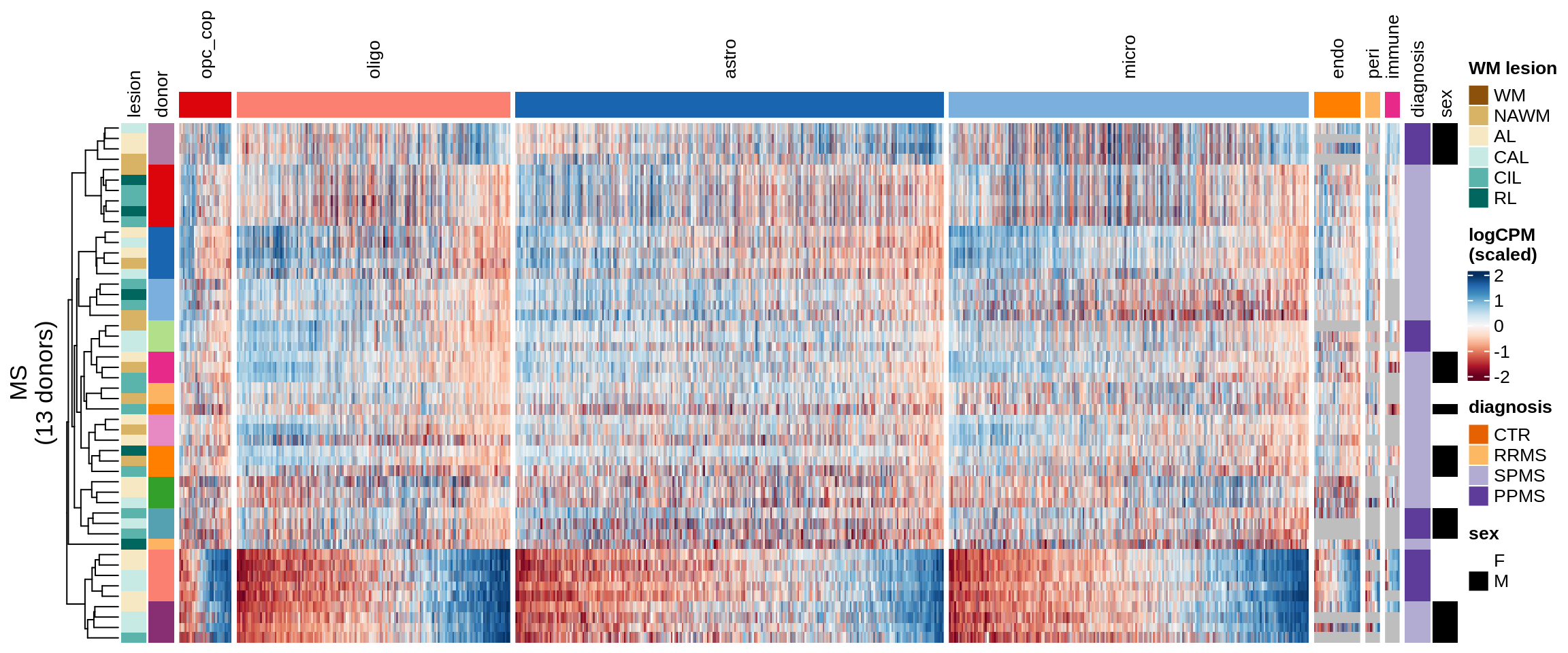

Factor1
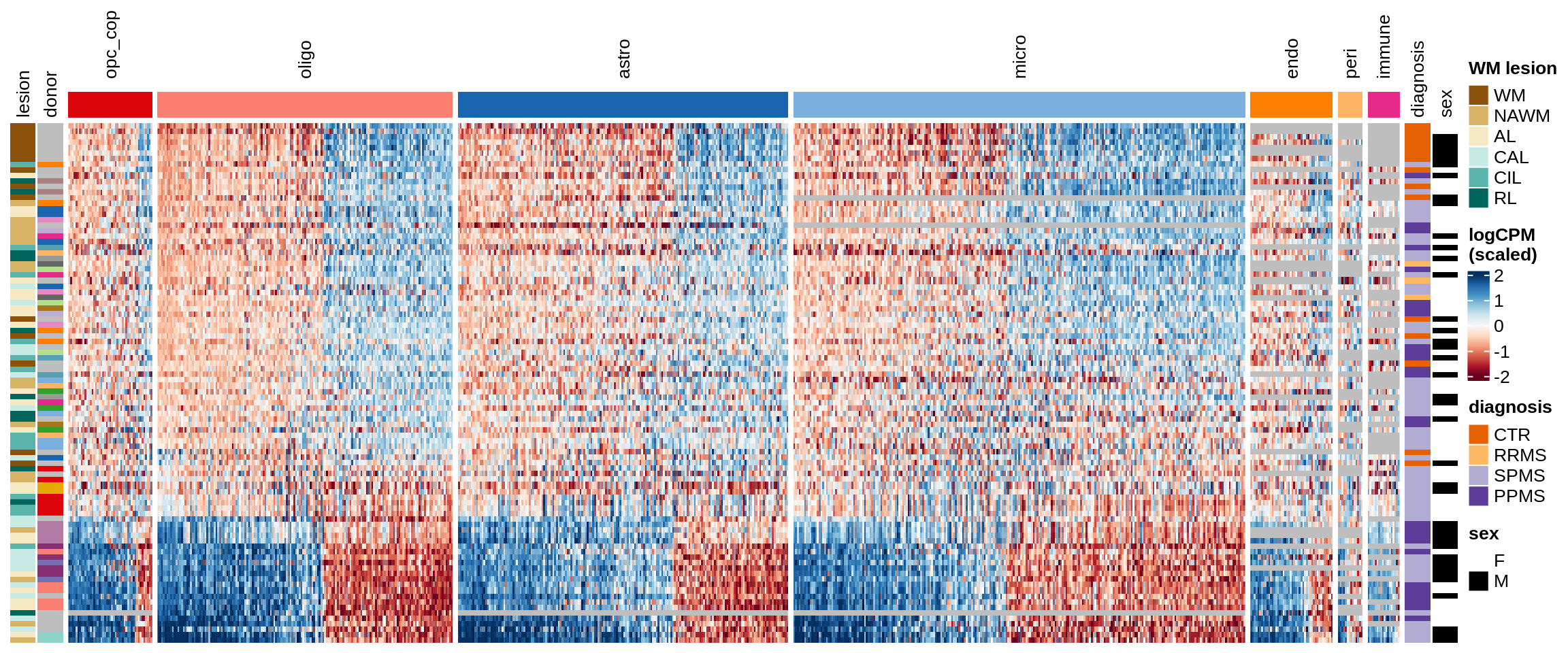
Factor2
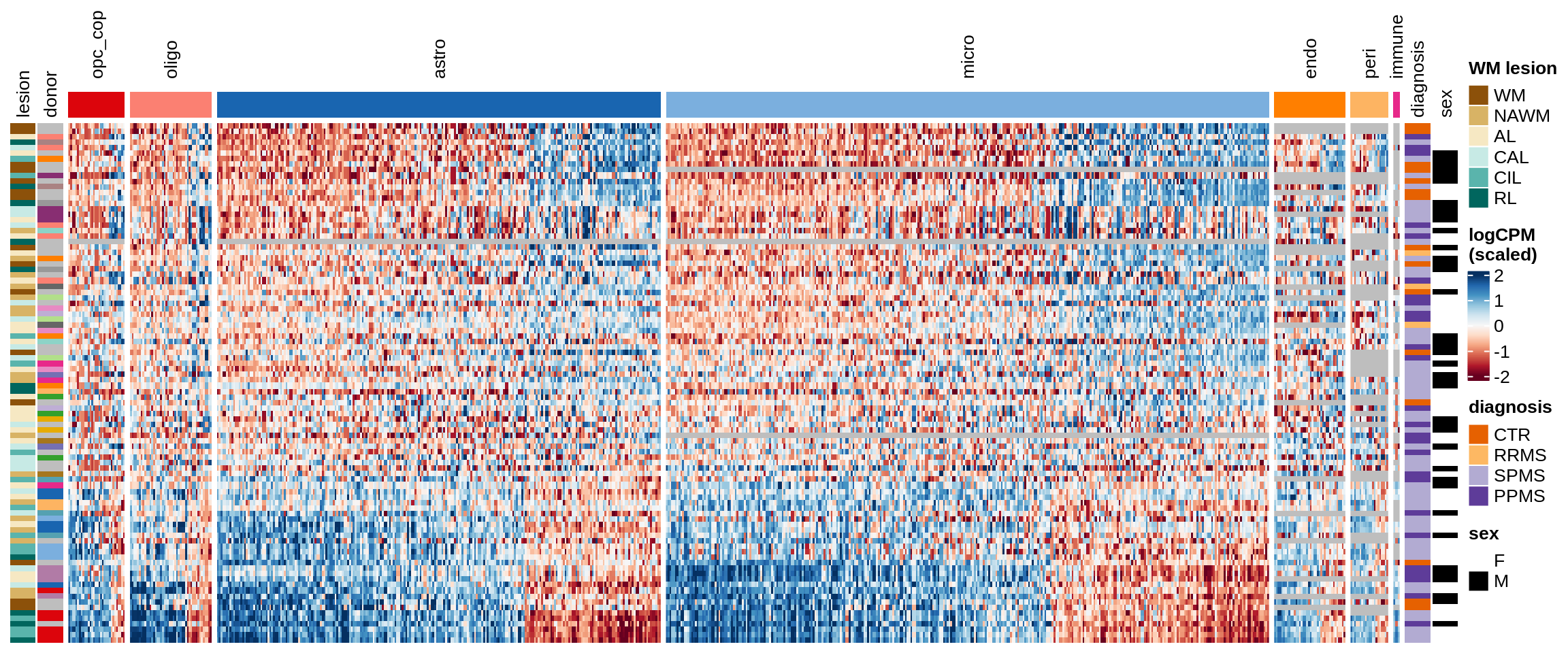
Factor3
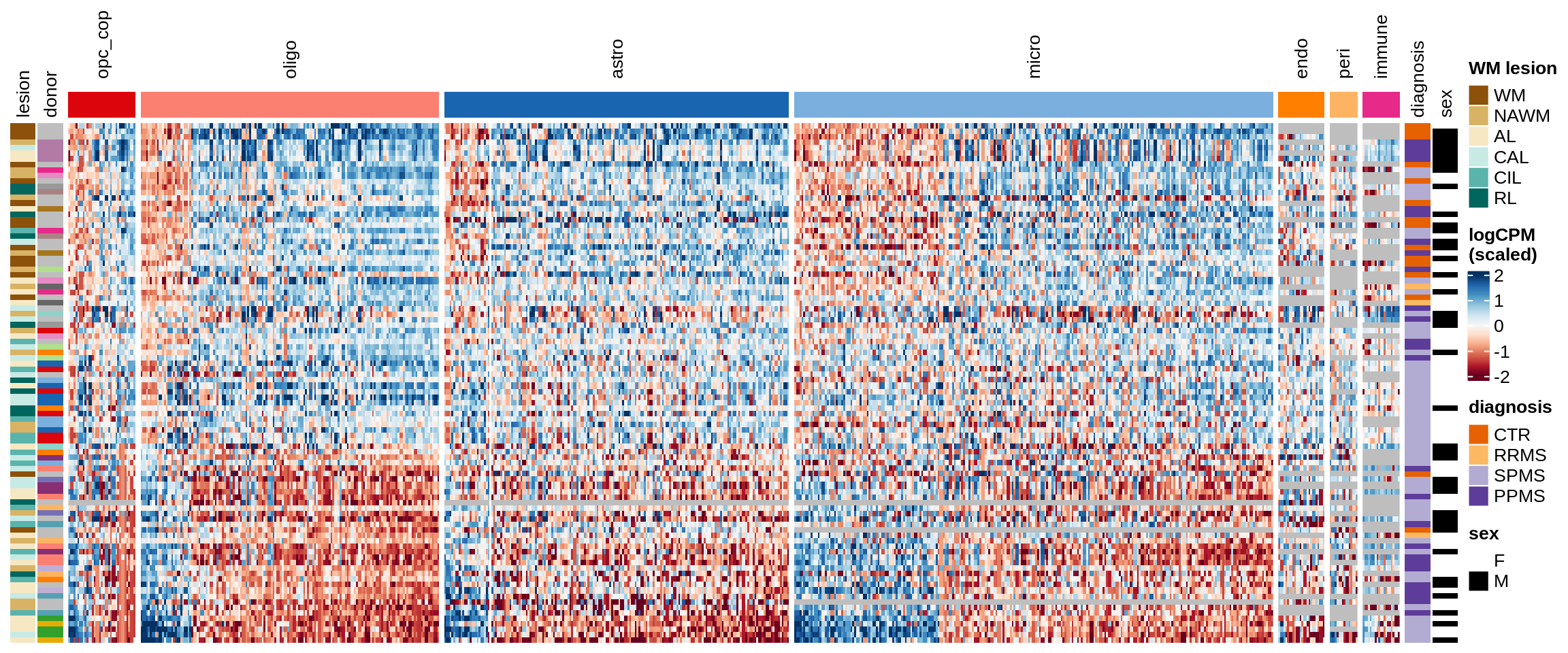
Factor4
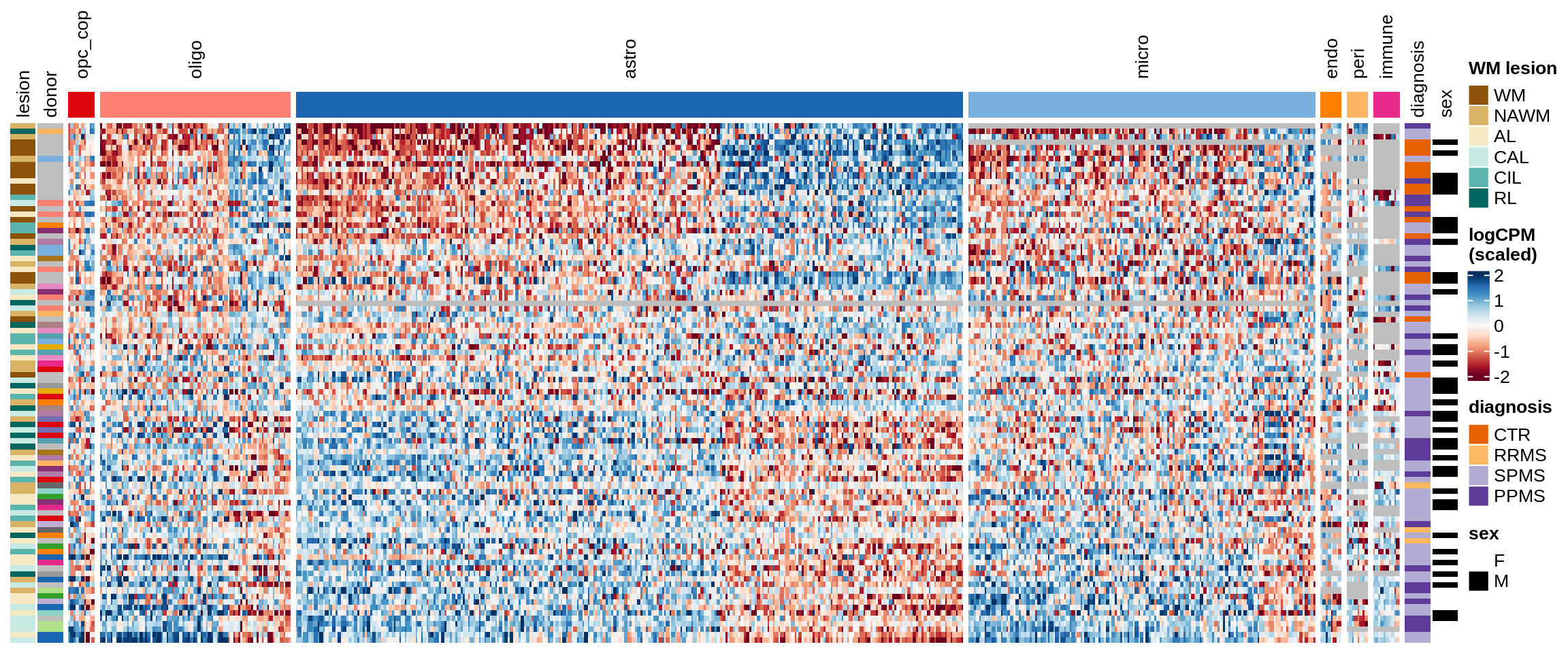
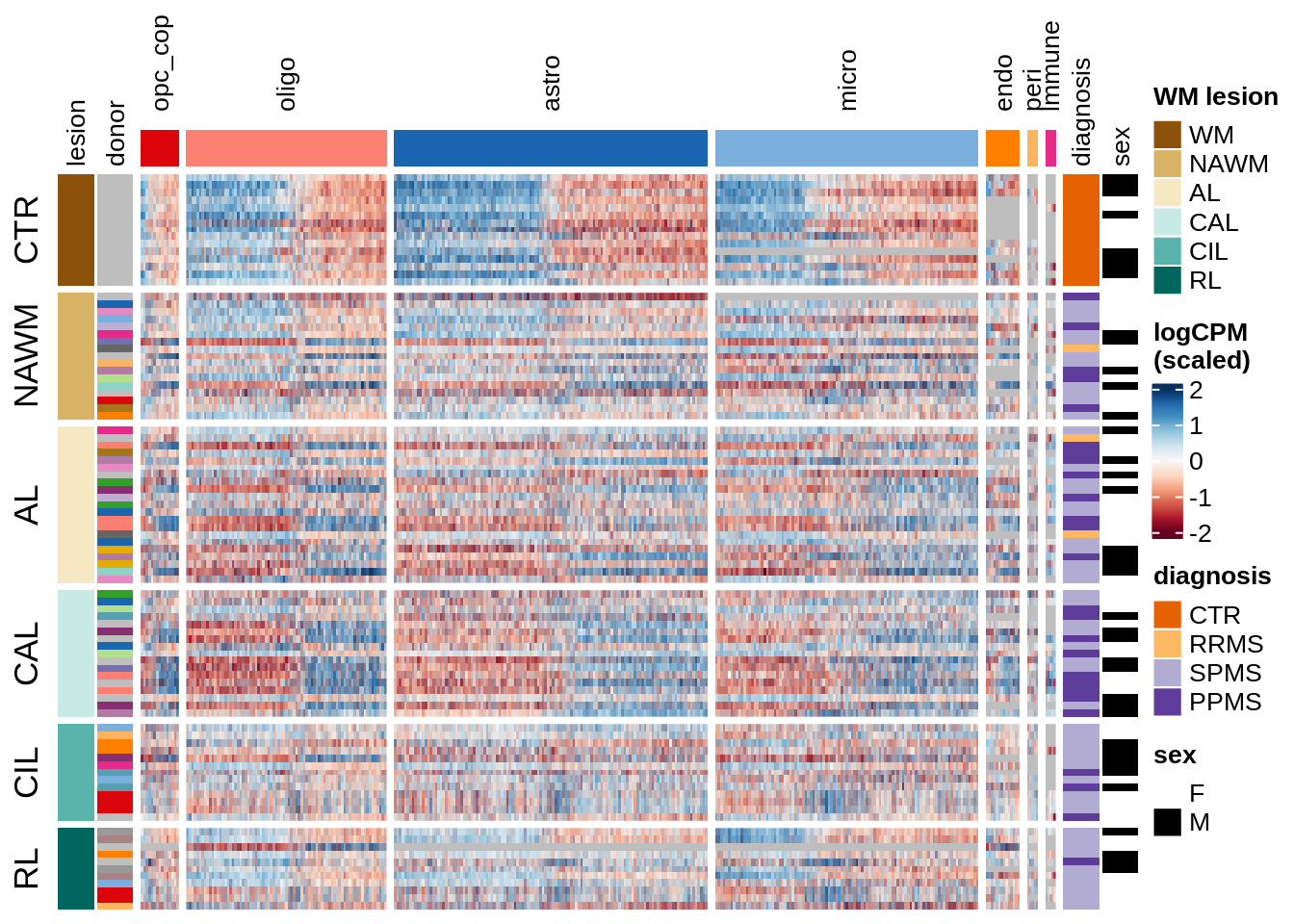
Expression heatmap, narrower
for (o in c("clustered", "by_lesion")) {
cat("### ", o, "\n")
draw(plot_expression_heatmap_samples(pb, filtered_dt, annots_dt, sel_cl,
model, ordering = o)
, merge_legend = TRUE)
cat("\n\n")
}clustered
MOFA+ factors - diagnosis
(plot_factors_univariate(model, annots_dt, pb, by = 'diagnosis'))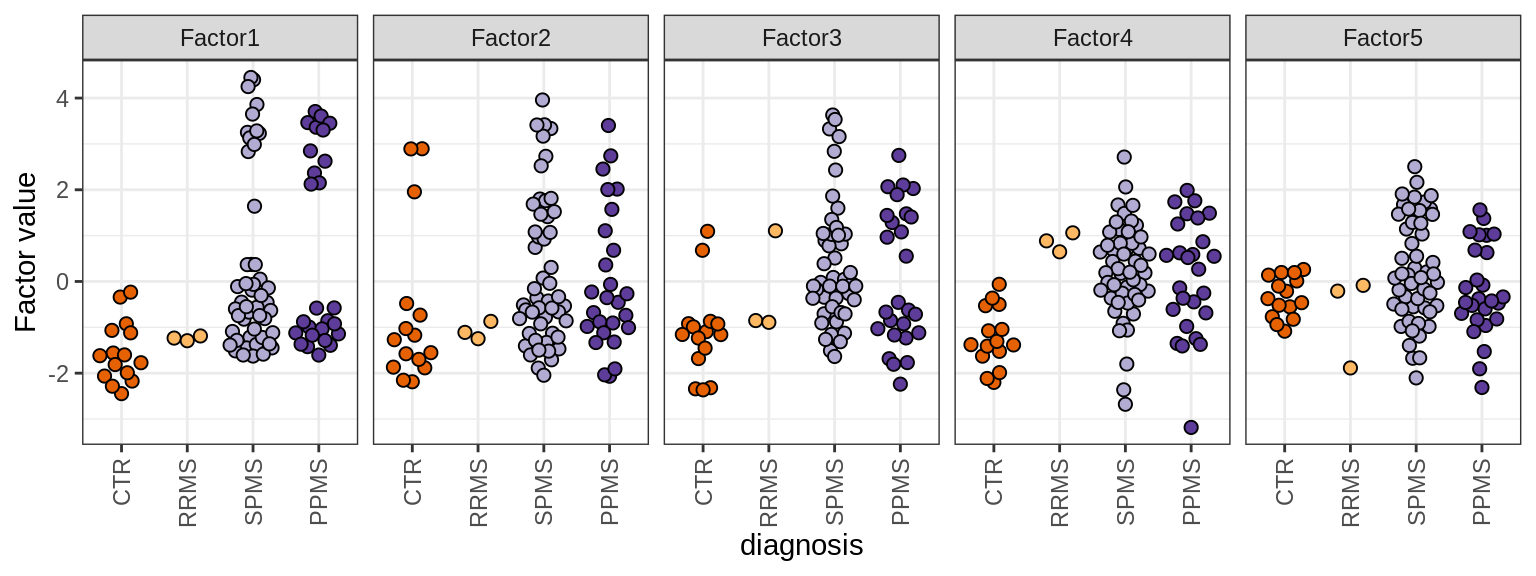
MOFA+ factors - lesions
(plot_factors_univariate(model, annots_dt, pb, by = 'lesion_type'))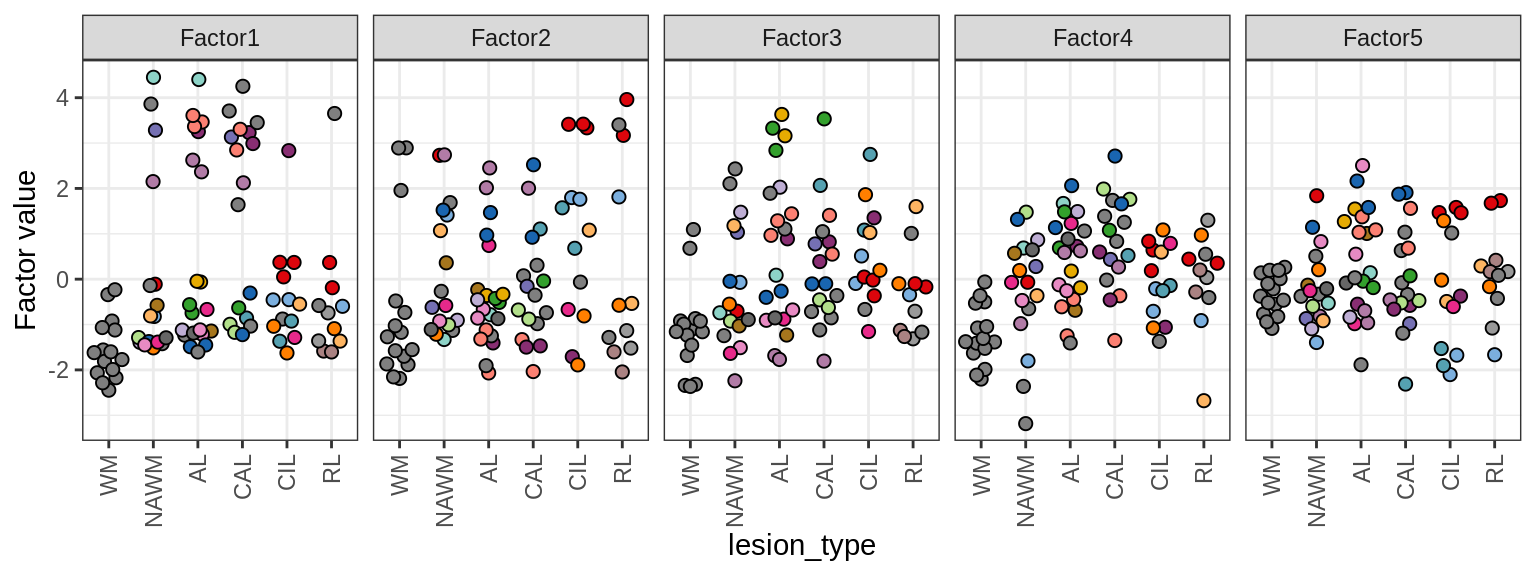
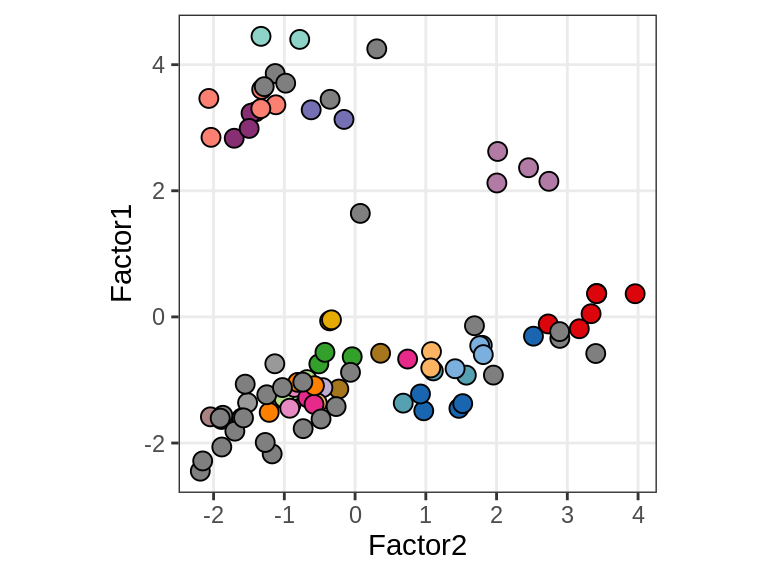
Factor 1 vs Factor 2
for (what in c("diagnosis", "lesion_type", "subject_id")) {
cat('### ', what, '\n', sep = '')
print(plot_factors_pair(model, annots_dt, pb,
f_pair = c("Factor2", "Factor1"), by = what))
cat('\n\n')
}diagnosis
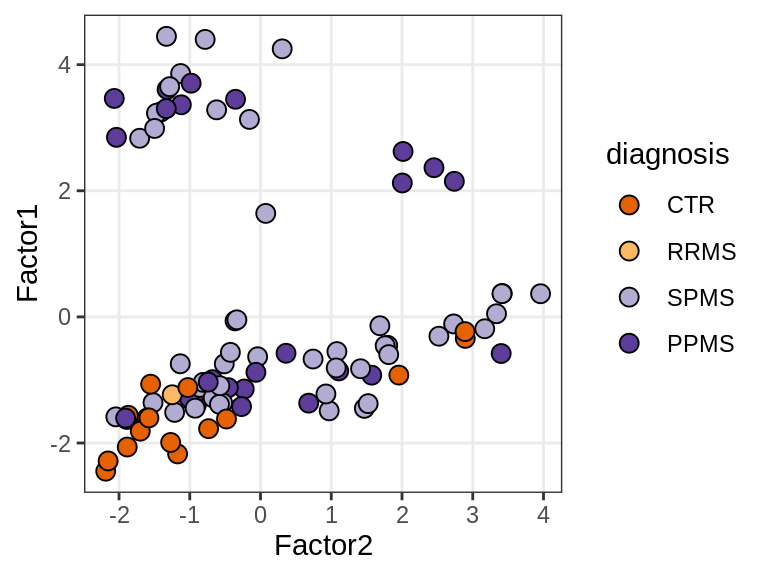
lesion_type

Interactions between factors and model components
(plot_factor_r2s(r2_dt, var_exp_dt))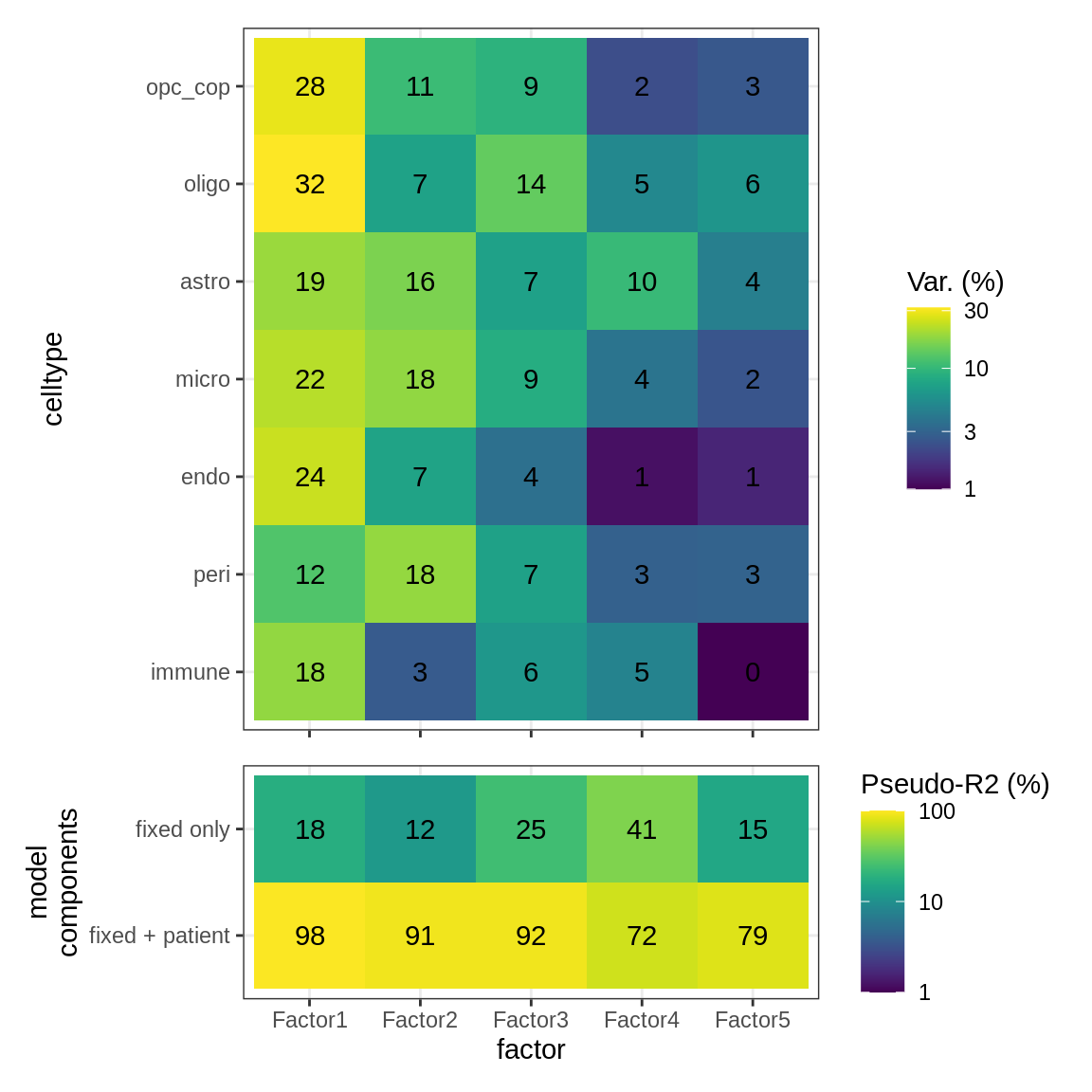
Does metadata explain factors?
(plot_factor_anovas(anova_dt))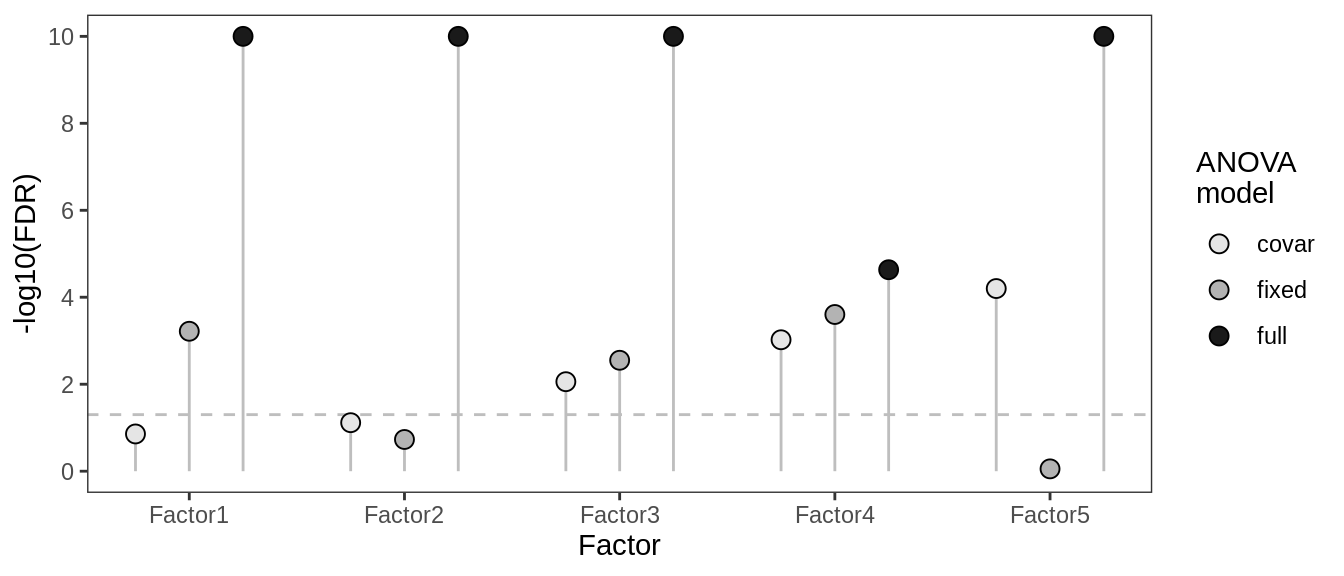
GO terms for factors
print(plot_mofa_gsea_dotplot(gsea_list[['go_bp']], labels_dt,
fgsea_cut = fgsea_cut, n_total = 50))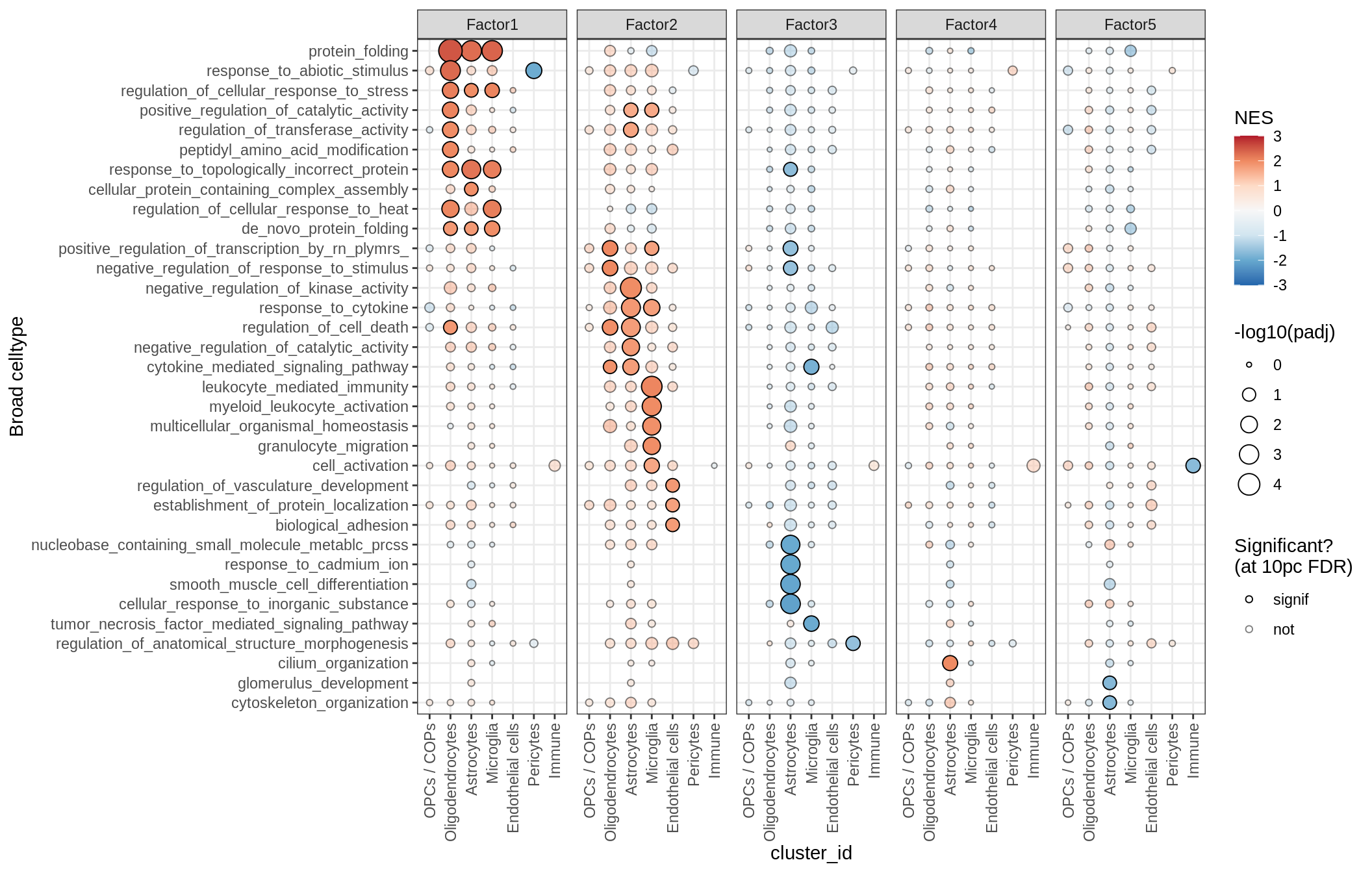
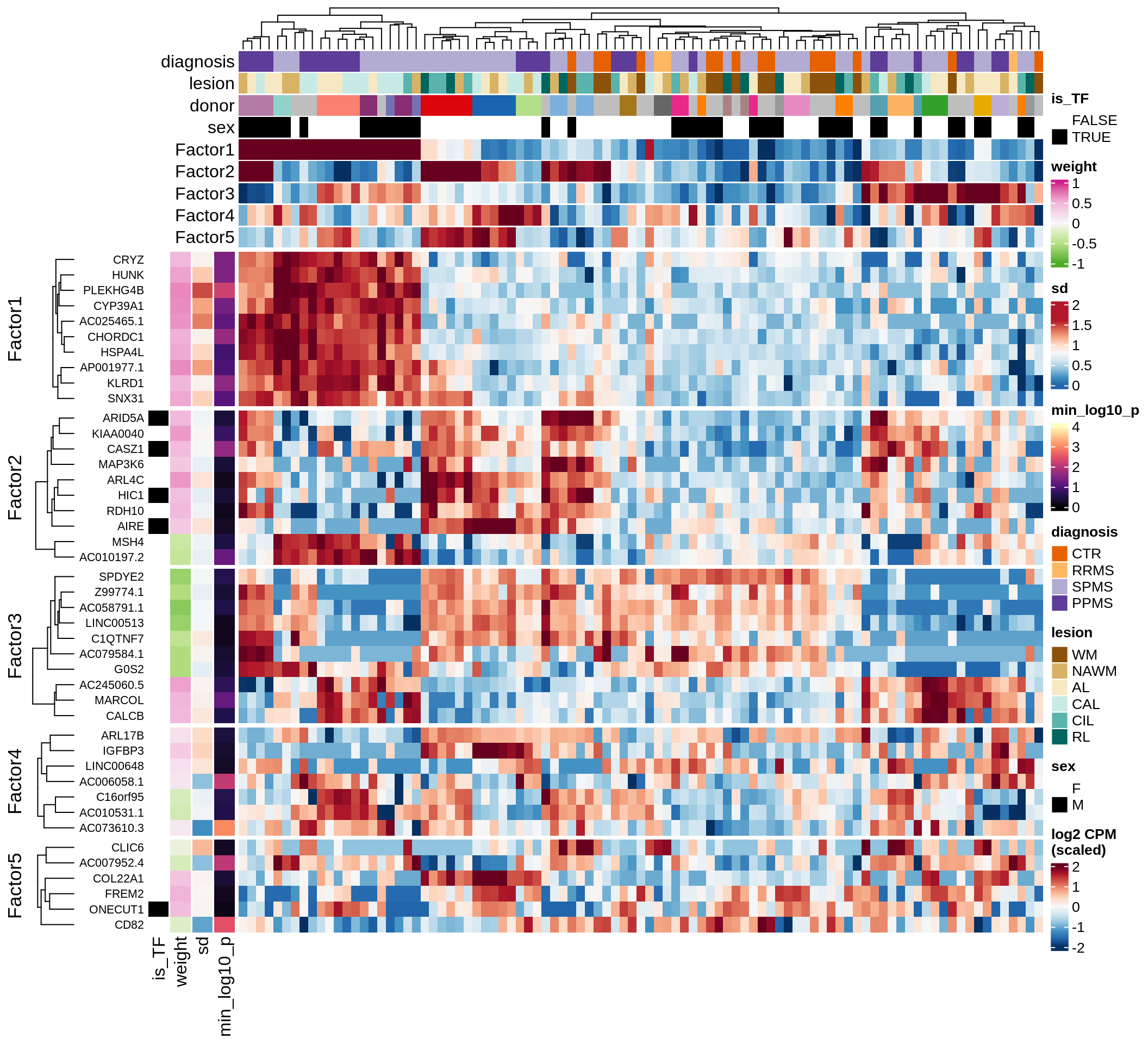
Top genes for Factor 1
draw( plot_top_genes_expression(model, pb, annots_dt,
filter_dt, tfs_dt, var_exp_no_endoperi,
sel_f = 'Factor1', min_var = 5, min_w = 0.2, n_top = 15, ok_genes = ok_genes),
heatmap_legend_side = 'left', annotation_legend_side = 'left')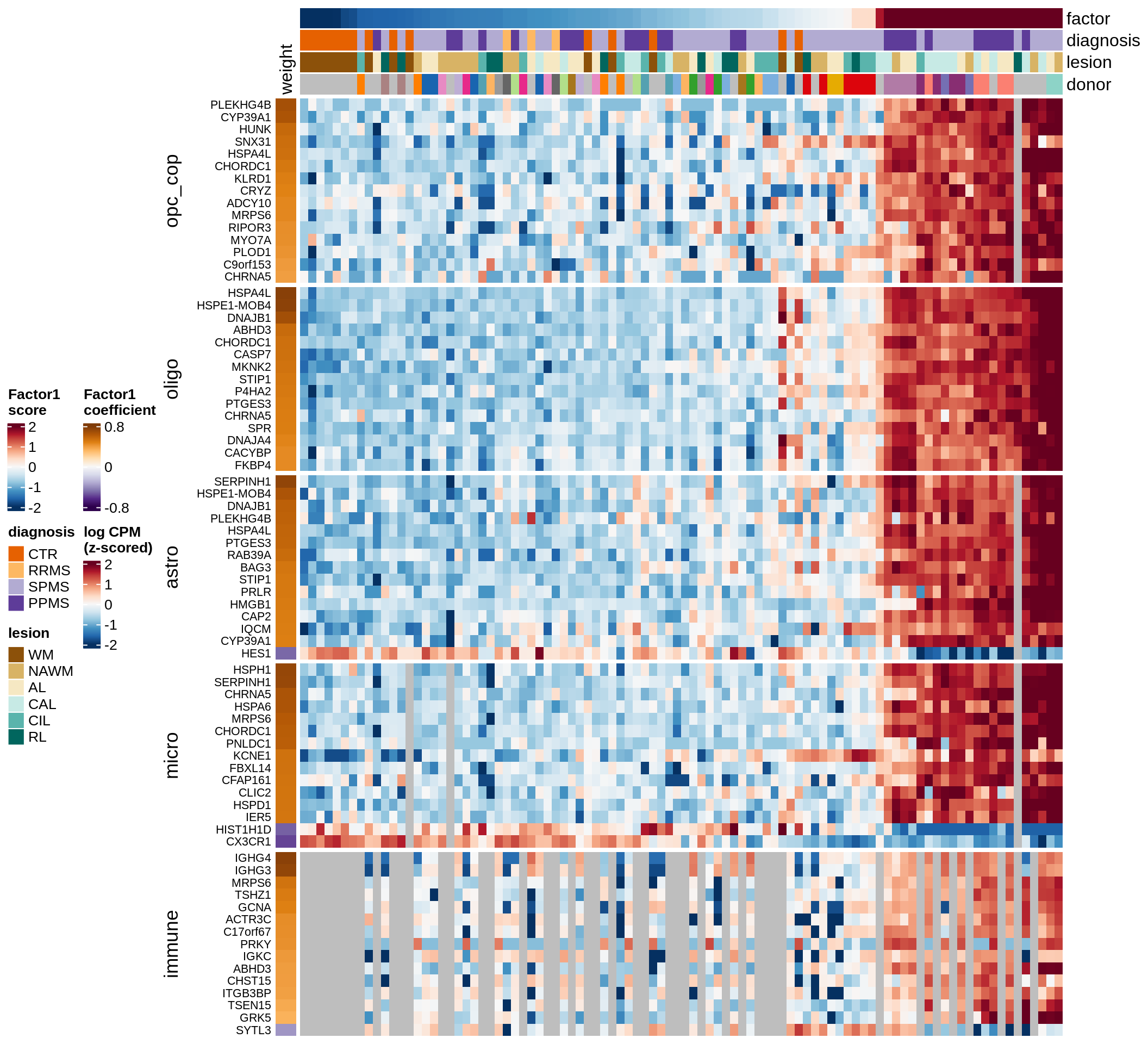
Top genes for Factor 2
draw( plot_top_genes_expression(model, pb, annots_dt,
filter_dt, tfs_dt, var_exp_no_endoperi,
sel_f = 'Factor2', min_var = 5, min_w = 0.2, n_top = 15, ok_genes = ok_genes),
heatmap_legend_side = 'left', annotation_legend_side = 'left')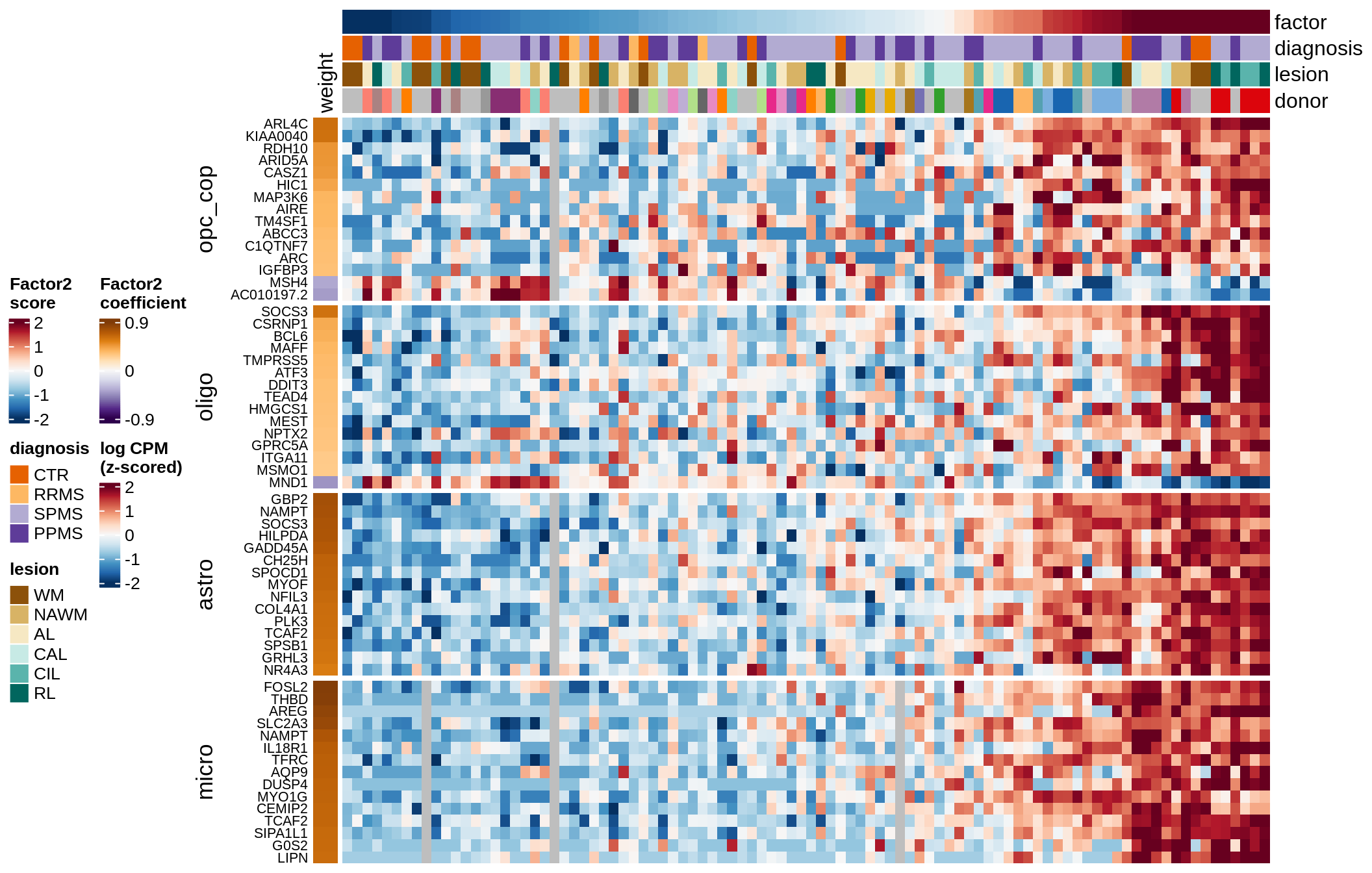
Top genes for Factor 3
draw( plot_top_genes_expression(model, pb, annots_dt,
filter_dt, tfs_dt, var_exp_no_endoperi,
sel_f = 'Factor3', min_var = 5, min_w = 0.2, n_top = 15, ok_genes = ok_genes),
heatmap_legend_side = 'left', annotation_legend_side = 'left')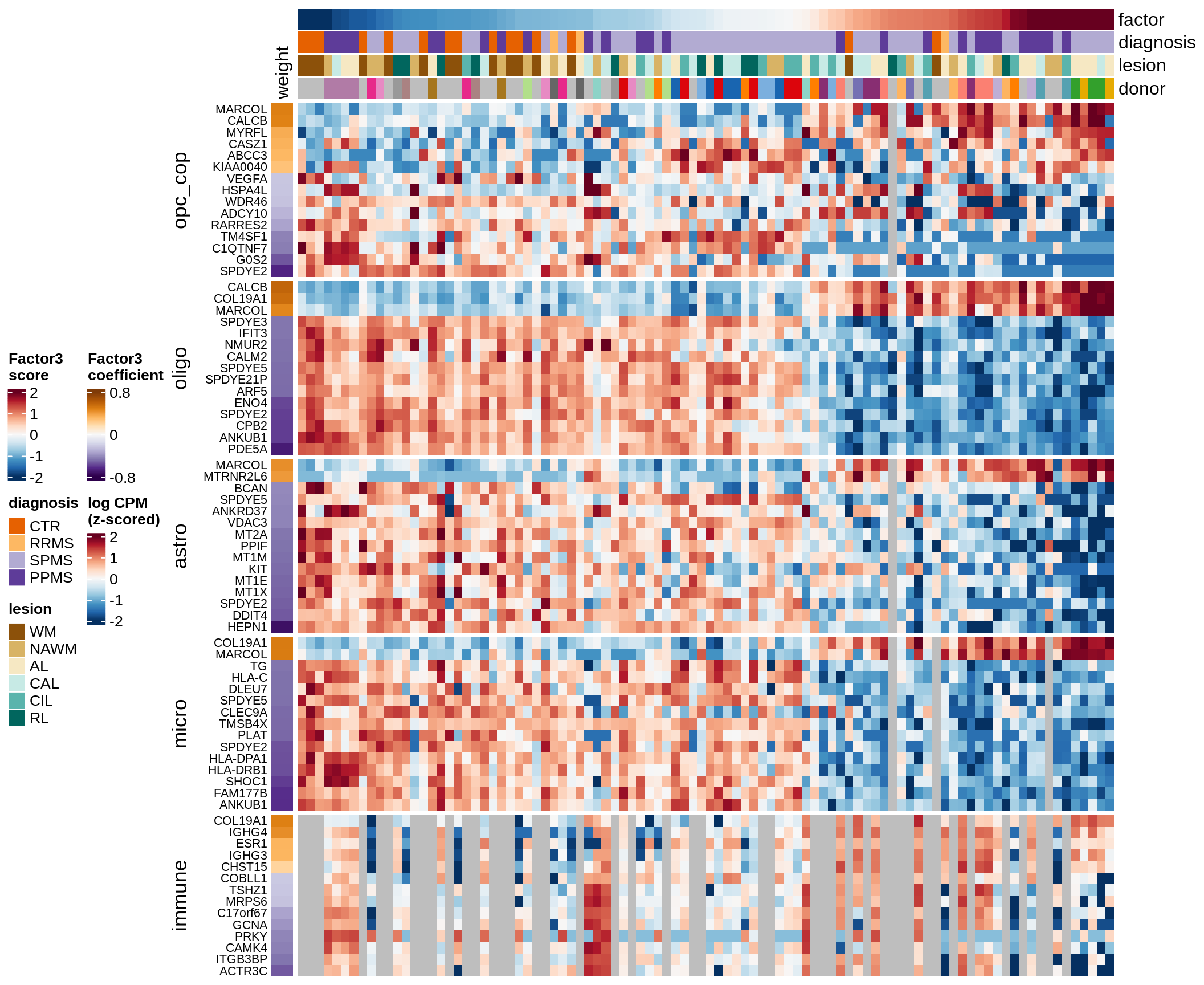
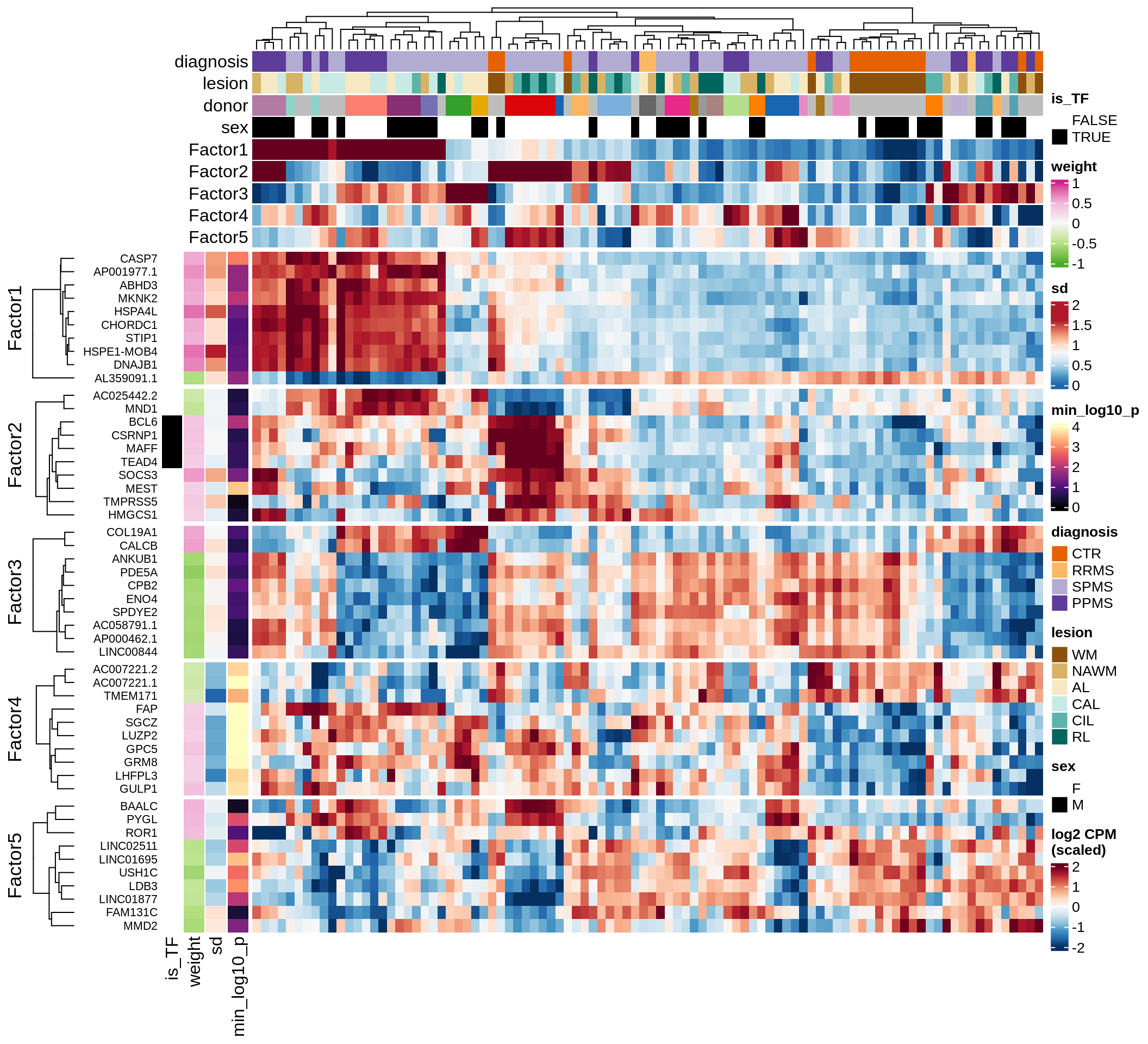
Top genes for Factor 4
draw( plot_top_genes_expression(model, pb, annots_dt,
filter_dt, tfs_dt, var_exp_no_endoperi,
sel_f = 'Factor4', min_var = 5, min_w = 0.2, n_top = 15, ok_genes = ok_genes),
heatmap_legend_side = 'left', annotation_legend_side = 'left')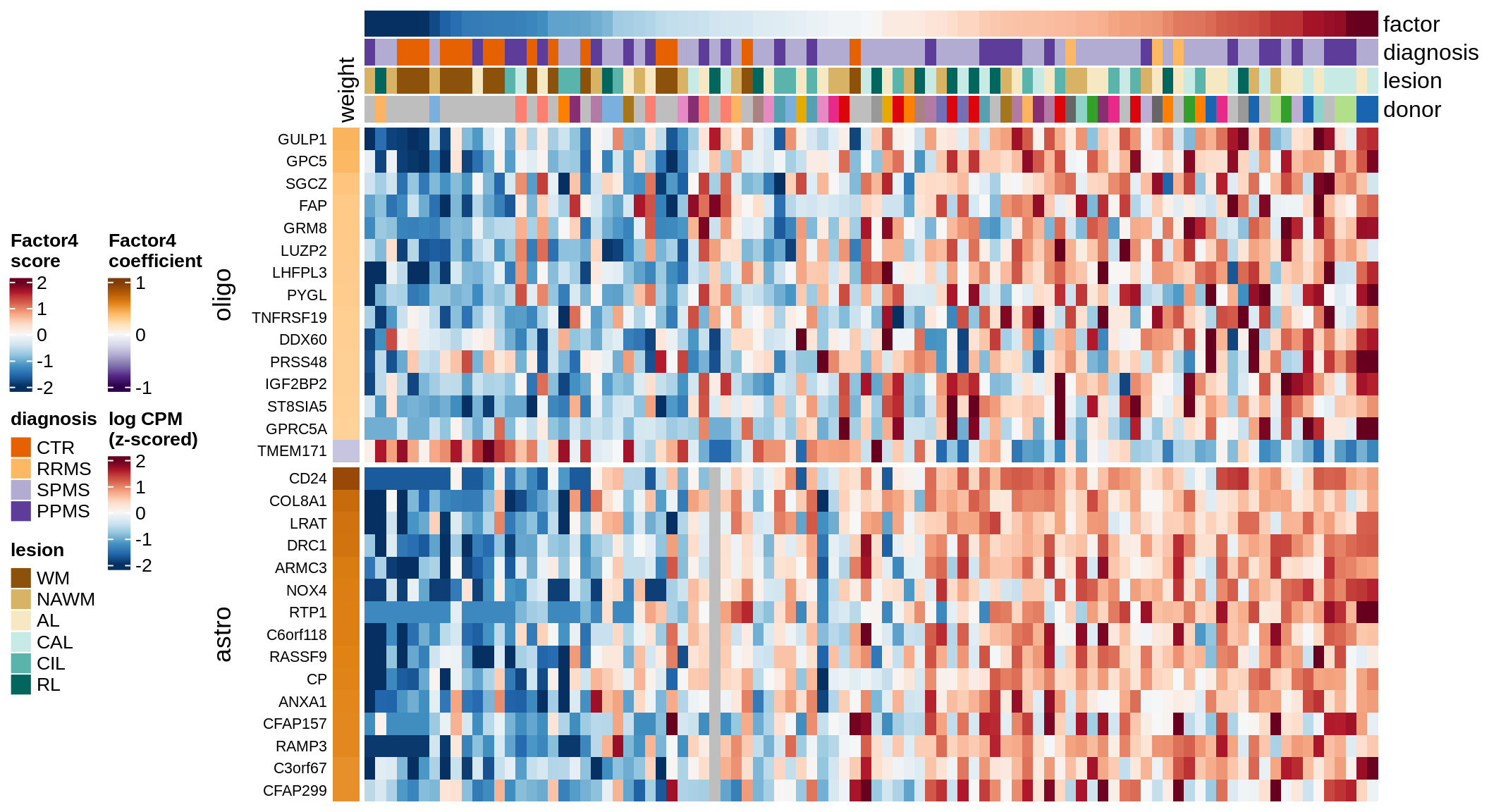
Top genes for Factor 5
draw( plot_top_genes_expression(model, pb, annots_dt,
filter_dt, tfs_dt, var_exp_no_endoperi,
sel_f = 'Factor5', min_var = 5, min_w = 0.2, n_top = 15, ok_genes = ok_genes),
heatmap_legend_side = 'left', annotation_legend_side = 'left')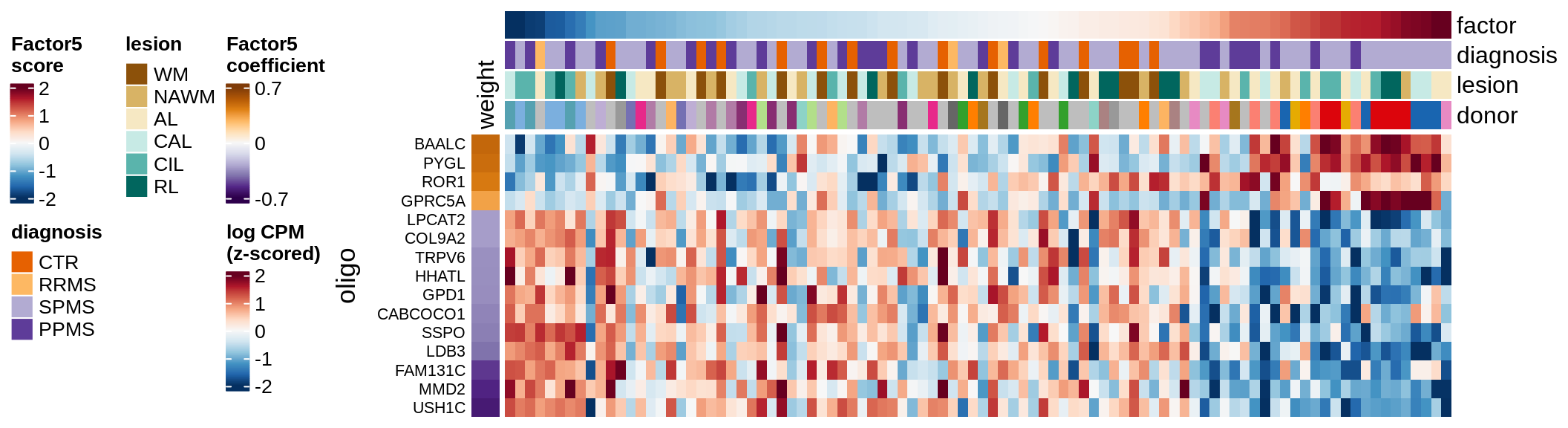
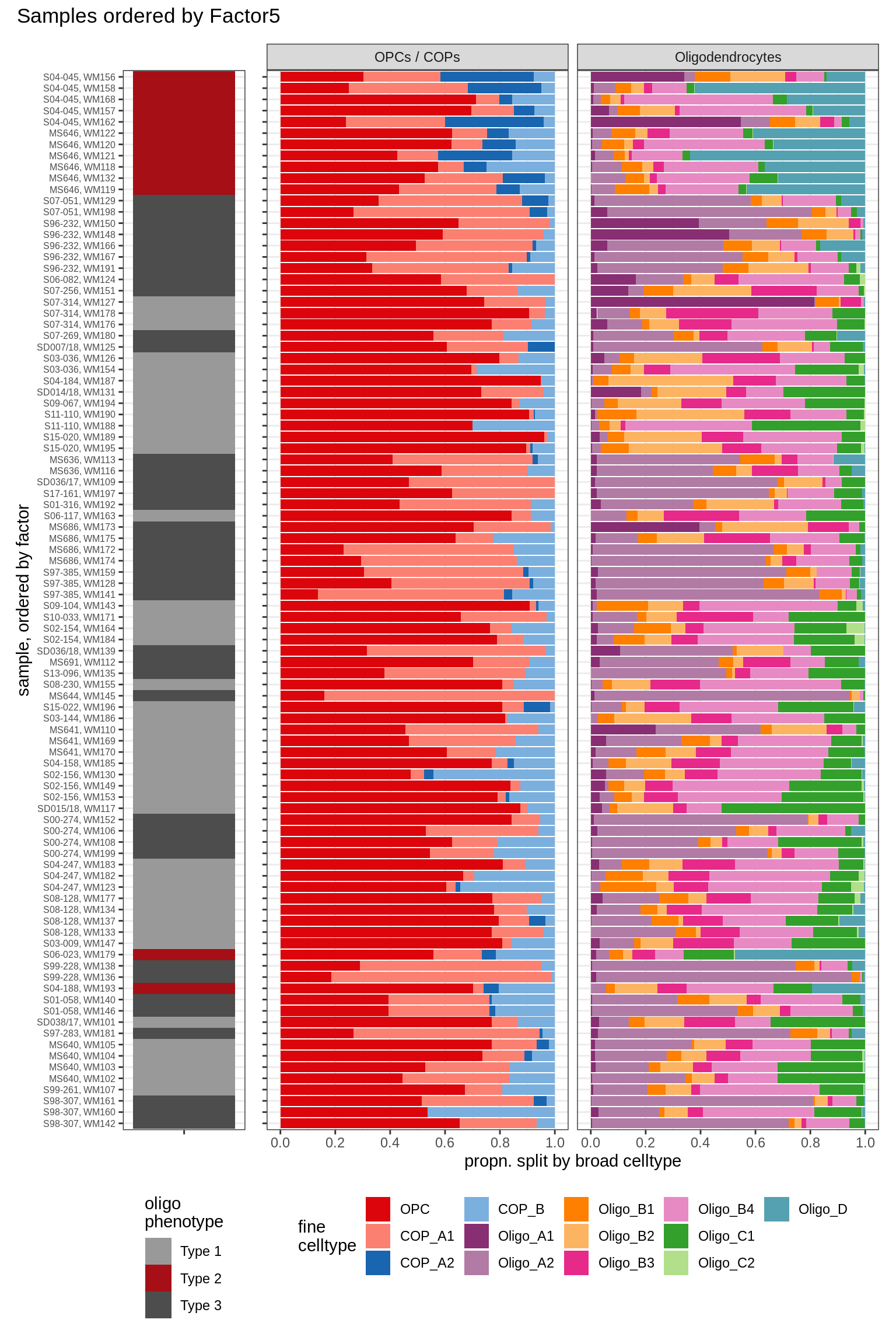
Barplots of celltype proportions ordered by factors
conos_dt = load_labelled_dt(labelled_f, labels_f)
types = c('OPCs / COPs', 'Oligodendrocytes')
m = "WM"
oligos_dt = conos_dt[ type_broad %in% types & str_detect(sample_id, "WM") ] %>%
.[, N_sample := .N, by = sample_id] %>%
.[, .N, by = .(sample_id, N_sample, type_broad, type_fine)] %>%
.[, prop := N / sum(N), by = .(sample_id, type_broad)] %>%
.[, type_fine := fct_relevel(type_fine, 'OPC')]
for (sel_f in factors_names(model)) {
cat('### ', sel_f, '\n', sep = '')
print(plot_barplots_ordered_by_factors(oligos_dt, model, sel_f))
cat('\n\n')
}Factor1

Factor2

Factor3
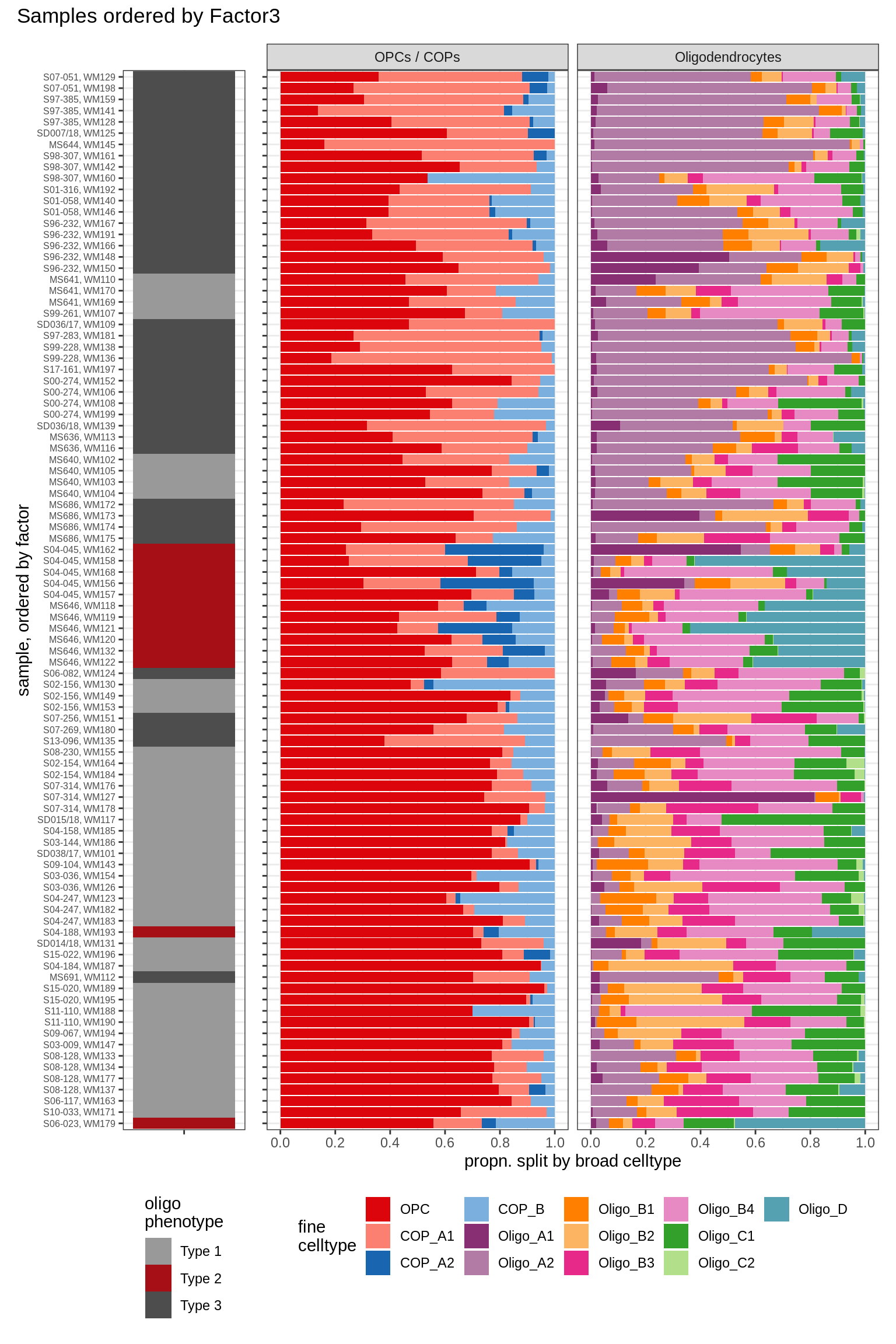
Factor4

Factor5
devtools::session_info()- Session info ---------------------------------------------------------------
setting value
version R version 4.1.2 (2021-11-01)
os Red Hat Enterprise Linux 8.2 (Ootpa)
system x86_64, linux-gnu
ui X11
language (EN)
collate en_US.UTF-8
ctype C
tz Europe/Amsterdam
date 2022-03-24
pandoc 2.5 @ /apps/rocs/pRED/2020.08/cascadelake/software/Pandoc/2.5/bin/ (via rmarkdown)
- Packages -------------------------------------------------------------------
package * version date (UTC) lib source
abind 1.4-5 2016-07-21 [5] CRAN (R 4.1.2)
ade4 1.7-18 2021-09-16 [5] CRAN (R 4.1.2)
ANCOMBC * 1.4.0 2021-10-26 [3] Bioconductor
annotate 1.72.0 2021-10-26 [3] Bioconductor
AnnotationDbi 1.56.2 2021-11-09 [3] Bioconductor
ape 5.5 2021-04-25 [5] CRAN (R 4.1.2)
assertthat * 0.2.1 2019-03-21 [5] CRAN (R 4.1.2)
backports 1.4.0 2021-11-23 [5] CRAN (R 4.1.2)
basilisk 1.6.0 2021-10-26 [1] Bioconductor
basilisk.utils 1.6.0 2021-10-26 [1] Bioconductor
beachmat 2.10.0 2021-10-26 [3] Bioconductor
beeswarm 0.4.0 2021-06-01 [3] CRAN (R 4.1.2)
Biobase * 2.54.0 2021-10-26 [3] Bioconductor
BiocGenerics * 0.40.0 2021-10-26 [3] Bioconductor
BiocIO 1.4.0 2021-10-26 [3] Bioconductor
BiocManager 1.30.16 2021-06-15 [3] CRAN (R 4.1.2)
BiocNeighbors 1.12.0 2021-10-26 [3] Bioconductor
BiocParallel * 1.28.3 2021-12-09 [1] Bioconductor
BiocSingular 1.10.0 2021-10-26 [3] Bioconductor
BiocStyle * 2.22.0 2021-10-26 [3] Bioconductor
biomformat 1.22.0 2021-10-26 [3] Bioconductor
Biostrings 2.62.0 2021-10-26 [3] Bioconductor
bit 4.0.4 2020-08-04 [5] CRAN (R 4.1.2)
bit64 4.0.5 2020-08-30 [5] CRAN (R 4.1.2)
bitops 1.0-7 2021-04-24 [5] CRAN (R 4.1.2)
blme 1.0-5 2021-01-05 [3] CRAN (R 4.1.2)
blob 1.2.2 2021-07-23 [5] CRAN (R 4.1.2)
boot 1.3-28 2021-05-03 [5] CRAN (R 4.1.2)
brew 1.0-6 2011-04-13 [5] CRAN (R 4.1.2)
broom 0.7.10 2021-10-31 [5] CRAN (R 4.1.2)
bslib 0.3.1 2021-10-06 [5] CRAN (R 4.1.2)
cachem 1.0.6 2021-08-19 [5] CRAN (R 4.1.2)
callr 3.7.0 2021-04-20 [5] CRAN (R 4.1.2)
caTools 1.18.2 2021-03-28 [5] CRAN (R 4.1.2)
cellranger 1.1.0 2016-07-27 [5] CRAN (R 4.1.2)
circlize * 0.4.13 2021-06-09 [3] CRAN (R 4.1.2)
cli 3.2.0 2022-02-14 [1] CRAN (R 4.1.2)
clue 0.3-60 2021-10-11 [5] CRAN (R 4.1.2)
cluster 2.1.2 2021-04-17 [5] CRAN (R 4.1.2)
coda 0.19-4 2020-09-30 [5] CRAN (R 4.1.2)
codetools 0.2-18 2020-11-04 [5] CRAN (R 4.1.2)
colorspace 2.0-3 2022-02-21 [1] CRAN (R 4.1.2)
ComplexHeatmap * 2.10.0 2021-10-26 [3] Bioconductor
corrplot 0.92 2021-11-18 [3] CRAN (R 4.1.2)
cowplot 1.1.1 2020-12-30 [5] CRAN (R 4.1.2)
crayon 1.5.0 2022-02-14 [1] CRAN (R 4.1.2)
data.table * 1.14.2 2021-09-27 [5] CRAN (R 4.1.2)
DBI 1.1.1 2021-01-15 [5] CRAN (R 4.1.2)
dbplyr 2.1.1 2021-04-06 [5] CRAN (R 4.1.2)
DelayedArray 0.20.0 2021-10-26 [3] Bioconductor
DelayedMatrixStats 1.16.0 2021-10-26 [3] Bioconductor
deldir 1.0-6 2021-10-23 [5] CRAN (R 4.1.2)
dendsort 0.3.4 2021-04-20 [1] CRAN (R 4.1.2)
desc 1.4.0 2021-09-28 [5] CRAN (R 4.1.2)
DESeq2 1.34.0 2021-10-26 [3] Bioconductor
devtools 2.4.3 2021-11-30 [5] CRAN (R 4.1.2)
digest 0.6.29 2021-12-01 [5] CRAN (R 4.1.2)
dir.expiry 1.2.0 2021-10-26 [1] Bioconductor
doParallel 1.0.16 2020-10-16 [5] CRAN (R 4.1.2)
dplyr * 1.0.7 2021-06-18 [5] CRAN (R 4.1.2)
drat 0.2.2 2021-12-02 [1] CRAN (R 4.1.2)
edgeR * 3.36.0 2021-10-26 [3] Bioconductor
ellipsis 0.3.2 2021-04-29 [5] CRAN (R 4.1.2)
emmeans 1.7.1-1 2021-11-29 [3] CRAN (R 4.1.2)
estimability 1.3 2018-02-11 [3] CRAN (R 4.1.2)
evaluate 0.15 2022-02-18 [1] CRAN (R 4.1.2)
fansi 1.0.2 2022-01-14 [1] CRAN (R 4.1.2)
farver 2.1.0 2021-02-28 [5] CRAN (R 4.1.2)
fastmap 1.1.0 2021-01-25 [5] CRAN (R 4.1.2)
fastmatch 1.1-3 2021-07-23 [3] CRAN (R 4.1.2)
fgsea * 1.20.0 2021-10-26 [3] Bioconductor
filelock 1.0.2 2018-10-05 [3] CRAN (R 4.1.2)
fitdistrplus 1.1-6 2021-09-28 [5] CRAN (R 4.1.2)
forcats * 0.5.1 2021-01-27 [5] CRAN (R 4.1.2)
foreach 1.5.1 2020-10-15 [5] CRAN (R 4.1.2)
fs 1.5.1 2021-11-30 [5] CRAN (R 4.1.2)
future * 1.23.0 2021-10-31 [5] CRAN (R 4.1.2)
future.apply 1.8.1 2021-08-10 [5] CRAN (R 4.1.2)
genefilter 1.76.0 2021-10-26 [3] Bioconductor
geneplotter 1.72.0 2021-10-26 [3] Bioconductor
generics 0.1.1 2021-10-25 [5] CRAN (R 4.1.2)
GenomeInfoDb * 1.30.1 2022-01-30 [1] Bioconductor
GenomeInfoDbData 1.2.7 2022-03-15 [3] Bioconductor
GenomicAlignments 1.30.0 2021-10-26 [3] Bioconductor
GenomicRanges * 1.46.1 2021-11-18 [3] Bioconductor
GetoptLong 1.0.5 2020-12-15 [3] CRAN (R 4.1.2)
ggbeeswarm * 0.6.0 2017-08-07 [3] CRAN (R 4.1.2)
ggplot.multistats * 1.0.0 2019-10-28 [1] CRAN (R 4.1.2)
ggplot2 * 3.3.5 2021-06-25 [5] CRAN (R 4.1.2)
ggrepel * 0.9.1 2021-01-15 [5] CRAN (R 4.1.2)
ggridges 0.5.3 2021-01-08 [5] CRAN (R 4.1.2)
git2r 0.29.0 2021-11-22 [5] CRAN (R 4.1.2)
glmmTMB 1.1.2.3 2021-09-20 [3] CRAN (R 4.1.2)
GlobalOptions 0.1.2 2020-06-10 [3] CRAN (R 4.1.2)
globals 0.14.0 2020-11-22 [5] CRAN (R 4.1.2)
glue 1.6.2 2022-02-24 [1] CRAN (R 4.1.2)
goftest 1.2-3 2021-10-07 [5] CRAN (R 4.1.2)
gplots 3.1.1 2020-11-28 [5] CRAN (R 4.1.2)
gridExtra 2.3 2017-09-09 [5] CRAN (R 4.1.2)
grr 0.9.5 2016-08-26 [1] CRAN (R 4.1.2)
gtable 0.3.0 2019-03-25 [5] CRAN (R 4.1.2)
gtools 3.9.2 2021-06-06 [5] CRAN (R 4.1.2)
haven 2.4.3 2021-08-04 [5] CRAN (R 4.1.2)
HDF5Array 1.22.1 2021-11-14 [3] Bioconductor
hexbin 1.28.2 2021-01-08 [5] CRAN (R 4.1.2)
highr 0.9 2021-04-16 [5] CRAN (R 4.1.2)
hms 1.1.1 2021-09-26 [5] CRAN (R 4.1.2)
htmltools 0.5.2 2021-08-25 [5] CRAN (R 4.1.2)
htmlwidgets 1.5.4 2021-09-08 [5] CRAN (R 4.1.2)
httpuv 1.6.3 2021-09-09 [5] CRAN (R 4.1.2)
httr 1.4.2 2020-07-20 [5] CRAN (R 4.1.2)
ica 1.0-2 2018-05-24 [5] CRAN (R 4.1.2)
igraph * 1.2.11 2022-01-04 [1] CRAN (R 4.1.2)
insight 0.16.0 2022-02-17 [1] CRAN (R 4.1.2)
IRanges * 2.28.0 2021-10-26 [3] Bioconductor
irlba 2.3.5 2021-12-06 [5] CRAN (R 4.1.2)
iterators 1.0.13 2020-10-15 [5] CRAN (R 4.1.2)
janitor 2.1.0 2021-01-05 [2] CRAN (R 4.1.2)
jquerylib 0.1.4 2021-04-26 [5] CRAN (R 4.1.2)
jsonlite 1.8.0 2022-02-22 [1] CRAN (R 4.1.2)
KEGGREST 1.34.0 2021-10-26 [3] Bioconductor
KernSmooth 2.23-20 2021-05-03 [5] CRAN (R 4.1.2)
knitr 1.37 2021-12-16 [1] CRAN (R 4.1.2)
labeling 0.4.2 2020-10-20 [5] CRAN (R 4.1.2)
later 1.3.0 2021-08-18 [5] CRAN (R 4.1.2)
lattice 0.20-45 2021-09-22 [5] CRAN (R 4.1.2)
lazyeval 0.2.2 2019-03-15 [5] CRAN (R 4.1.2)
leiden 0.3.9 2021-07-27 [5] CRAN (R 4.1.2)
lifecycle 1.0.1 2021-09-24 [5] CRAN (R 4.1.2)
limma * 3.50.0 2021-10-26 [3] Bioconductor
listenv 0.8.0 2019-12-05 [5] CRAN (R 4.1.2)
lme4 1.1-27.1 2021-06-22 [5] CRAN (R 4.1.2)
lmerTest 3.1-3 2020-10-23 [3] CRAN (R 4.1.2)
lmtest 0.9-39 2021-11-07 [5] CRAN (R 4.1.2)
locfit 1.5-9.4 2020-03-25 [5] CRAN (R 4.1.2)
lubridate 1.8.0 2021-10-07 [5] CRAN (R 4.1.2)
magick 2.7.3 2021-08-18 [2] CRAN (R 4.1.2)
magrittr * 2.0.2 2022-01-26 [1] CRAN (R 4.1.2)
MASS * 7.3-54 2021-05-03 [5] CRAN (R 4.1.2)
Matrix * 1.3-4 2021-06-01 [5] CRAN (R 4.1.2)
Matrix.utils * 0.9.8 2020-02-26 [1] CRAN (R 4.1.2)
MatrixGenerics * 1.6.0 2021-10-26 [3] Bioconductor
matrixStats * 0.61.0 2021-09-17 [5] CRAN (R 4.1.2)
memoise 2.0.1 2021-11-26 [5] CRAN (R 4.1.2)
mgcv 1.8-38 2021-10-06 [5] CRAN (R 4.1.2)
microbiome 1.16.0 2021-10-26 [3] Bioconductor
mime 0.12 2021-09-28 [5] CRAN (R 4.1.2)
miniUI 0.1.1.1 2018-05-18 [5] CRAN (R 4.1.2)
minqa 1.2.4 2014-10-09 [5] CRAN (R 4.1.2)
modelr 0.1.8 2020-05-19 [5] CRAN (R 4.1.2)
MOFA2 * 1.4.0 2021-10-26 [1] Bioconductor
multcomp 1.4-17 2021-04-29 [5] CRAN (R 4.1.2)
multtest 2.50.0 2021-10-26 [3] Bioconductor
munsell 0.5.0 2018-06-12 [5] CRAN (R 4.1.2)
muscat * 1.8.0 2021-10-26 [3] Bioconductor
mvtnorm 1.1-3 2021-10-08 [5] CRAN (R 4.1.2)
N2R 1.0.1 2022-01-18 [1] CRAN (R 4.1.2)
nlme 3.1-153 2021-09-07 [5] CRAN (R 4.1.2)
nloptr 1.2.2.3 2021-11-02 [5] CRAN (R 4.1.2)
numDeriv 2016.8-1.1 2019-06-06 [5] CRAN (R 4.1.2)
pagoda2 * 1.0.9 2022-03-02 [1] CRAN (R 4.1.2)
parallelly 1.29.0 2021-11-21 [5] CRAN (R 4.1.2)
patchwork * 1.1.0.9000 2022-03-23 [1] Github (thomasp85/patchwork@79223d3)
pbapply 1.5-0 2021-09-16 [5] CRAN (R 4.1.2)
pbkrtest 0.5.1 2021-03-09 [5] CRAN (R 4.1.2)
performance * 0.8.0 2021-10-01 [1] CRAN (R 4.1.2)
permute 0.9-5 2019-03-12 [3] CRAN (R 4.1.2)
pheatmap 1.0.12 2019-01-04 [3] CRAN (R 4.1.2)
phyloseq * 1.38.0 2021-10-26 [3] Bioconductor
pillar 1.7.0 2022-02-01 [1] CRAN (R 4.1.2)
pkgbuild 1.2.1 2021-11-30 [5] CRAN (R 4.1.2)
pkgconfig 2.0.3 2019-09-22 [5] CRAN (R 4.1.2)
pkgload 1.2.4 2021-11-30 [5] CRAN (R 4.1.2)
plotly 4.10.0 2021-10-09 [5] CRAN (R 4.1.2)
plyr 1.8.6 2020-03-03 [5] CRAN (R 4.1.2)
png 0.1-7 2013-12-03 [5] CRAN (R 4.1.2)
polyclip 1.10-0 2019-03-14 [5] CRAN (R 4.1.2)
prettyunits 1.1.1 2020-01-24 [5] CRAN (R 4.1.2)
processx 3.5.2 2021-04-30 [5] CRAN (R 4.1.2)
progress 1.2.2 2019-05-16 [5] CRAN (R 4.1.2)
promises 1.2.0.1 2021-02-11 [5] CRAN (R 4.1.2)
ps 1.6.0 2021-02-28 [5] CRAN (R 4.1.2)
purrr * 0.3.4 2020-04-17 [5] CRAN (R 4.1.2)
R.methodsS3 1.8.1 2020-08-26 [5] CRAN (R 4.1.2)
R.oo 1.24.0 2020-08-26 [5] CRAN (R 4.1.2)
R.utils 2.11.0 2021-09-26 [5] CRAN (R 4.1.2)
R6 2.5.1 2021-08-19 [5] CRAN (R 4.1.2)
RANN 2.6.1 2019-01-08 [5] CRAN (R 4.1.2)
rappdirs 0.3.3 2021-01-31 [5] CRAN (R 4.1.2)
rbibutils 2.2.7 2021-12-07 [5] CRAN (R 4.1.2)
RColorBrewer * 1.1-2 2014-12-07 [5] CRAN (R 4.1.2)
Rcpp 1.0.8.3 2022-03-17 [1] CRAN (R 4.1.2)
RcppAnnoy 0.0.19 2021-07-30 [5] CRAN (R 4.1.2)
RCurl 1.98-1.6 2022-02-08 [1] CRAN (R 4.1.2)
Rdpack 2.1.3 2021-12-08 [5] CRAN (R 4.1.2)
readr * 2.1.1 2021-11-30 [5] CRAN (R 4.1.2)
readxl * 1.3.1 2019-03-13 [5] CRAN (R 4.1.2)
registry 0.5-1 2019-03-05 [5] CRAN (R 4.1.2)
remotes 2.4.2 2021-11-30 [5] CRAN (R 4.1.2)
reprex 2.0.1 2021-08-05 [5] CRAN (R 4.1.2)
reshape2 * 1.4.4 2020-04-09 [5] CRAN (R 4.1.2)
restfulr 0.0.13 2017-08-06 [3] CRAN (R 4.1.2)
reticulate * 1.22 2021-09-17 [5] CRAN (R 4.1.2)
rhdf5 2.38.0 2021-10-26 [3] Bioconductor
rhdf5filters 1.6.0 2021-10-26 [3] Bioconductor
Rhdf5lib 1.16.0 2021-10-26 [3] Bioconductor
rjson 0.2.20 2018-06-08 [5] CRAN (R 4.1.2)
rlang 1.0.2 2022-03-04 [1] CRAN (R 4.1.2)
rmarkdown * 2.13 2022-03-10 [1] CRAN (R 4.1.2)
RMTstat 0.3 2014-11-01 [1] CRAN (R 4.1.2)
ROCR 1.0-11 2020-05-02 [5] CRAN (R 4.1.2)
Rook 1.1-1 2014-10-20 [1] CRAN (R 4.1.2)
rpart 4.1-15 2019-04-12 [5] CRAN (R 4.1.2)
rprojroot 2.0.2 2020-11-15 [5] CRAN (R 4.1.2)
Rsamtools 2.10.0 2021-10-26 [3] Bioconductor
RSQLite 2.2.9 2021-12-06 [5] CRAN (R 4.1.2)
rstudioapi 0.13 2020-11-12 [5] CRAN (R 4.1.2)
rsvd 1.0.5 2021-04-16 [5] CRAN (R 4.1.2)
rtracklayer * 1.54.0 2021-10-26 [3] Bioconductor
Rtsne 0.15 2018-11-10 [5] CRAN (R 4.1.2)
rvest 1.0.2 2021-10-16 [5] CRAN (R 4.1.2)
S4Vectors * 0.32.3 2021-11-21 [3] Bioconductor
sandwich 3.0-1 2021-05-18 [5] CRAN (R 4.1.2)
sass 0.4.0 2021-05-12 [5] CRAN (R 4.1.2)
ScaledMatrix 1.2.0 2021-10-26 [3] Bioconductor
scales * 1.1.1 2020-05-11 [5] CRAN (R 4.1.2)
scater * 1.22.0 2021-10-26 [3] Bioconductor
scattermore 0.7 2020-11-24 [5] CRAN (R 4.1.2)
sccore 1.0.1 2021-12-12 [1] CRAN (R 4.1.2)
sctransform 0.3.2 2020-12-16 [5] CRAN (R 4.1.2)
scuttle * 1.4.0 2021-10-26 [3] Bioconductor
seriation * 1.3.1 2021-10-16 [3] CRAN (R 4.1.2)
sessioninfo 1.2.2 2021-12-06 [5] CRAN (R 4.1.2)
Seurat * 4.0.5 2021-10-17 [5] CRAN (R 4.1.2)
SeuratObject * 4.0.4 2021-11-23 [5] CRAN (R 4.1.2)
shape 1.4.6 2021-05-19 [3] CRAN (R 4.1.2)
shiny 1.7.1 2021-10-02 [5] CRAN (R 4.1.2)
SingleCellExperiment * 1.16.0 2021-10-26 [3] Bioconductor
snakecase 0.11.0 2019-05-25 [2] CRAN (R 4.1.2)
sparseMatrixStats 1.6.0 2021-10-26 [3] Bioconductor
spatstat.core 2.3-2 2021-11-26 [5] CRAN (R 4.1.2)
spatstat.data 2.1-0 2021-03-21 [5] CRAN (R 4.1.2)
spatstat.geom 2.3-0 2021-10-09 [5] CRAN (R 4.1.2)
spatstat.sparse 2.0-0 2021-03-16 [5] CRAN (R 4.1.2)
spatstat.utils 2.2-0 2021-06-14 [5] CRAN (R 4.1.2)
stringi 1.7.6 2021-11-29 [5] CRAN (R 4.1.2)
stringr * 1.4.0 2019-02-10 [5] CRAN (R 4.1.2)
SummarizedExperiment * 1.24.0 2021-10-26 [3] Bioconductor
survival 3.2-13 2021-08-24 [5] CRAN (R 4.1.2)
tensor 1.5 2012-05-05 [5] CRAN (R 4.1.2)
testthat 3.1.1 2021-12-03 [5] CRAN (R 4.1.2)
TH.data 1.1-0 2021-09-27 [5] CRAN (R 4.1.2)
tibble * 3.1.6 2021-11-07 [5] CRAN (R 4.1.2)
tictoc * 1.0.1 2021-04-19 [1] CRAN (R 4.1.2)
tidyr * 1.1.4 2021-09-27 [5] CRAN (R 4.1.2)
tidyselect 1.1.1 2021-04-30 [5] CRAN (R 4.1.2)
tidyverse * 1.3.1 2021-04-15 [5] CRAN (R 4.1.2)
TMB 1.7.22 2021-09-28 [3] CRAN (R 4.1.2)
triebeard 0.3.0 2016-08-04 [2] CRAN (R 4.1.2)
TSP 1.1-11 2021-10-06 [3] CRAN (R 4.1.2)
tzdb 0.2.0 2021-10-27 [5] CRAN (R 4.1.2)
UpSetR * 1.4.0 2019-05-22 [1] CRAN (R 4.1.2)
urltools 1.7.3 2019-04-14 [2] CRAN (R 4.1.2)
usethis 2.1.3 2021-10-27 [5] CRAN (R 4.1.2)
utf8 1.2.2 2021-07-24 [5] CRAN (R 4.1.2)
uwot 0.1.11 2021-12-02 [5] CRAN (R 4.1.2)
variancePartition 1.24.0 2021-10-26 [3] Bioconductor
vctrs 0.3.8 2021-04-29 [5] CRAN (R 4.1.2)
vegan 2.5-7 2020-11-28 [3] CRAN (R 4.1.2)
vipor 0.4.5 2017-03-22 [3] CRAN (R 4.1.2)
viridis * 0.6.2 2021-10-13 [5] CRAN (R 4.1.2)
viridisLite * 0.4.0 2021-04-13 [5] CRAN (R 4.1.2)
whisker 0.4 2019-08-28 [5] CRAN (R 4.1.2)
withr 2.5.0 2022-03-03 [1] CRAN (R 4.1.2)
workflowr 1.7.0 2021-12-21 [1] CRAN (R 4.1.2)
writexl * 1.4.0 2021-04-20 [1] CRAN (R 4.1.2)
xfun 0.30 2022-03-02 [1] CRAN (R 4.1.2)
XML 3.99-0.8 2021-09-17 [5] CRAN (R 4.1.2)
xml2 1.3.3 2021-11-30 [5] CRAN (R 4.1.2)
xtable 1.8-4 2019-04-21 [5] CRAN (R 4.1.2)
XVector 0.34.0 2021-10-26 [3] Bioconductor
yaml 2.3.5 2022-02-21 [1] CRAN (R 4.1.2)
zlibbioc 1.40.0 2021-10-26 [3] Bioconductor
zoo 1.8-9 2021-03-09 [5] CRAN (R 4.1.2)
[1] /gpfs/homefs/global/home/macnairw/R/x86_64-pc-linux-gnu-library/4.1.2-foss
[2] /apps/rocs/2020.08/cascadelake/software/R-Roche-bundle/2021.12-foss-2020a-R-4.1.2
[3] /apps/rocs/2020.08/cascadelake/software/R-bundle-Bioconductor/3.14-foss-2020a-R-4.1.2
[4] /apps/rocs/2020.08/cascadelake/software/ncdf4/1.18-foss-2020a-R-4.1.2
[5] /apps/rocs/2020.08/cascadelake/software/R/4.1.2-foss-2020a/lib64/R/library
------------------------------------------------------------------------------
sessionInfo()R version 4.1.2 (2021-11-01)
Platform: x86_64-pc-linux-gnu (64-bit)
Running under: Red Hat Enterprise Linux 8.2 (Ootpa)
Matrix products: default
BLAS/LAPACK: /apps/rocs/2020.08/cascadelake/software/OpenBLAS/0.3.9-GCC-9.3.0/lib/libopenblas_skylakexp-r0.3.9.so
locale:
[1] LC_CTYPE=C LC_NUMERIC=C
[3] LC_TIME=en_US.UTF-8 LC_COLLATE=en_US.UTF-8
[5] LC_MONETARY=en_US.UTF-8 LC_MESSAGES=en_US.UTF-8
[7] LC_PAPER=en_US.UTF-8 LC_NAME=C
[9] LC_ADDRESS=C LC_TELEPHONE=C
[11] LC_MEASUREMENT=en_US.UTF-8 LC_IDENTIFICATION=C
attached base packages:
[1] grid stats4 stats graphics grDevices utils datasets
[8] methods base
other attached packages:
[1] MOFA2_1.4.0 rmarkdown_2.13
[3] tictoc_1.0.1 performance_0.8.0
[5] edgeR_3.36.0 limma_3.50.0
[7] reshape2_1.4.4 scater_1.22.0
[9] scuttle_1.4.0 Matrix.utils_0.9.8
[11] UpSetR_1.4.0 muscat_1.8.0
[13] dplyr_1.0.7 readr_2.1.1
[15] tidyr_1.1.4 tibble_3.1.6
[17] tidyverse_1.3.1 rtracklayer_1.54.0
[19] ggbeeswarm_0.6.0 ggrepel_0.9.1
[21] MASS_7.3-54 phyloseq_1.38.0
[23] ANCOMBC_1.4.0 patchwork_1.1.0.9000
[25] writexl_1.4.0 reticulate_1.22
[27] fgsea_1.20.0 BiocParallel_1.28.3
[29] ggplot.multistats_1.0.0 seriation_1.3.1
[31] ComplexHeatmap_2.10.0 pagoda2_1.0.9
[33] igraph_1.2.11 SeuratObject_4.0.4
[35] Seurat_4.0.5 future_1.23.0
[37] Matrix_1.3-4 SingleCellExperiment_1.16.0
[39] SummarizedExperiment_1.24.0 Biobase_2.54.0
[41] GenomicRanges_1.46.1 GenomeInfoDb_1.30.1
[43] IRanges_2.28.0 S4Vectors_0.32.3
[45] BiocGenerics_0.40.0 MatrixGenerics_1.6.0
[47] matrixStats_0.61.0 purrr_0.3.4
[49] readxl_1.3.1 forcats_0.5.1
[51] ggplot2_3.3.5 scales_1.1.1
[53] viridis_0.6.2 viridisLite_0.4.0
[55] assertthat_0.2.1 stringr_1.4.0
[57] data.table_1.14.2 magrittr_2.0.2
[59] circlize_0.4.13 RColorBrewer_1.1-2
[61] BiocStyle_2.22.0
loaded via a namespace (and not attached):
[1] rsvd_1.0.5 ica_1.0-2
[3] ps_1.6.0 Rsamtools_2.10.0
[5] foreach_1.5.1 lmtest_0.9-39
[7] rprojroot_2.0.2 crayon_1.5.0
[9] spatstat.core_2.3-2 rbibutils_2.2.7
[11] rhdf5filters_1.6.0 nlme_3.1-153
[13] backports_1.4.0 reprex_2.0.1
[15] basilisk_1.6.0 rlang_1.0.2
[17] XVector_0.34.0 ROCR_1.0-11
[19] microbiome_1.16.0 irlba_2.3.5
[21] callr_3.7.0 nloptr_1.2.2.3
[23] filelock_1.0.2 rjson_0.2.20
[25] bit64_4.0.5 glue_1.6.2
[27] pheatmap_1.0.12 sctransform_0.3.2
[29] processx_3.5.2 pbkrtest_0.5.1
[31] parallel_4.1.2 vipor_0.4.5
[33] spatstat.sparse_2.0-0 AnnotationDbi_1.56.2
[35] spatstat.geom_2.3-0 haven_2.4.3
[37] tidyselect_1.1.1 usethis_2.1.3
[39] fitdistrplus_1.1-6 variancePartition_1.24.0
[41] XML_3.99-0.8 zoo_1.8-9
[43] GenomicAlignments_1.30.0 xtable_1.8-4
[45] evaluate_0.15 Rdpack_2.1.3
[47] cli_3.2.0 zlibbioc_1.40.0
[49] rstudioapi_0.13 miniUI_0.1.1.1
[51] whisker_0.4 bslib_0.3.1
[53] rpart_4.1-15 fastmatch_1.1-3
[55] shiny_1.7.1 BiocSingular_1.10.0
[57] xfun_0.30 clue_0.3-60
[59] pkgbuild_1.2.1 multtest_2.50.0
[61] cluster_2.1.2 caTools_1.18.2
[63] TSP_1.1-11 biomformat_1.22.0
[65] KEGGREST_1.34.0 ape_5.5
[67] listenv_0.8.0 Biostrings_2.62.0
[69] png_0.1-7 permute_0.9-5
[71] withr_2.5.0 bitops_1.0-7
[73] plyr_1.8.6 cellranger_1.1.0
[75] coda_0.19-4 pillar_1.7.0
[77] gplots_3.1.1 GlobalOptions_0.1.2
[79] cachem_1.0.6 multcomp_1.4-17
[81] fs_1.5.1 GetoptLong_1.0.5
[83] DelayedMatrixStats_1.16.0 vctrs_0.3.8
[85] ellipsis_0.3.2 generics_0.1.1
[87] devtools_2.4.3 urltools_1.7.3
[89] tools_4.1.2 beeswarm_0.4.0
[91] munsell_0.5.0 emmeans_1.7.1-1
[93] DelayedArray_0.20.0 pkgload_1.2.4
[95] fastmap_1.1.0 compiler_4.1.2
[97] abind_1.4-5 httpuv_1.6.3
[99] sessioninfo_1.2.2 plotly_4.10.0
[101] GenomeInfoDbData_1.2.7 gridExtra_2.3
[103] glmmTMB_1.1.2.3 workflowr_1.7.0
[105] dir.expiry_1.2.0 lattice_0.20-45
[107] deldir_1.0-6 utf8_1.2.2
[109] later_1.3.0 jsonlite_1.8.0
[111] ScaledMatrix_1.2.0 dendsort_0.3.4
[113] sparseMatrixStats_1.6.0 pbapply_1.5-0
[115] estimability_1.3 genefilter_1.76.0
[117] lazyeval_0.2.2 promises_1.2.0.1
[119] doParallel_1.0.16 R.utils_2.11.0
[121] goftest_1.2-3 spatstat.utils_2.2-0
[123] brew_1.0-6 sandwich_3.0-1
[125] cowplot_1.1.1 blme_1.0-5
[127] Rtsne_0.15 uwot_0.1.11
[129] HDF5Array_1.22.1 Rook_1.1-1
[131] survival_3.2-13 numDeriv_2016.8-1.1
[133] yaml_2.3.5 htmltools_0.5.2
[135] memoise_2.0.1 BiocIO_1.4.0
[137] locfit_1.5-9.4 digest_0.6.29
[139] mime_0.12 rappdirs_0.3.3
[141] registry_0.5-1 N2R_1.0.1
[143] RSQLite_2.2.9 future.apply_1.8.1
[145] remotes_2.4.2 blob_1.2.2
[147] vegan_2.5-7 R.oo_1.24.0
[149] drat_0.2.2 labeling_0.4.2
[151] splines_4.1.2 Rhdf5lib_1.16.0
[153] RCurl_1.98-1.6 broom_0.7.10
[155] hms_1.1.1 modelr_0.1.8
[157] rhdf5_2.38.0 colorspace_2.0-3
[159] BiocManager_1.30.16 shape_1.4.6
[161] sass_0.4.0 Rcpp_1.0.8.3
[163] RANN_2.6.1 mvtnorm_1.1-3
[165] fansi_1.0.2 tzdb_0.2.0
[167] parallelly_1.29.0 R6_2.5.1
[169] ggridges_0.5.3 lifecycle_1.0.1
[171] minqa_1.2.4 testthat_3.1.1
[173] leiden_0.3.9 jquerylib_0.1.4
[175] snakecase_0.11.0 desc_1.4.0
[177] RcppAnnoy_0.0.19 TH.data_1.1-0
[179] iterators_1.0.13 TMB_1.7.22
[181] htmlwidgets_1.5.4 beachmat_2.10.0
[183] polyclip_1.10-0 triebeard_0.3.0
[185] RMTstat_0.3 rvest_1.0.2
[187] mgcv_1.8-38 globals_0.14.0
[189] insight_0.16.0 codetools_0.2-18
[191] lubridate_1.8.0 gtools_3.9.2
[193] prettyunits_1.1.1 dbplyr_2.1.1
[195] basilisk.utils_1.6.0 R.methodsS3_1.8.1
[197] gtable_0.3.0 DBI_1.1.1
[199] git2r_0.29.0 tensor_1.5
[201] httr_1.4.2 highr_0.9
[203] KernSmooth_2.23-20 stringi_1.7.6
[205] progress_1.2.2 farver_2.1.0
[207] annotate_1.72.0 hexbin_1.28.2
[209] magick_2.7.3 xml2_1.3.3
[211] sccore_1.0.1 grr_0.9.5
[213] boot_1.3-28 BiocNeighbors_1.12.0
[215] lme4_1.1-27.1 restfulr_0.0.13
[217] ade4_1.7-18 geneplotter_1.72.0
[219] scattermore_0.7 DESeq2_1.34.0
[221] bit_4.0.4 spatstat.data_2.1-0
[223] janitor_2.1.0 pkgconfig_2.0.3
[225] lmerTest_3.1-3 corrplot_0.92
[227] knitr_1.37